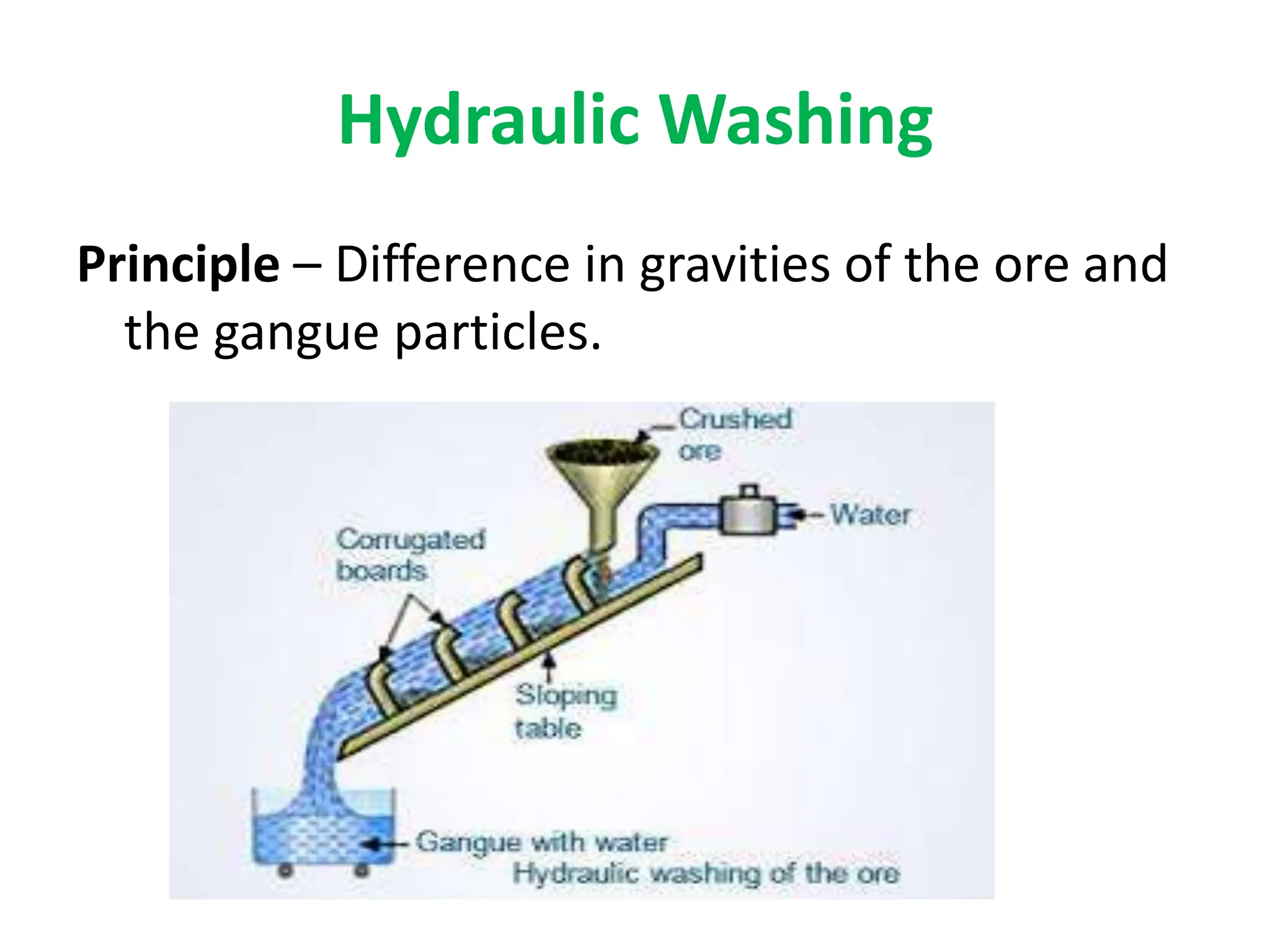
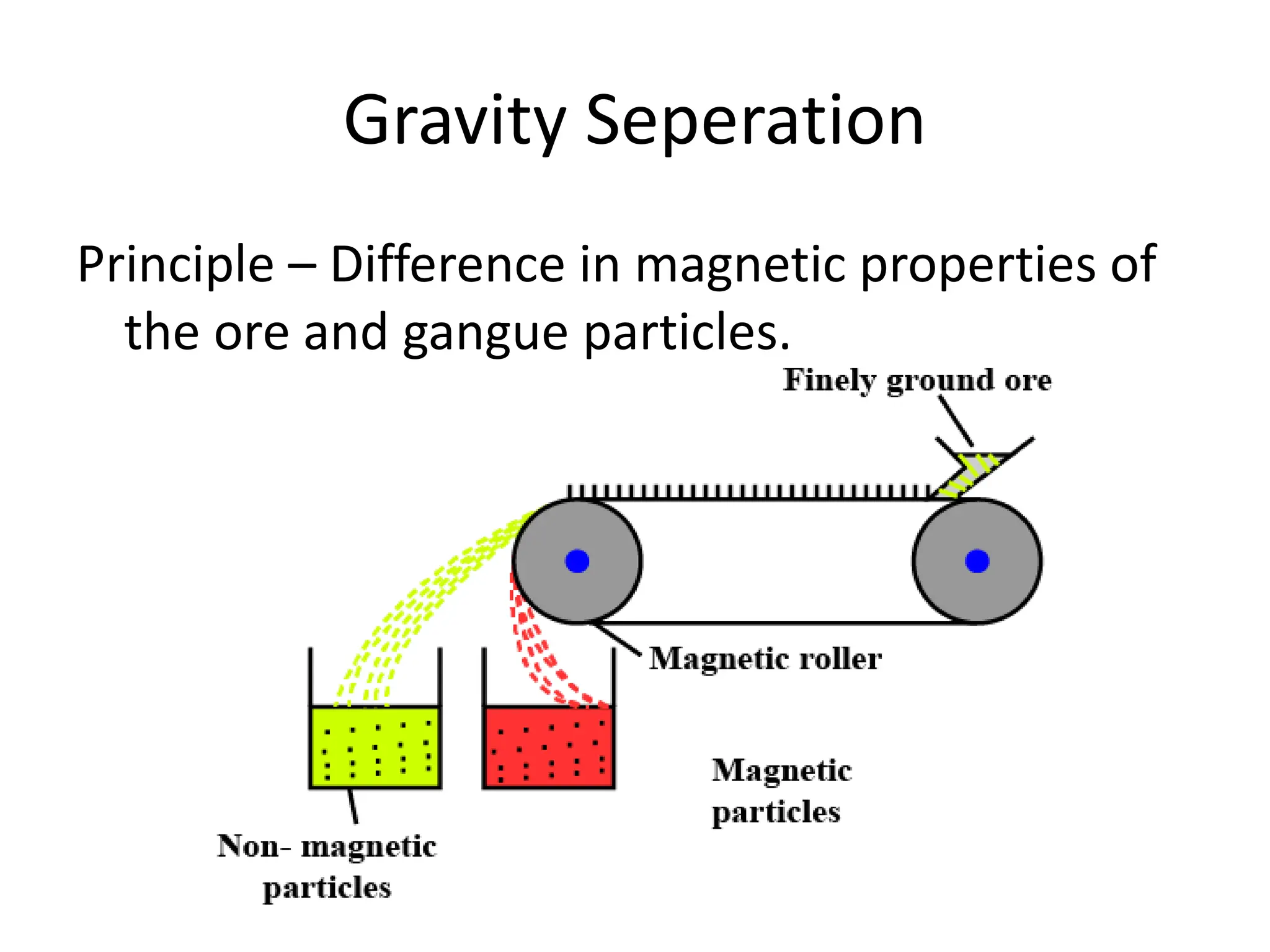
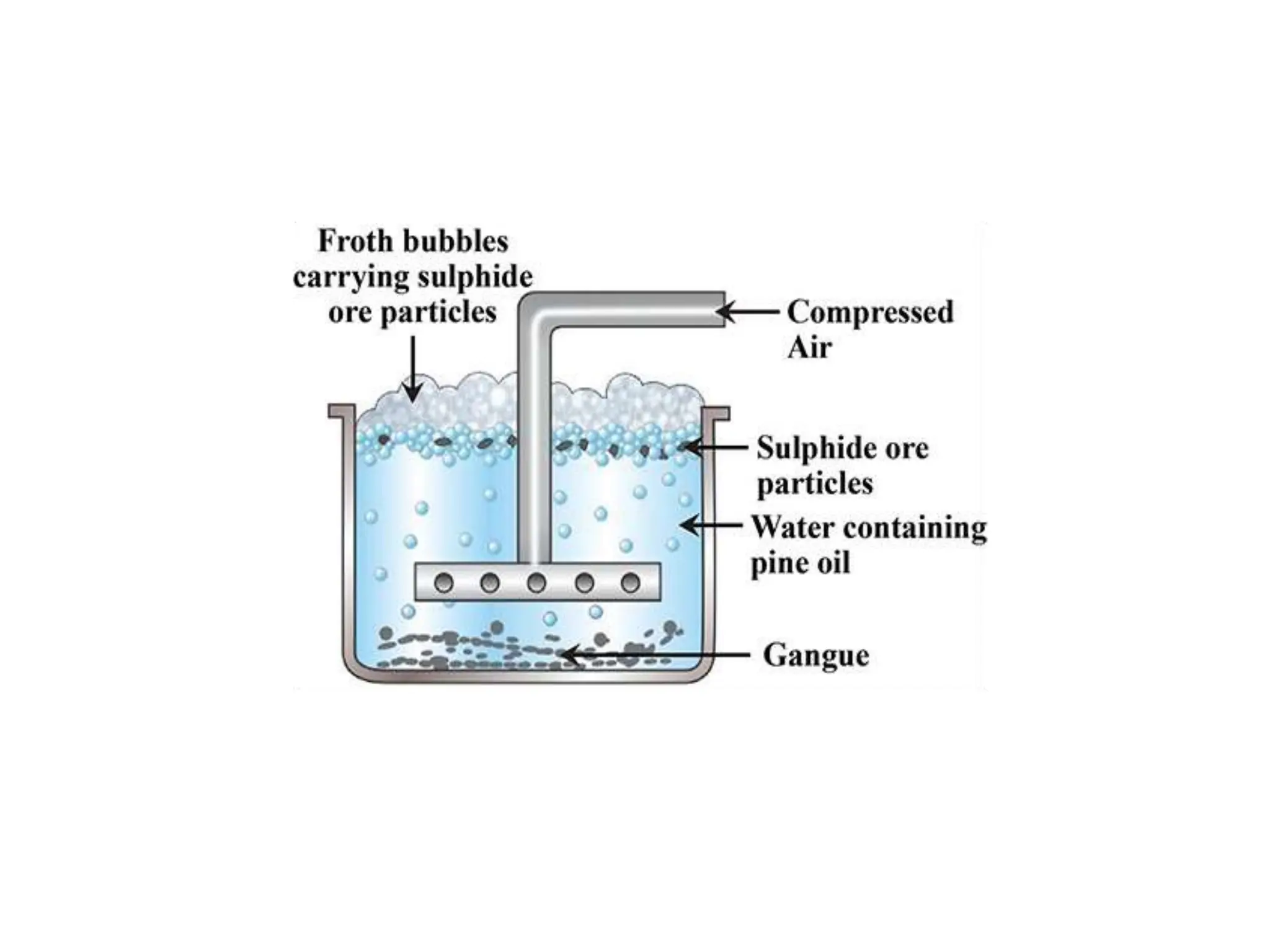
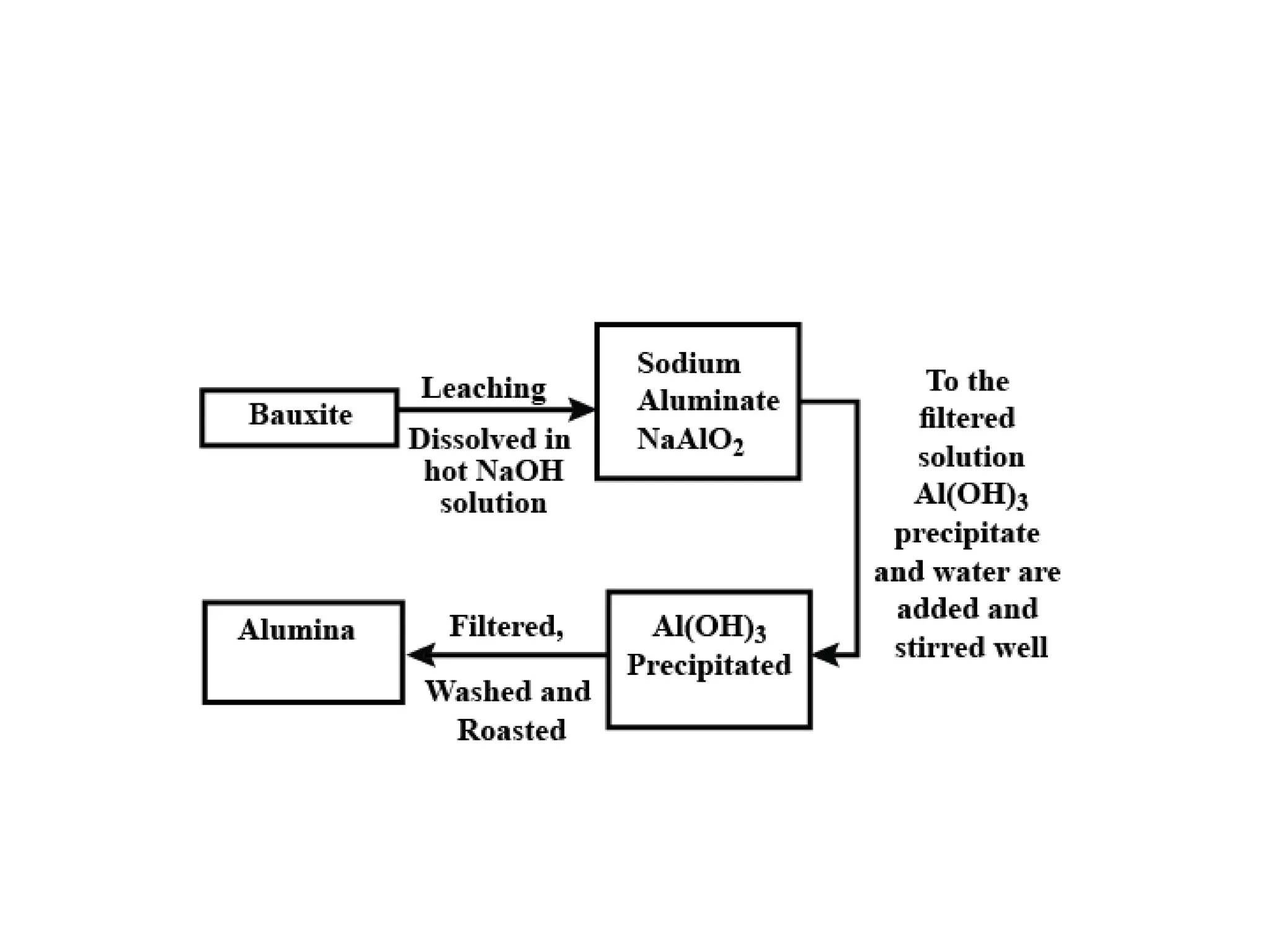
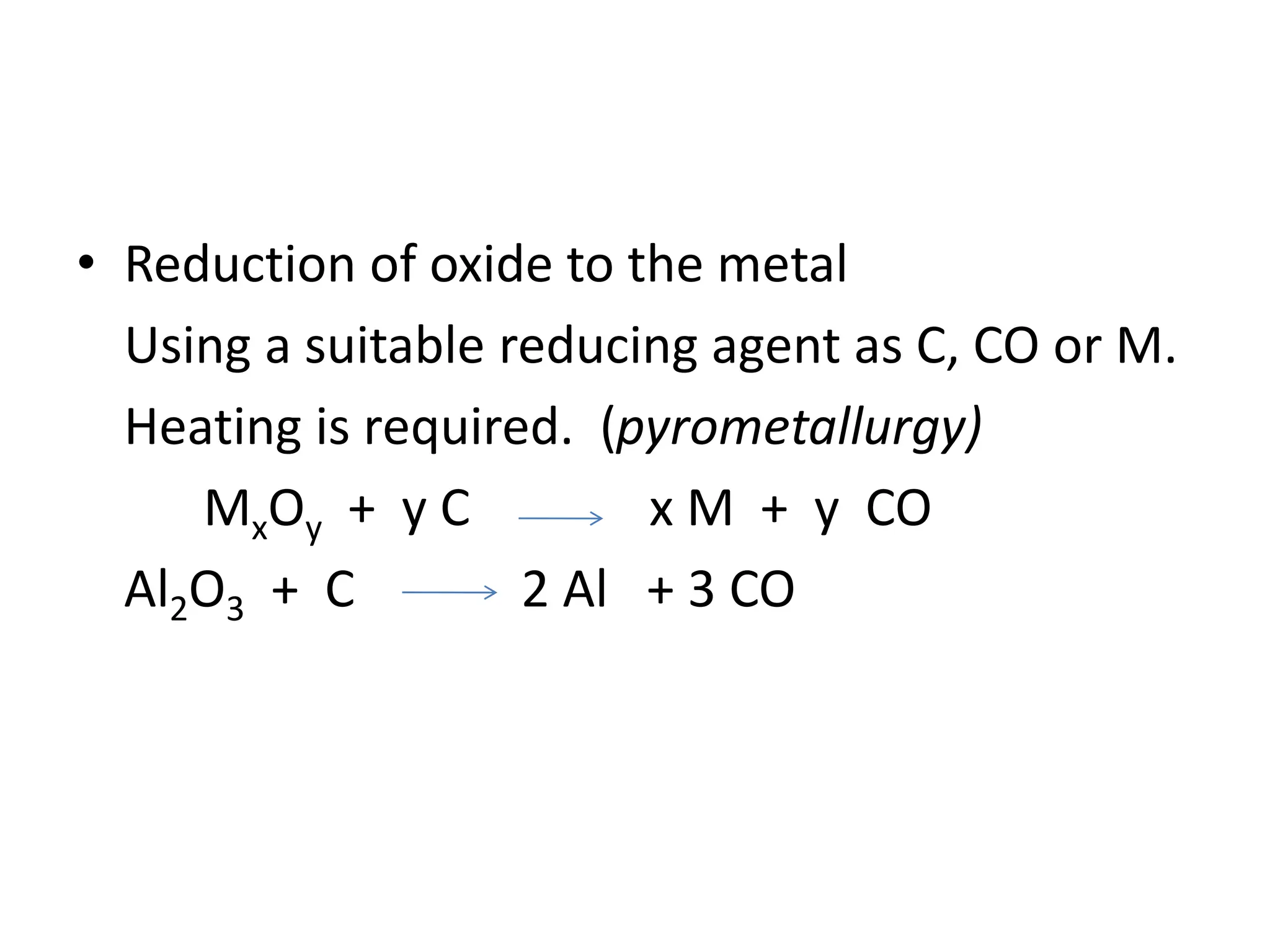
