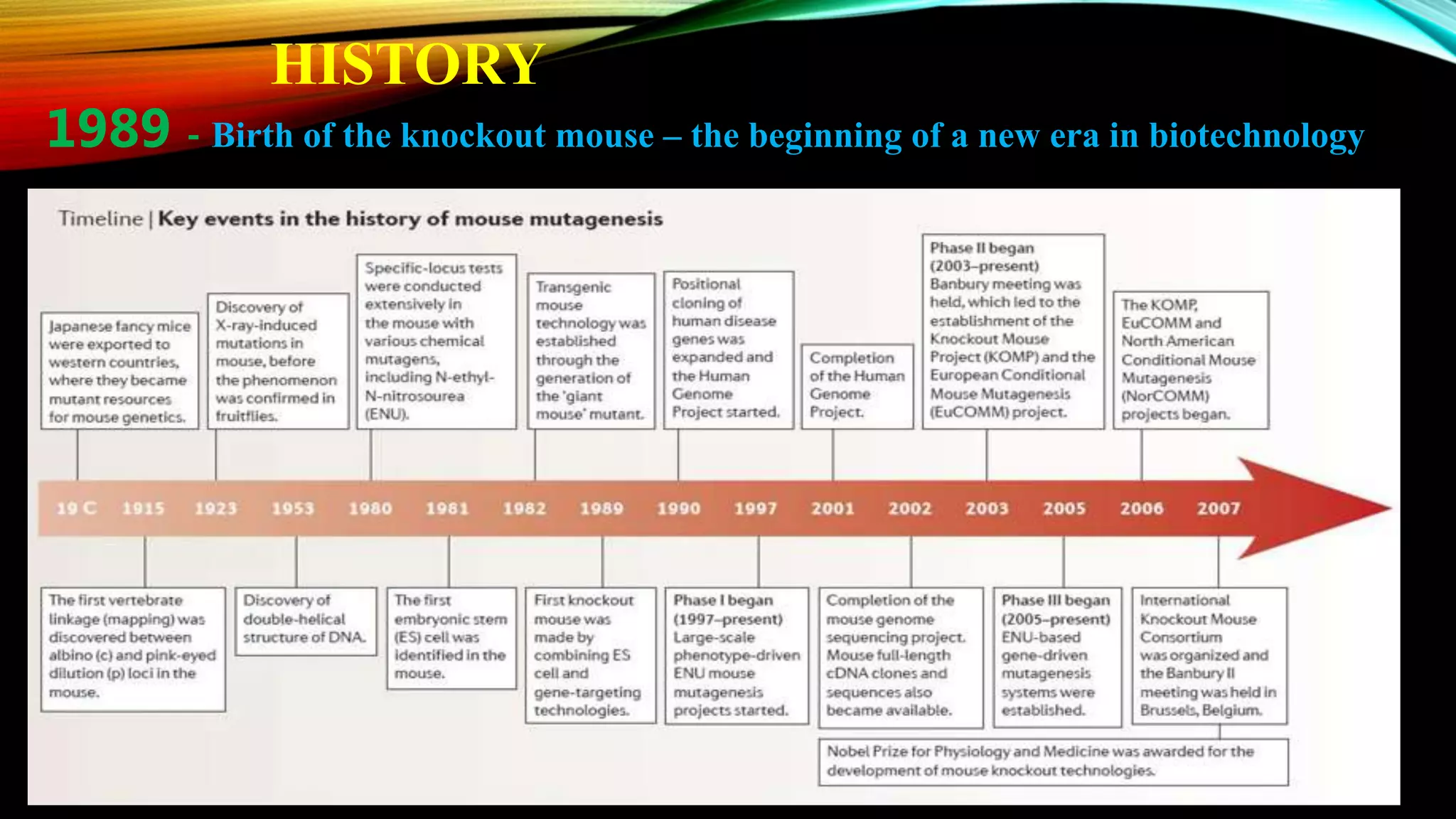
