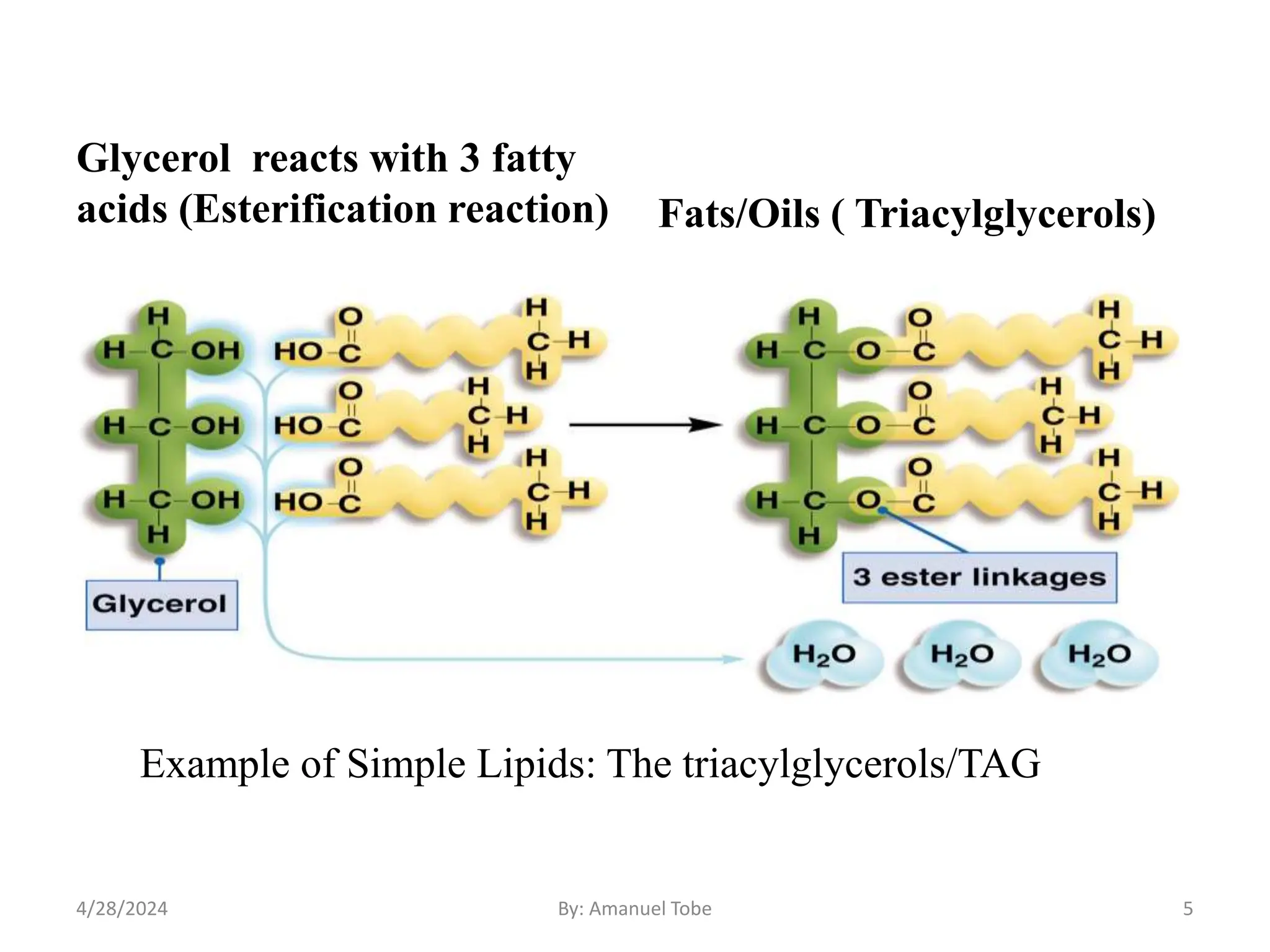
The document provides an overview of lipids, defining them as biochemical compounds that are insoluble in water but soluble in organic solvents, and classifying them into simple, compound, and derived lipids. It discusses the structure and functions of various lipid types, including triacylglycerols, phospholipids, and steroids, highlighting their roles in energy storage, cellular structure, and biological processes. Additionally, it emphasizes the importance of essential fatty acids and their impact on health and various physiological functions.