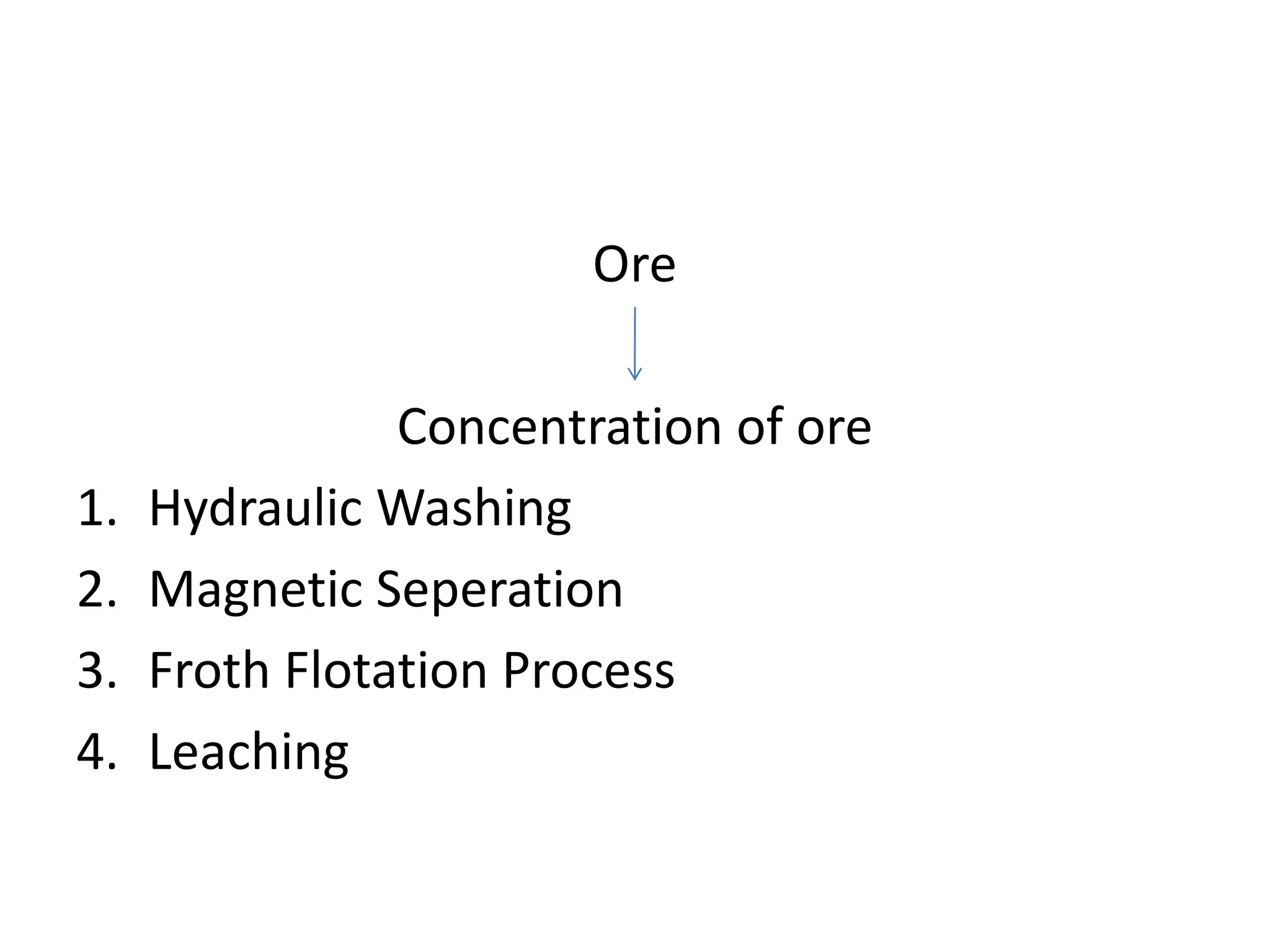
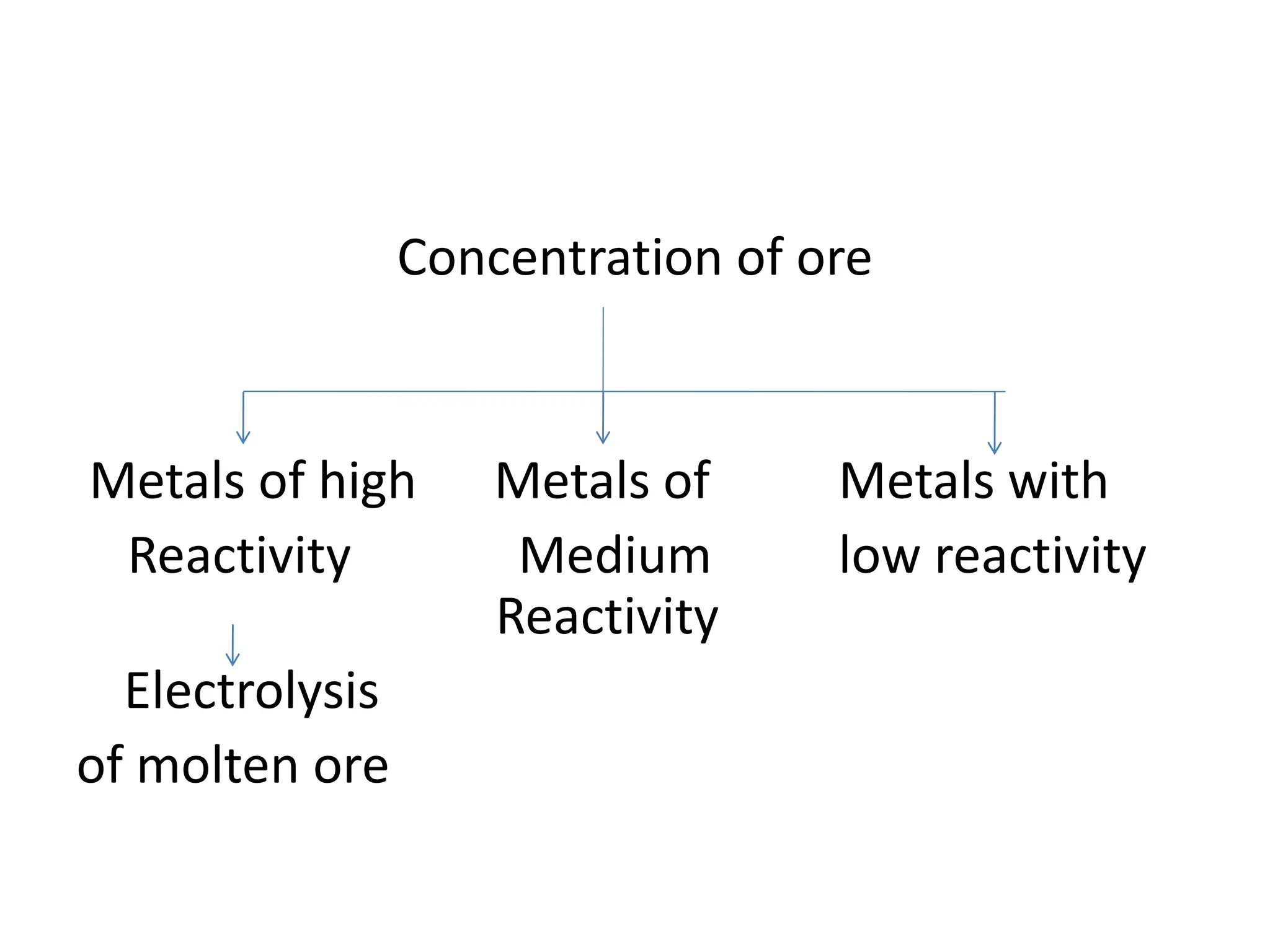
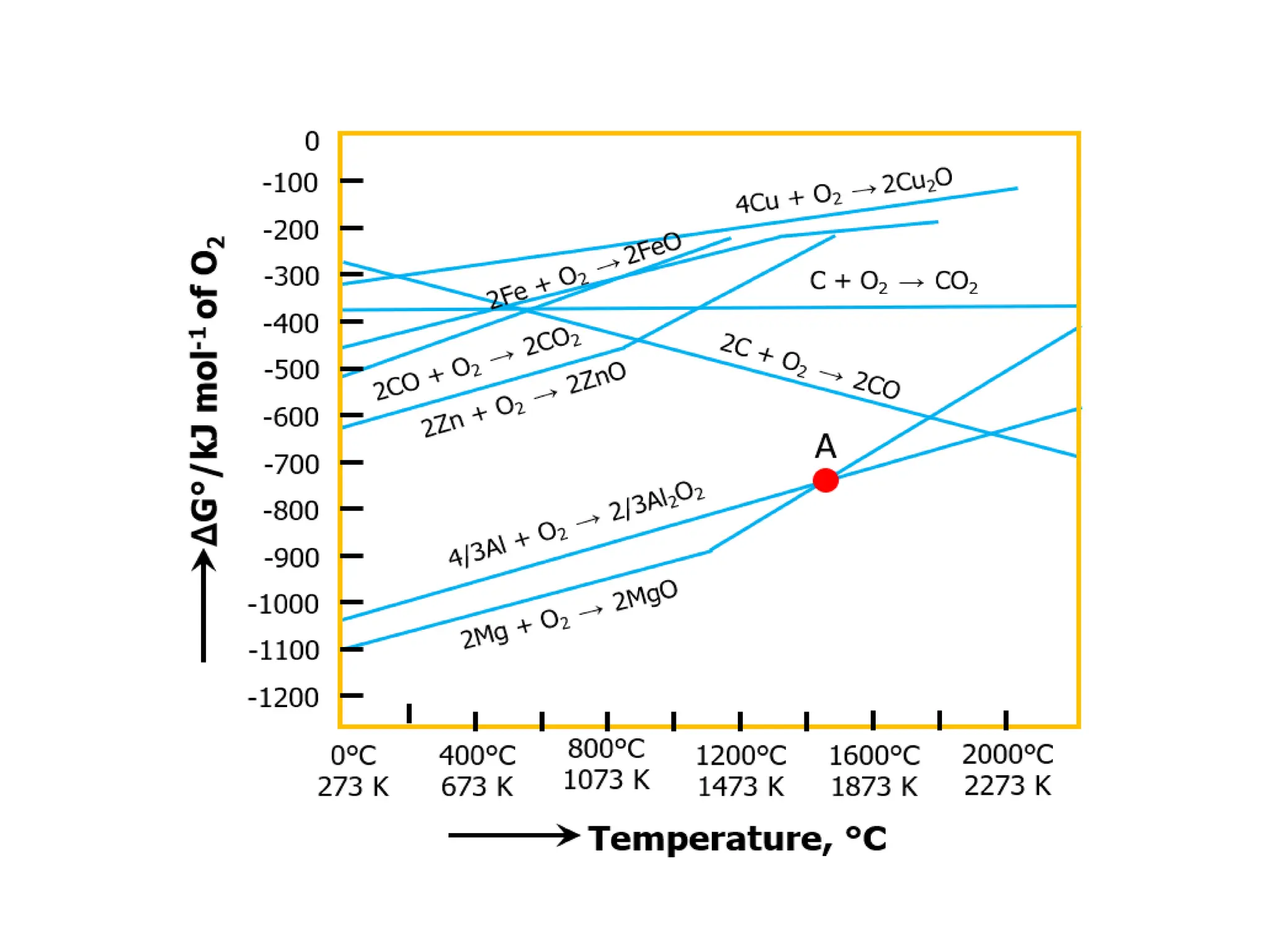
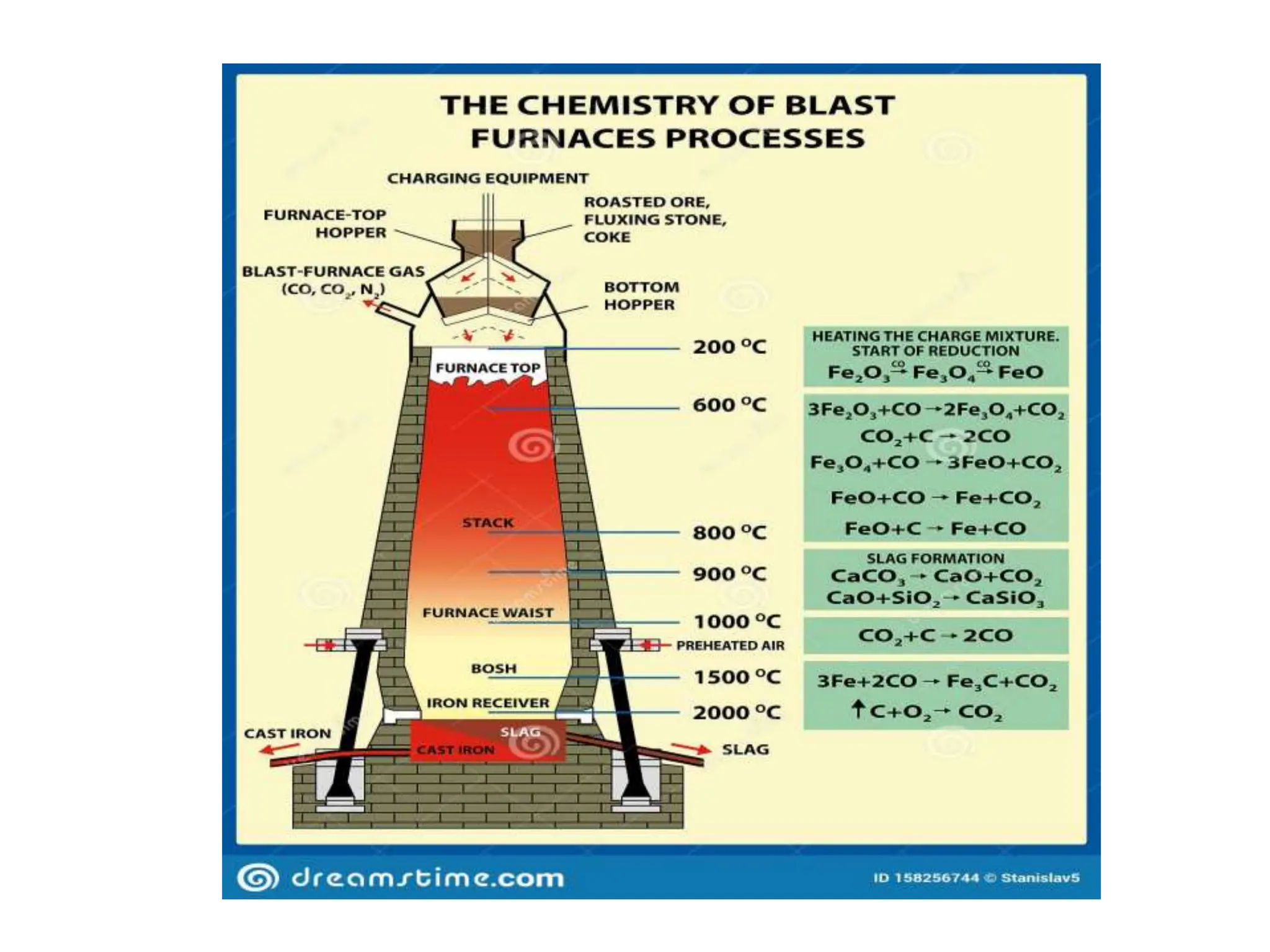