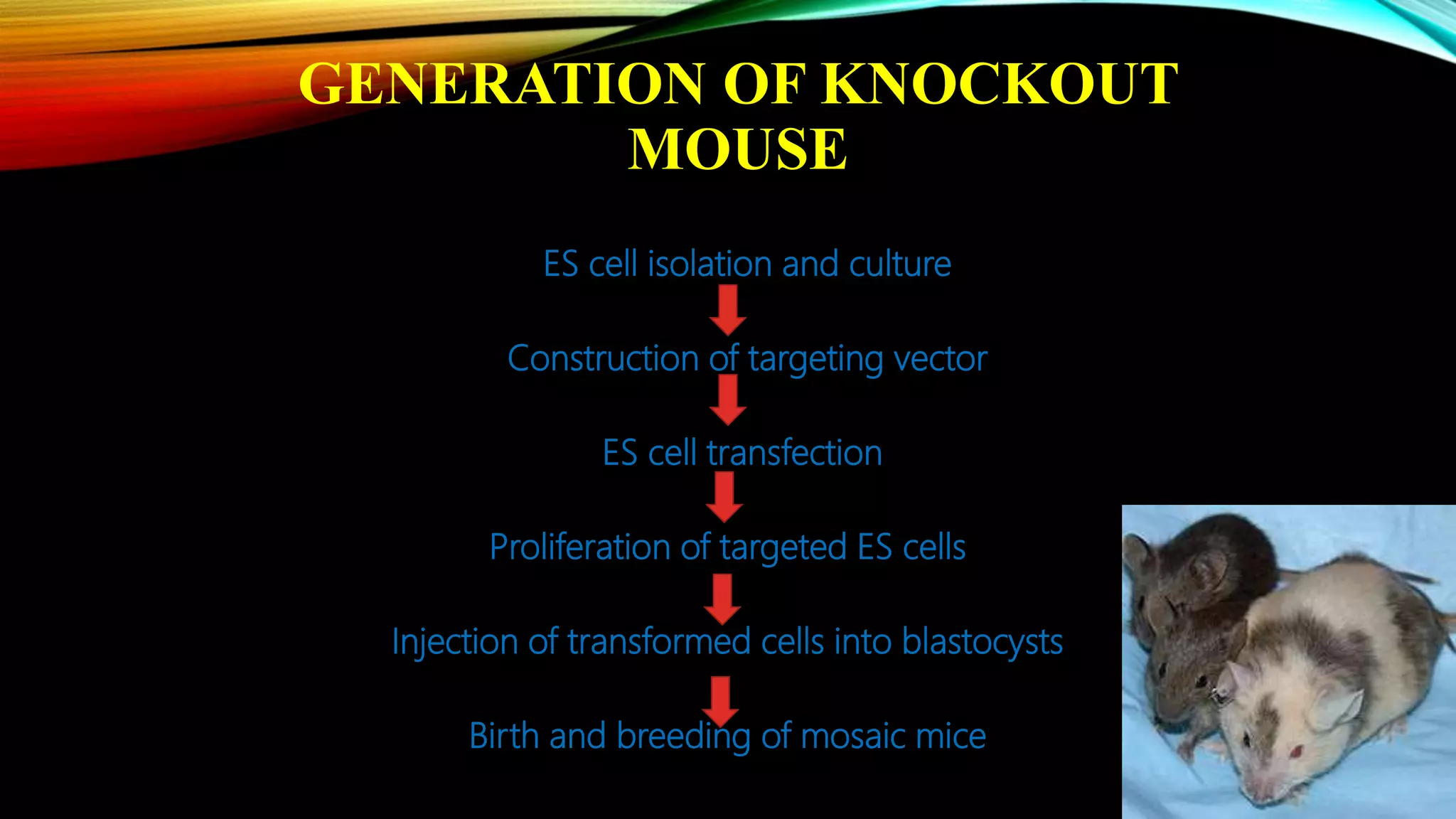
