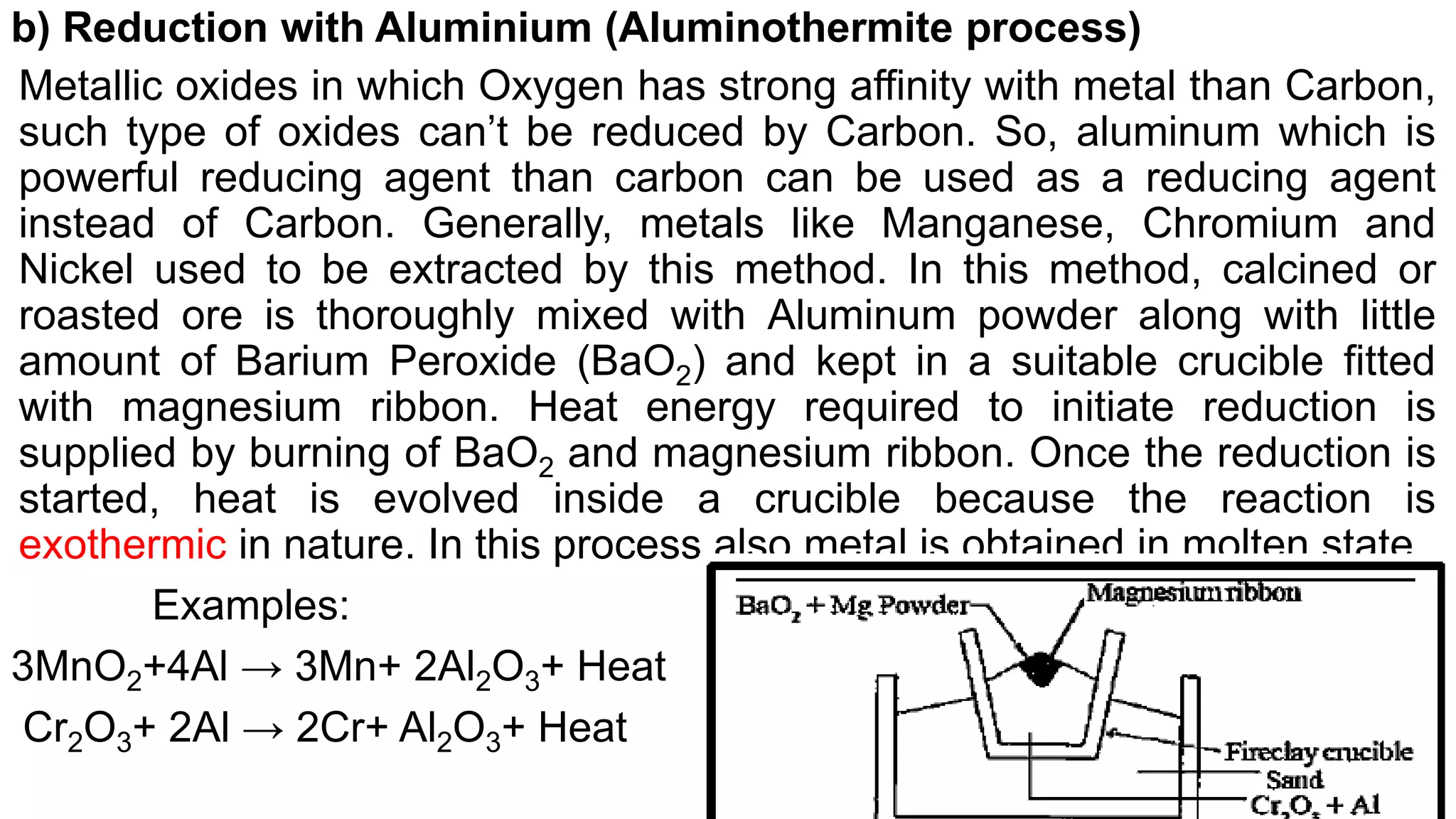
The document discusses various topics related to metals and metallurgy. It begins by defining metals and metallurgical principles, then discusses alkali metals, alkaline earth metals, and their compounds. It also covers minerals, ores, concentration processes, smelting, electrolytic reduction, and refining of metals. Key concentration methods described are magnetic separation, gravity separation, froth flotation, and leaching. The document also differentiates between minerals and ores, discusses alloys, amalgams, and existence of metals. It provides an overview of metallurgy and the steps involved in the metallurgical process.







![Importance of Alloys:
They are less rusting in nature in comparison to the pure metals. So, using
alloys we can save the loss from rusting.
Alloys are stronger than metal i.e. by making alloys we can increase the
mechanical strength of metals.
Alloys are useful for making colorful utensils and materials i.e. we can import
color if we make the alloy.
Amalgam
The homogeneous mixture of Mercury with metal is known as amalgam.
Mercury is in liquid form so by dissolving metal on Mercury we can obtain
an Amalgam.
E.g. Hg+ Na, Na-Hg [Sodium amalgam],Na/Hg
Hg+Cu,Cu.Hg[Copper amalgam],Cu/Hg
Note: metals like Iron (Fe), Cobalt (Co) and Nickel(Ni) don’t form amalgam
because they do not dissolve in Mercury.](https://image.slidesharecdn.com/metallurgicalprocess-220925112854-0ebb37eb/75/Metallurgical-process-pptx-8-2048.jpg)