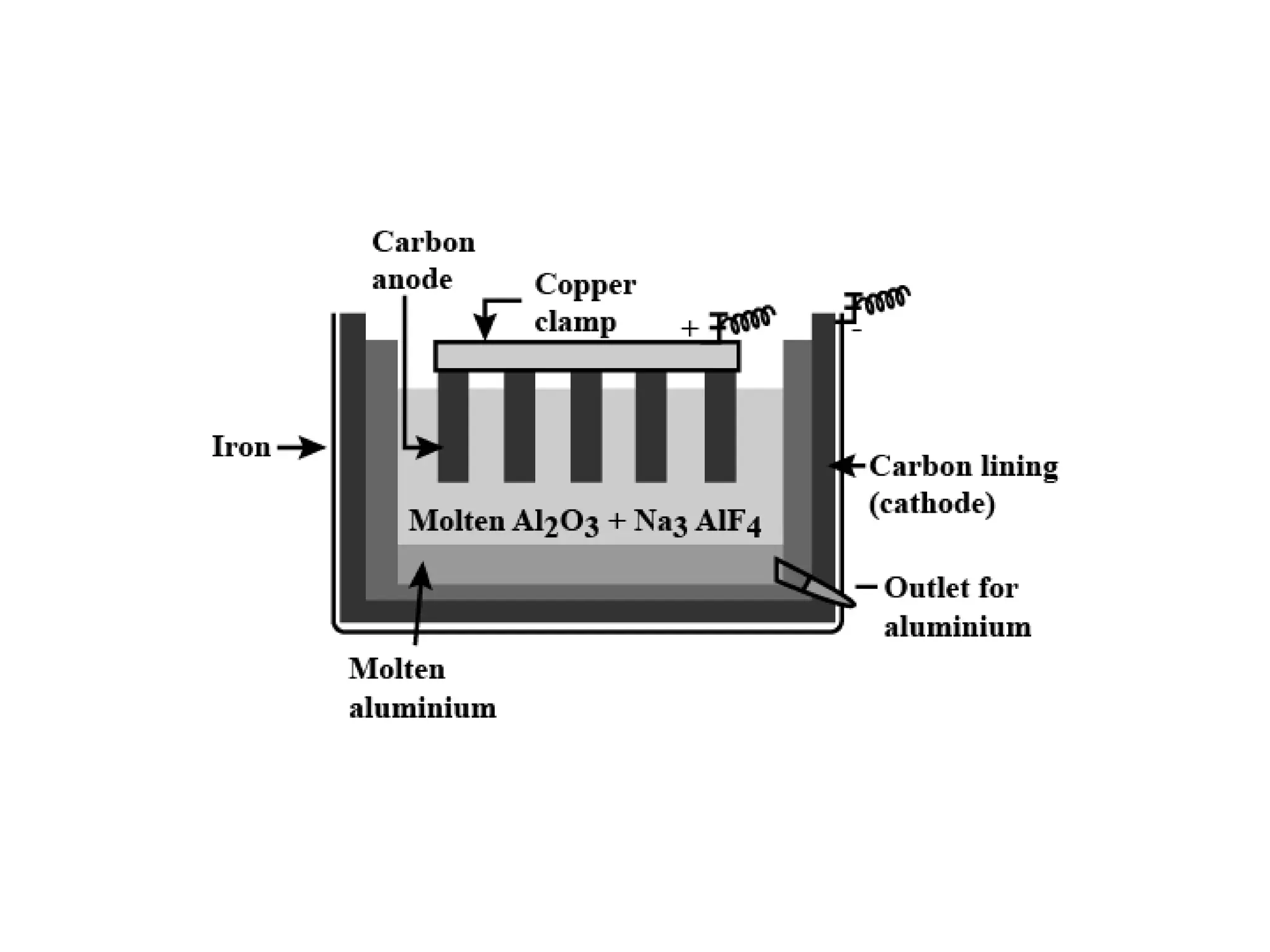
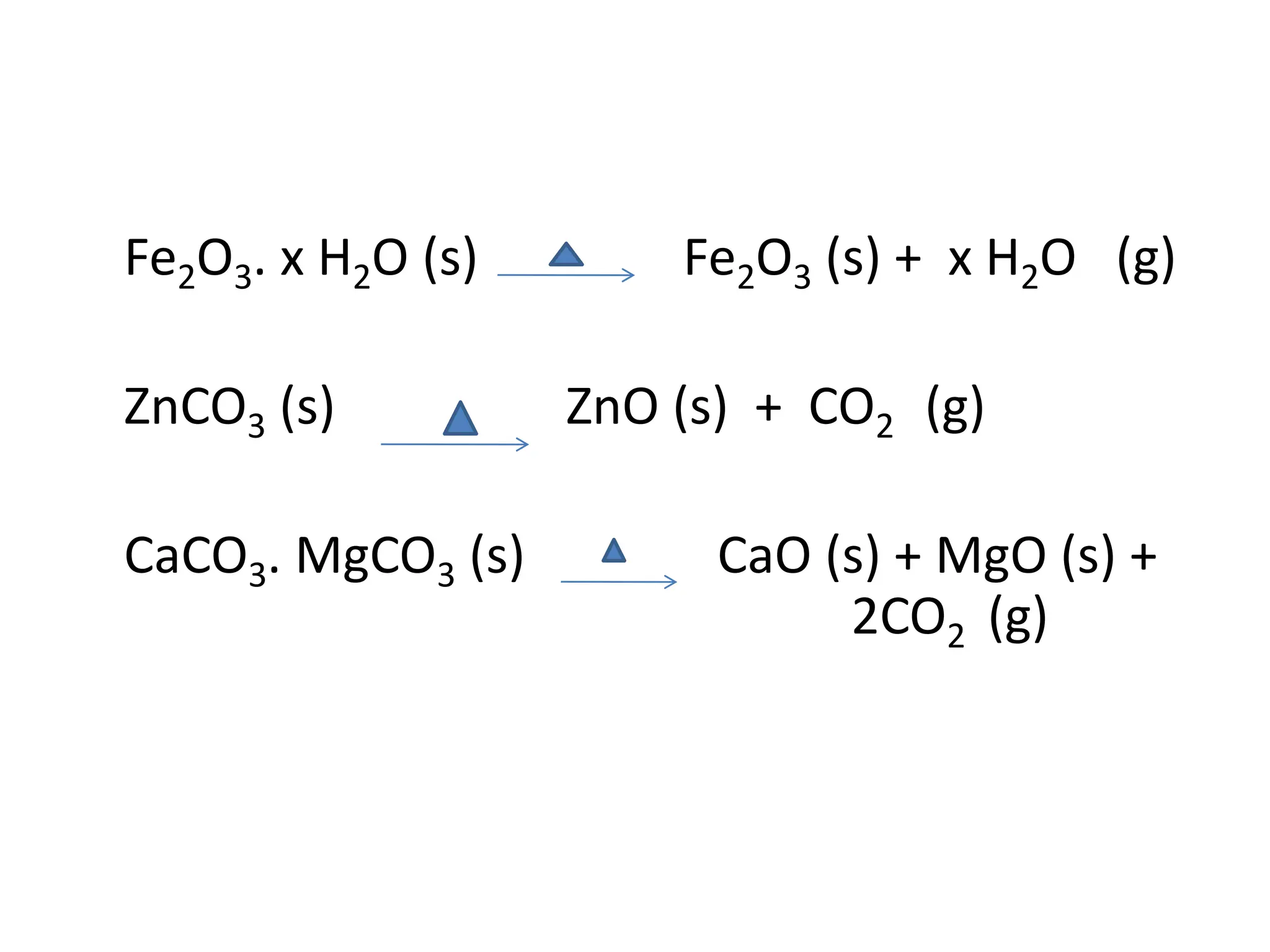The document describes the processes of isolating elements from ores, highlighting minerals, ores, and gangue, as well as the methods for metal extraction such as concentration, calcination, roasting, and electrolysis. It explains the principles of metallurgy, the significance of leaching, and the use of thermodynamic principles for selecting reducing agents. Additionally, the document covers specific extraction methods for iron, copper, and zinc, detailing the chemical reactions and the formation of various metal oxides and the resultant products.





![Occurrence of Metals
Metal Ores Compo0sitions
Aluminium Bauxite
Kaolinite [ Al2 (OH )4 Si2O5 ]
Iron Haematite Fe2O3
Magnetite Fe2O3
Siderite FeCO3
Iron Pyrites FeS2
Copper Copper Pyrites CeFeS2
Malachite CuCO3 . Cu (OH )2
Copper glance Cu2S
Cuprite Cu2O
Zinc Zinc blende ZnS
Calamine ZnCO3
Zincite ZnO](https://image.slidesharecdn.com/generalprinciplesandprocessesofisolationofelements-231029165819-9856c4bd/75/General-Principles-and-Processes-of-Isolation-of-Elements-pptx-6-2048.jpg)