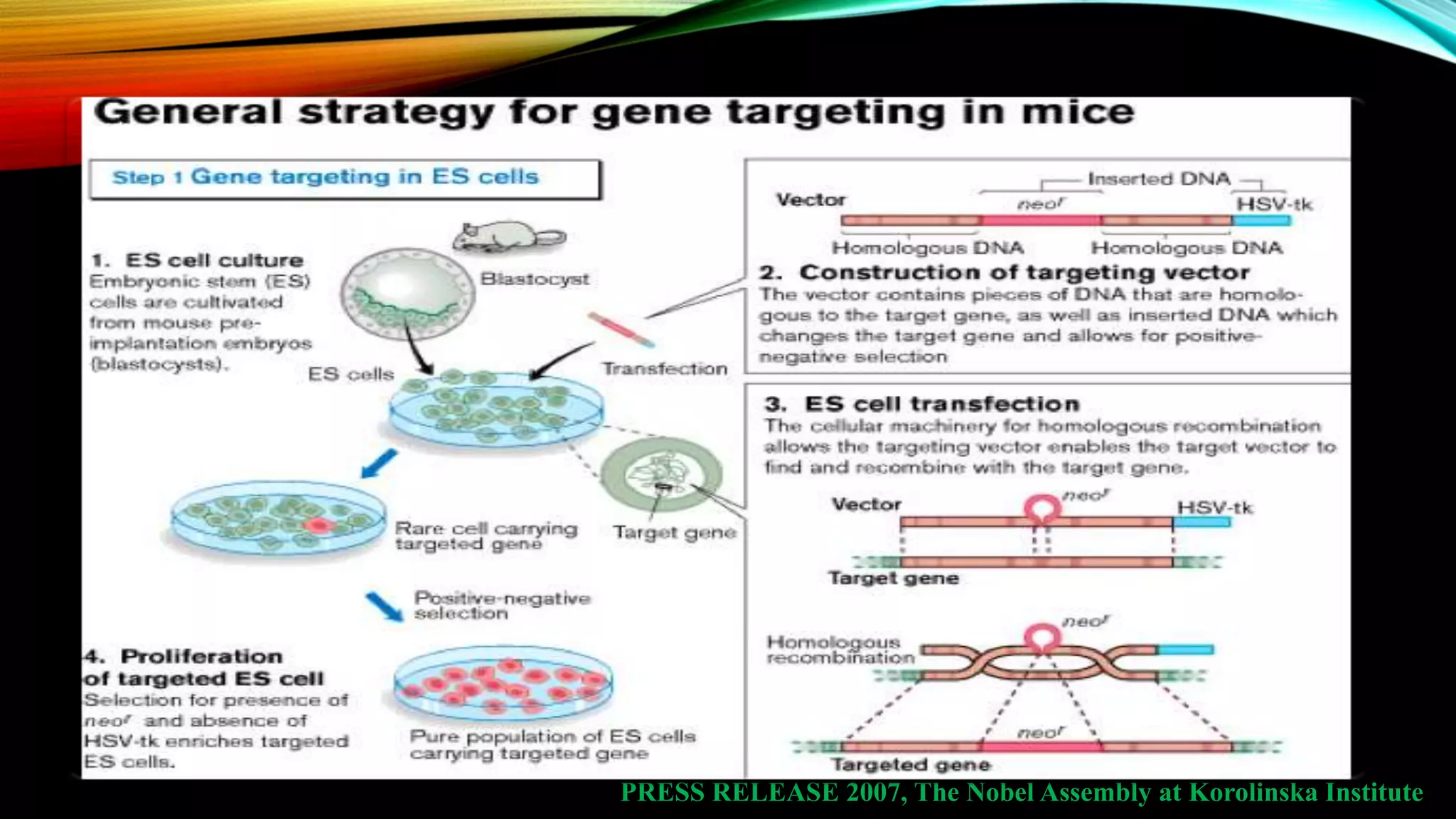
