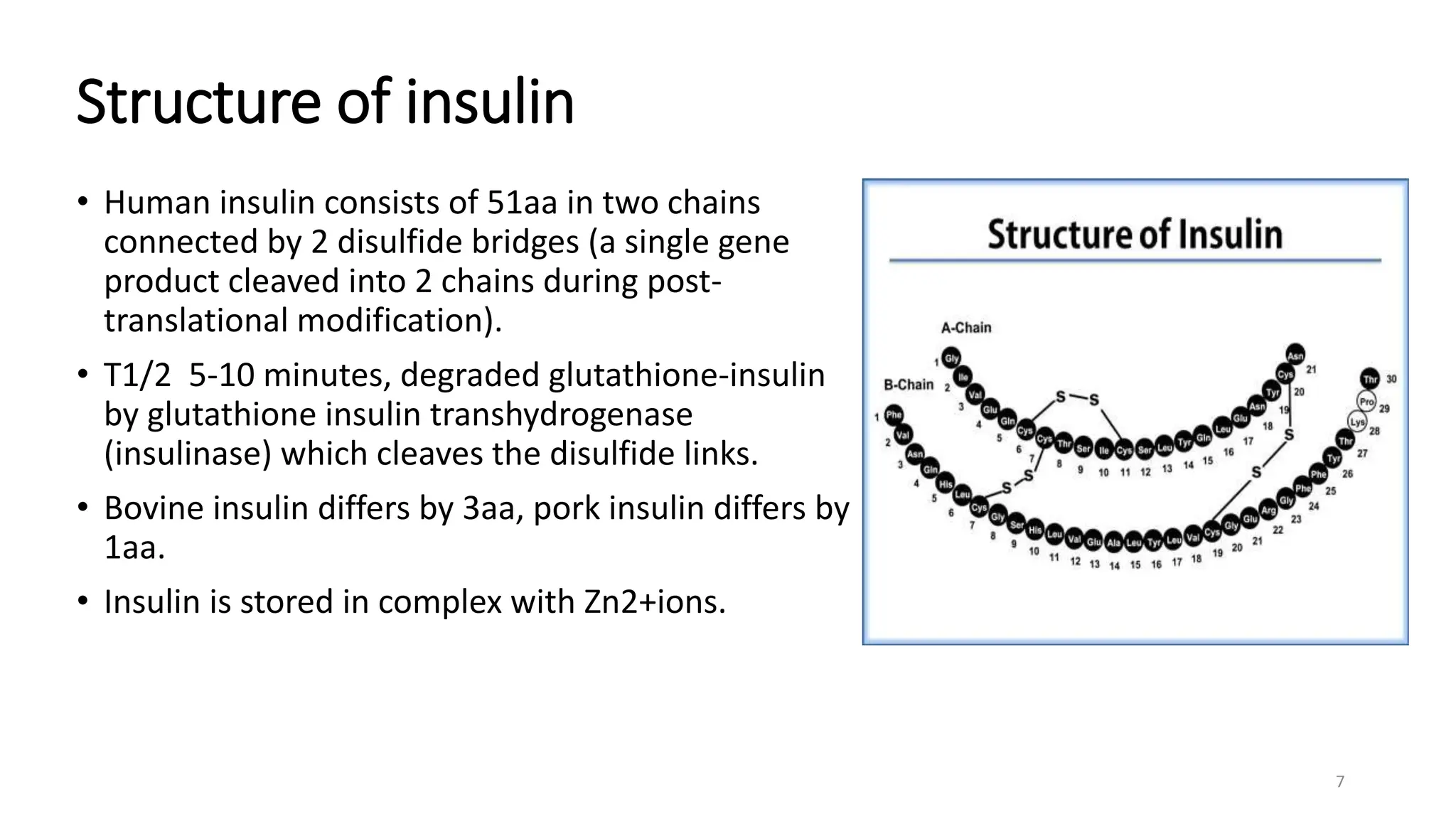
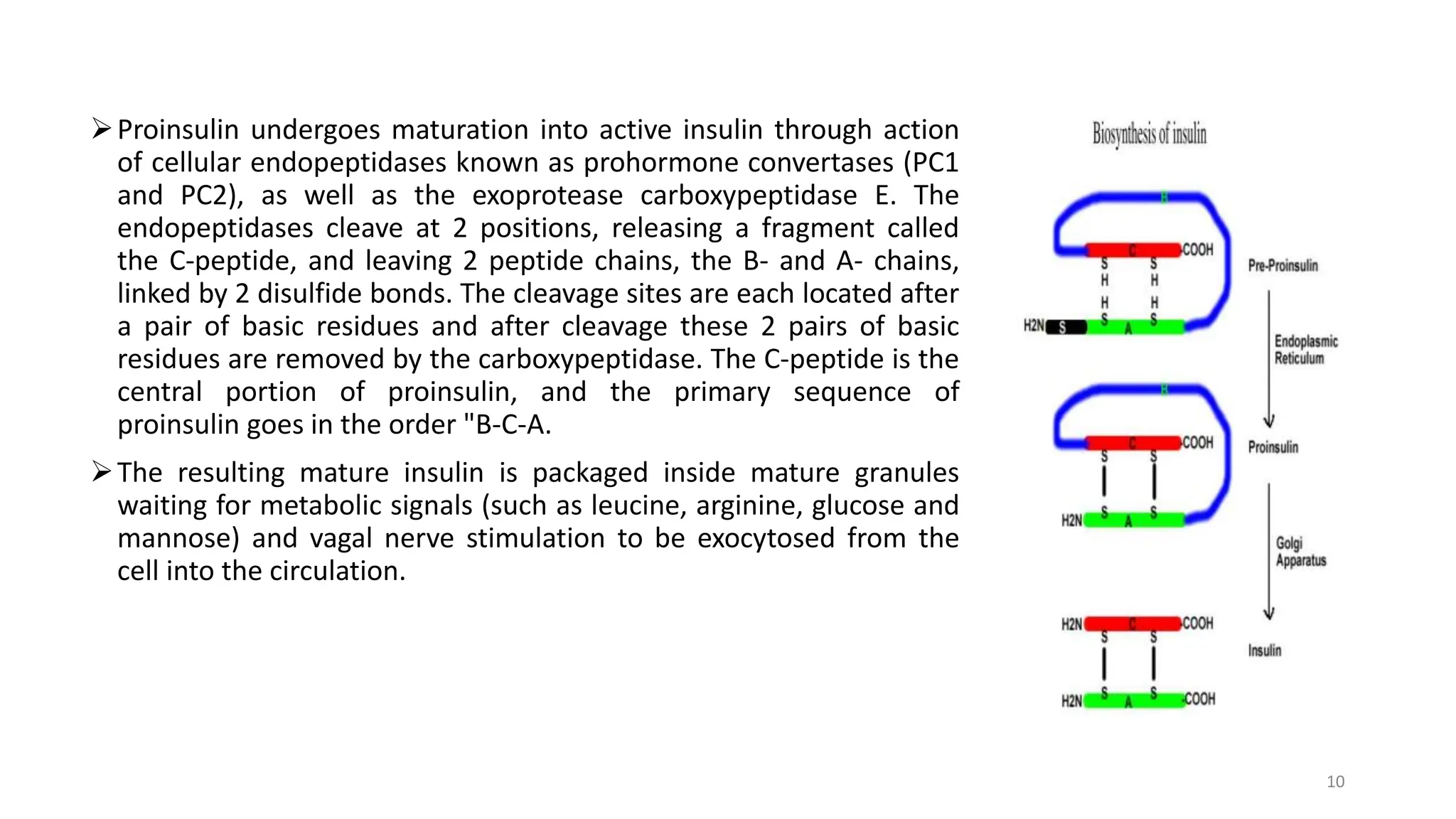
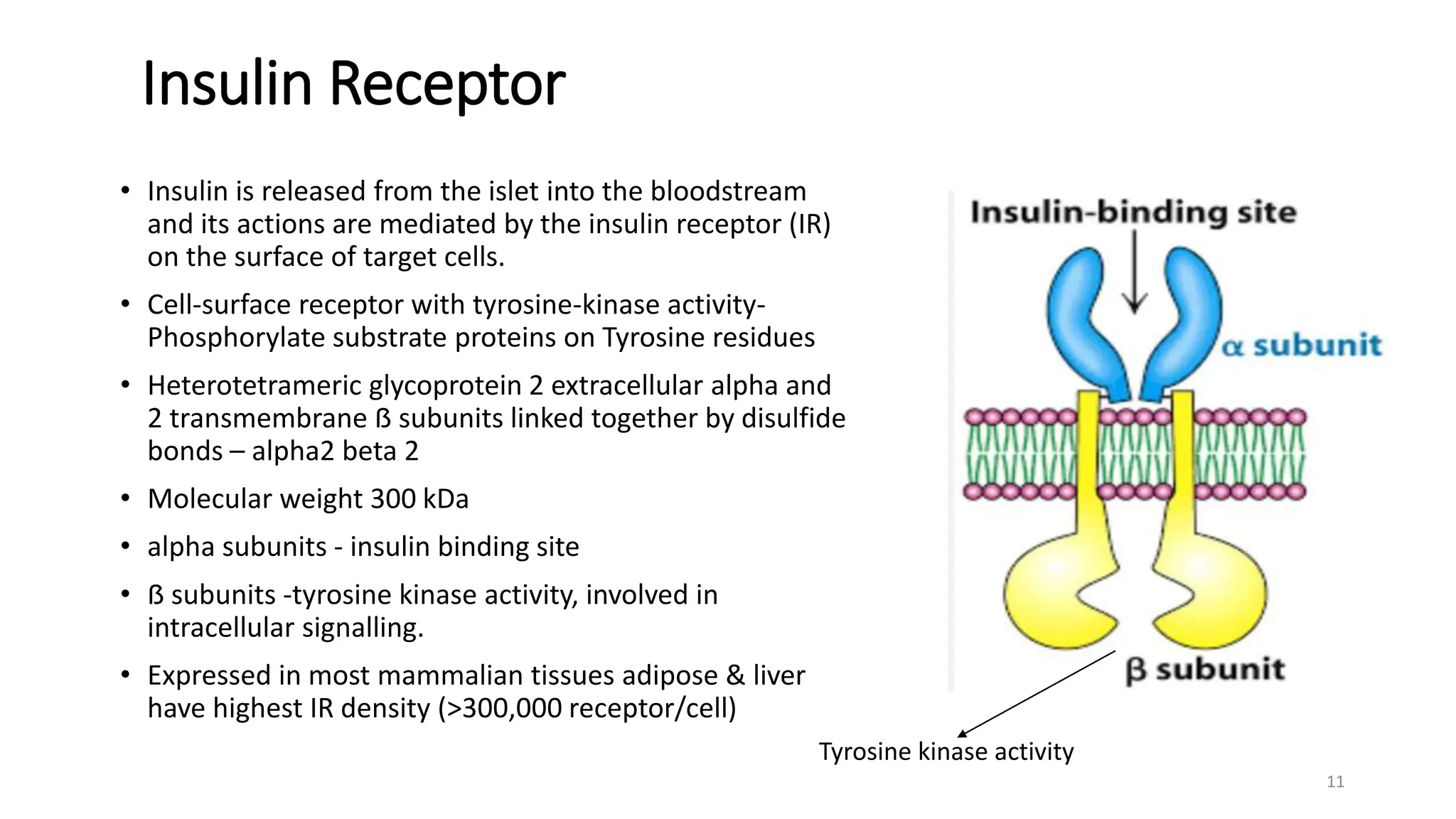