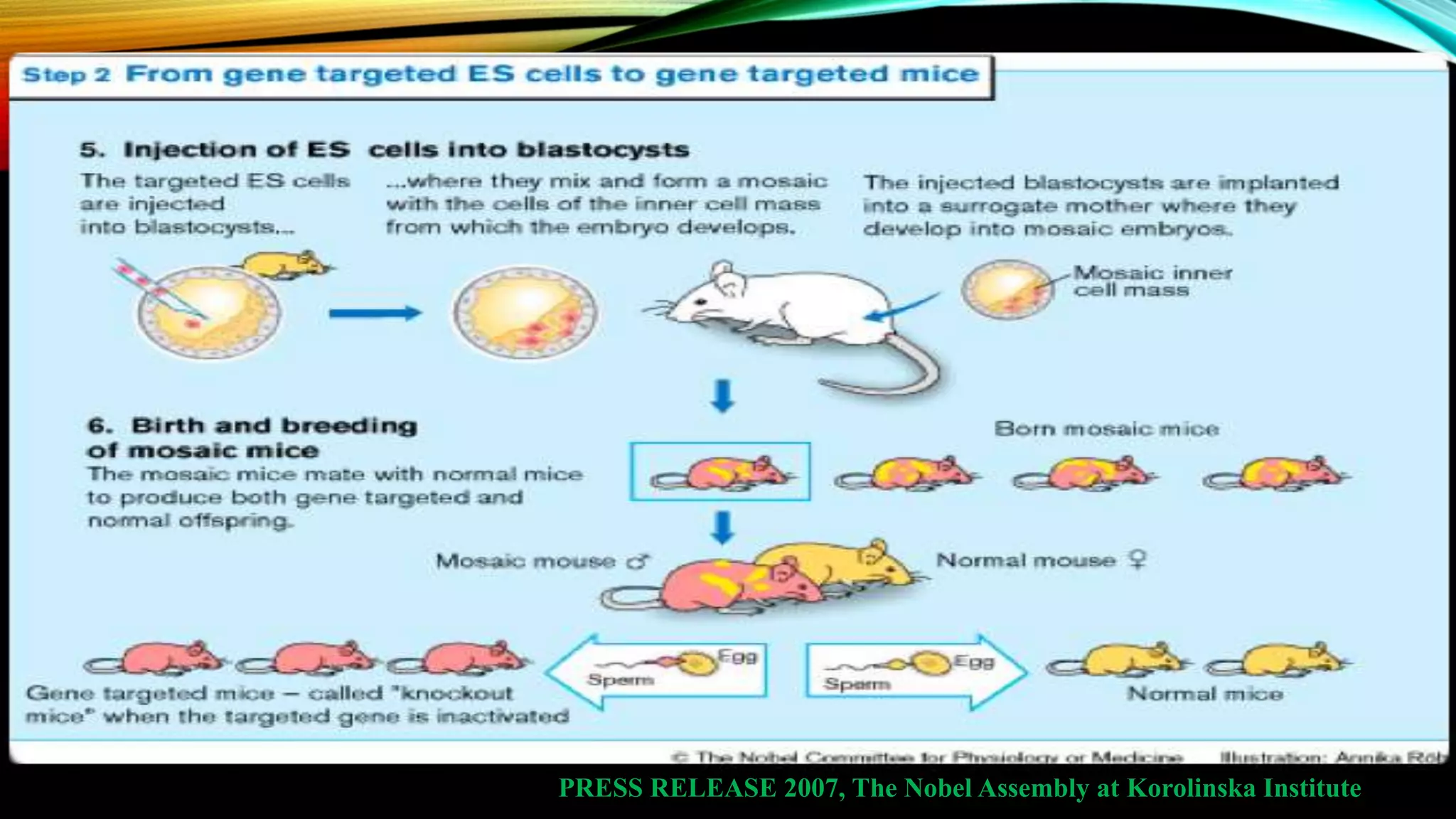