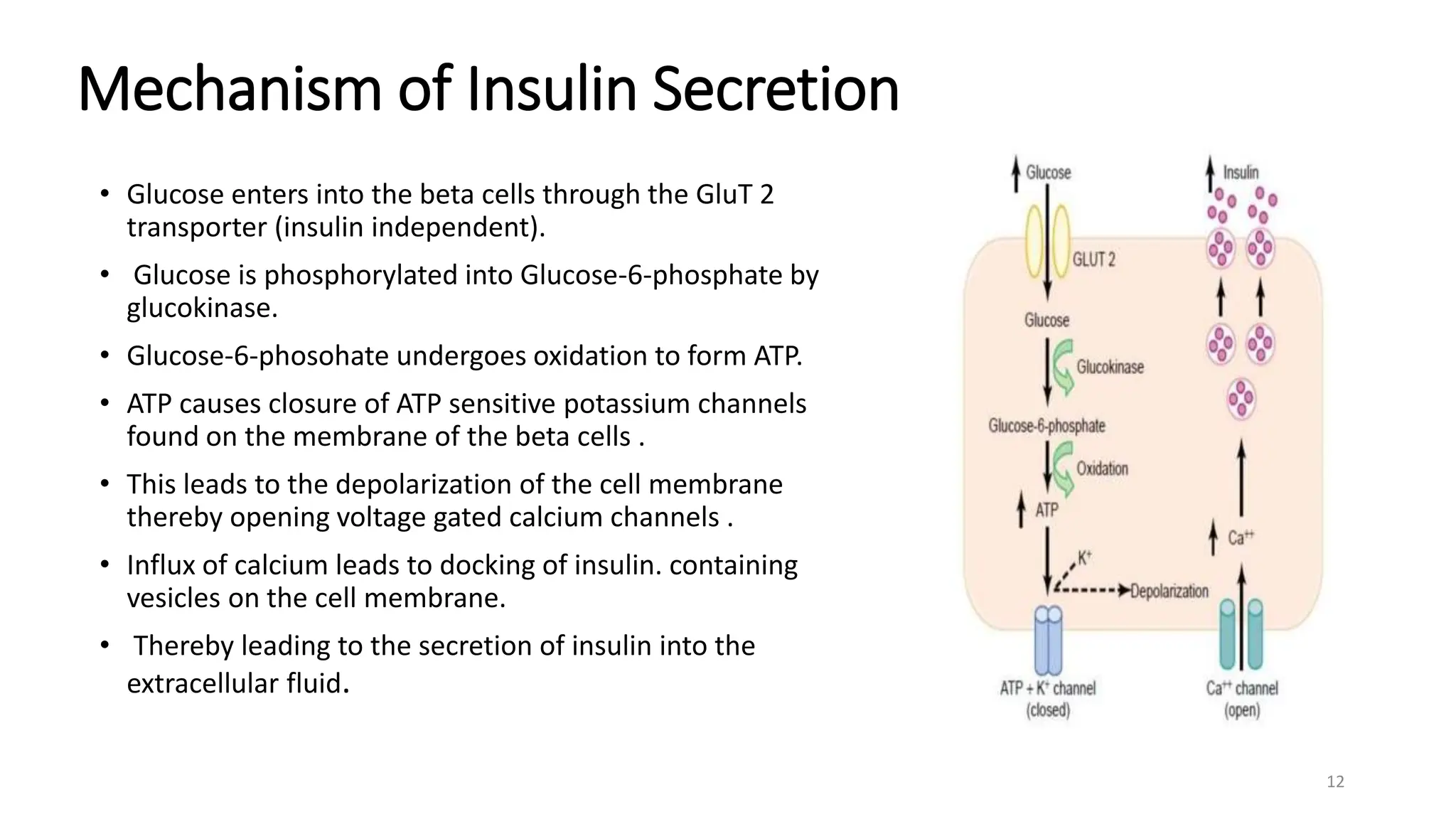
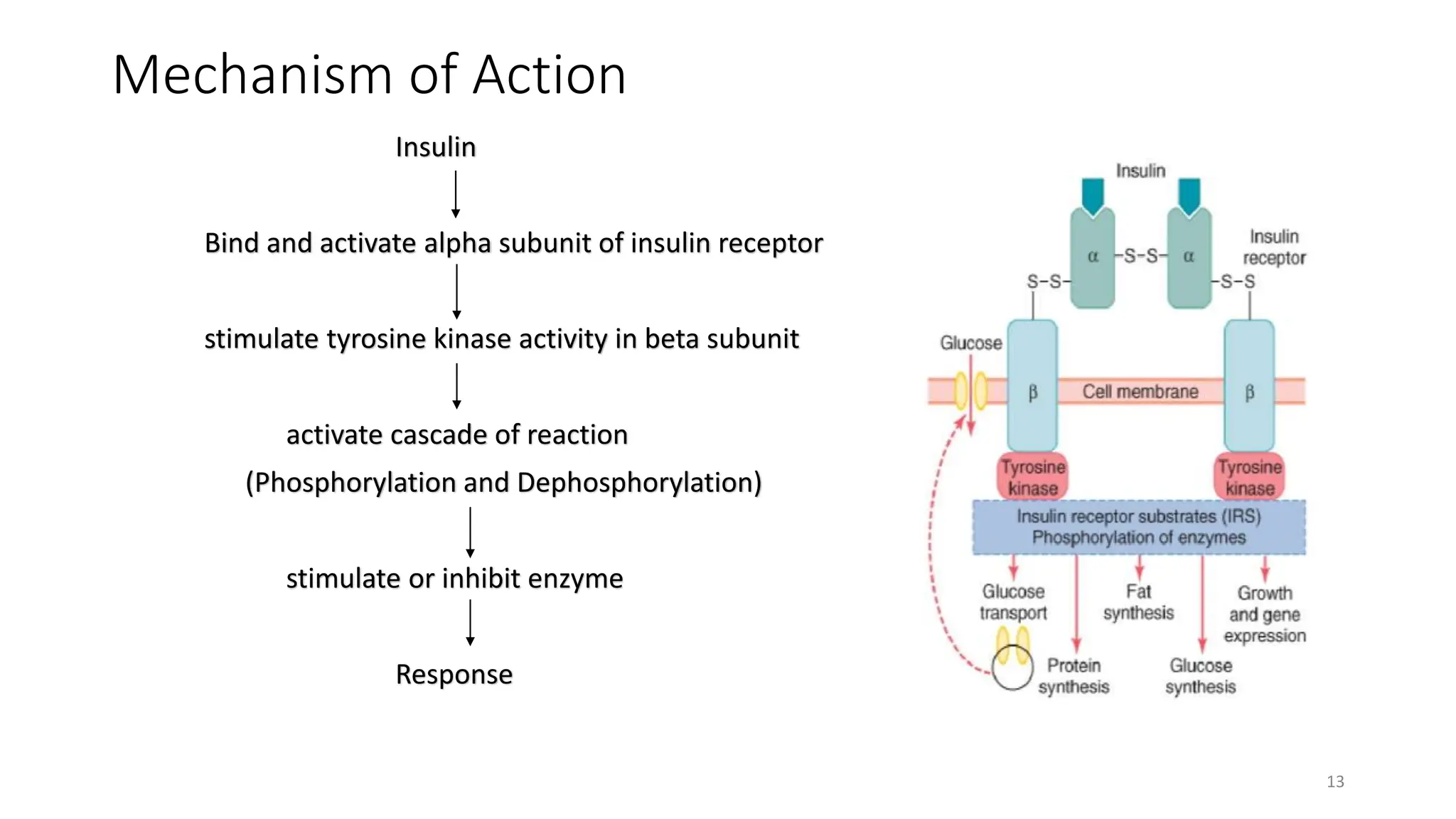
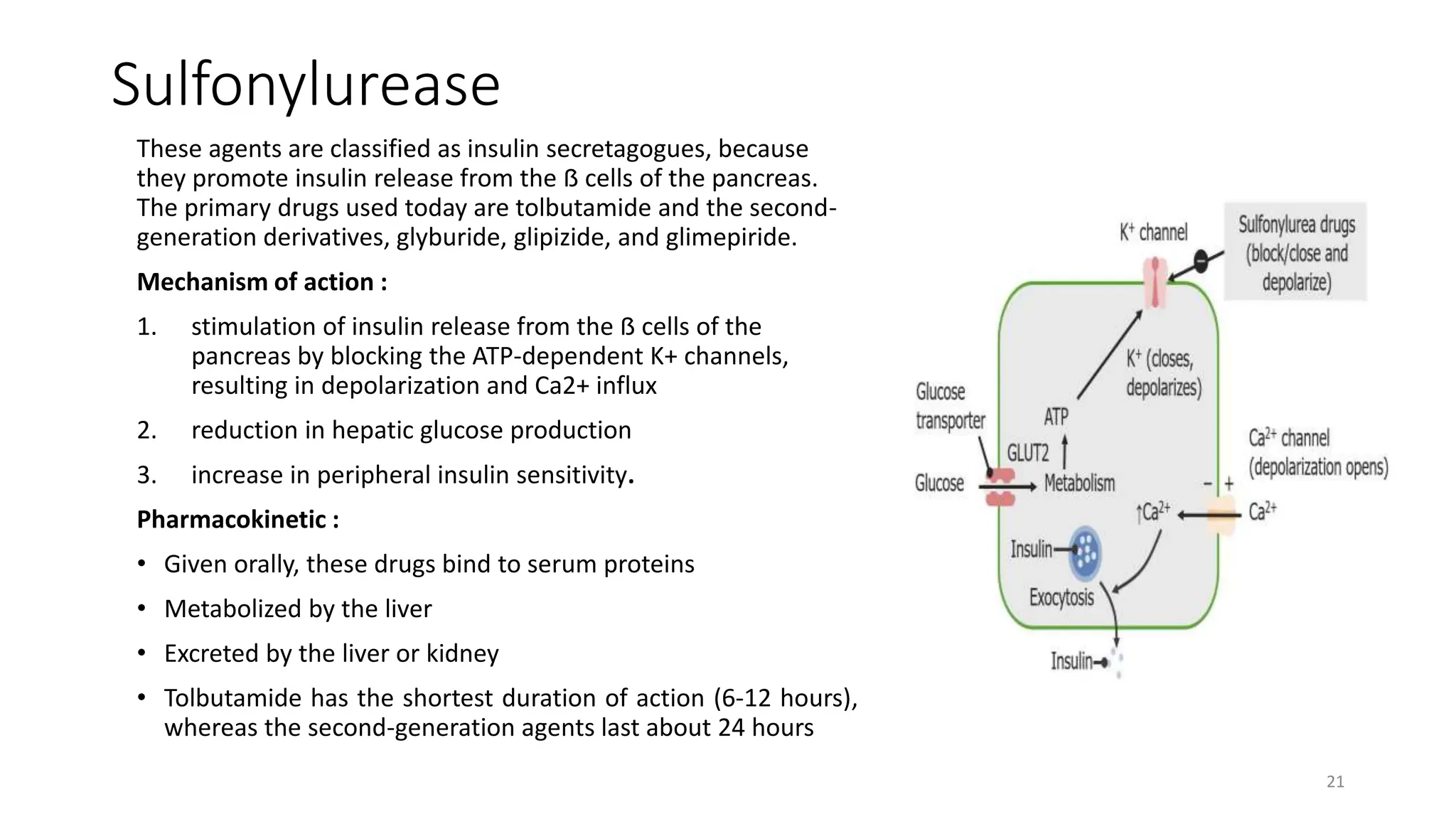
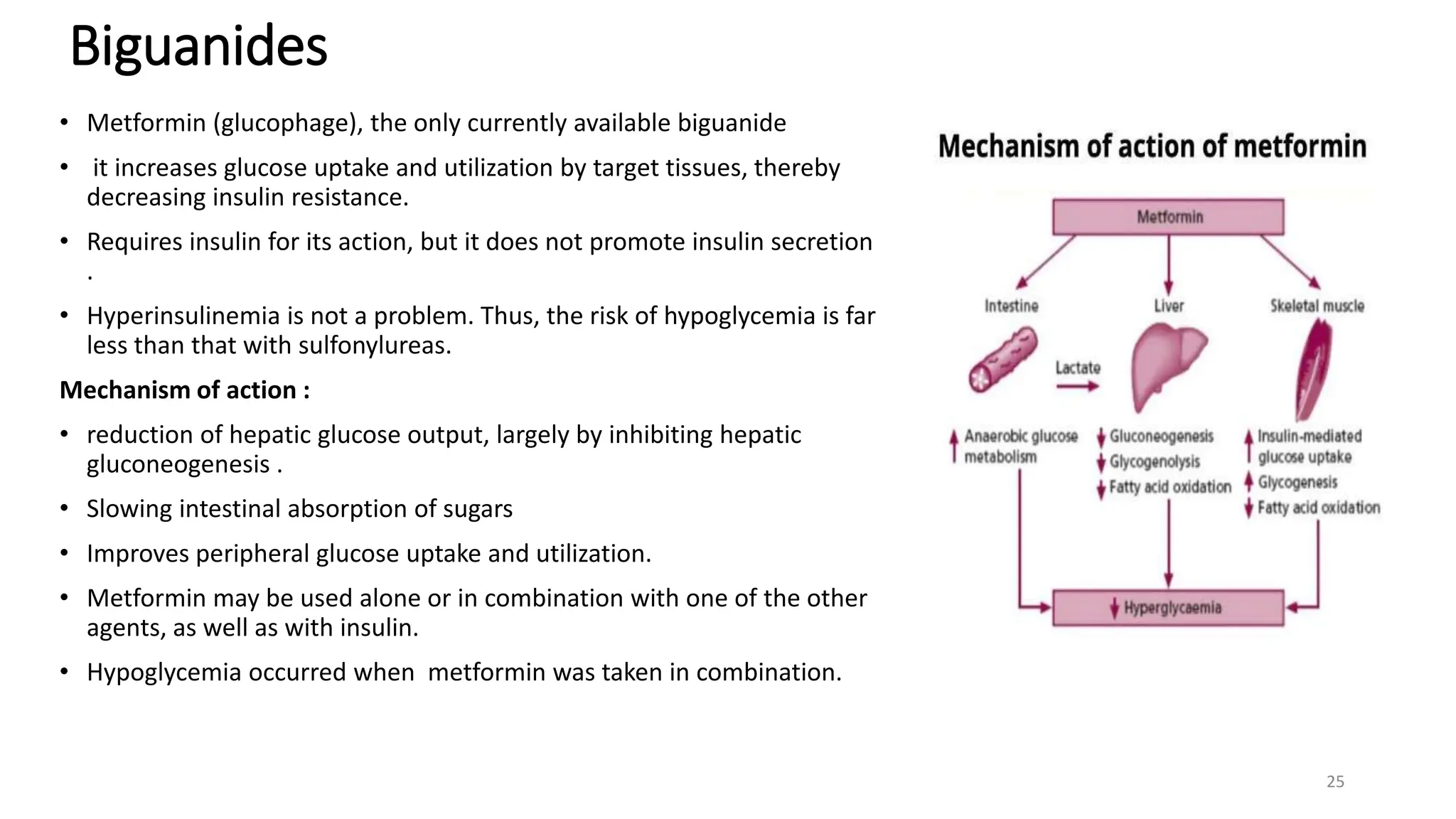
The document discusses diabetes mellitus, its types (insulin-dependent and non-insulin-dependent), and the molecular mechanism of insulin action alongside oral hypoglycemic agents. It outlines the biosynthesis of insulin, its receptor interactions, and the pharmacology of various oral antidiabetic drugs like sulfonylureas, biguanides, and thiazolidinediones. The document also covers the history of insulin discovery and highlights the importance of insulin analogues and their pharmacokinetics.