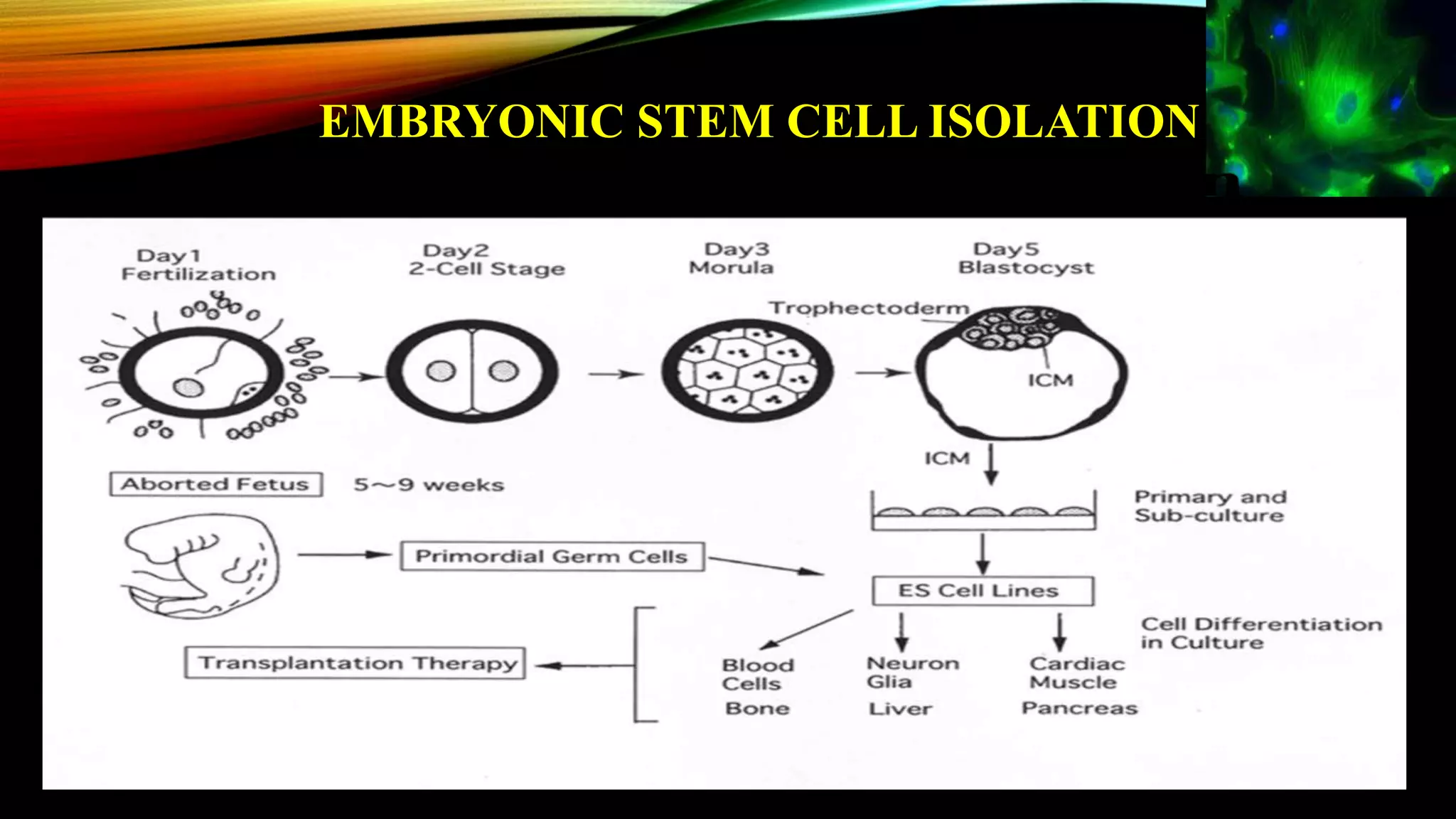
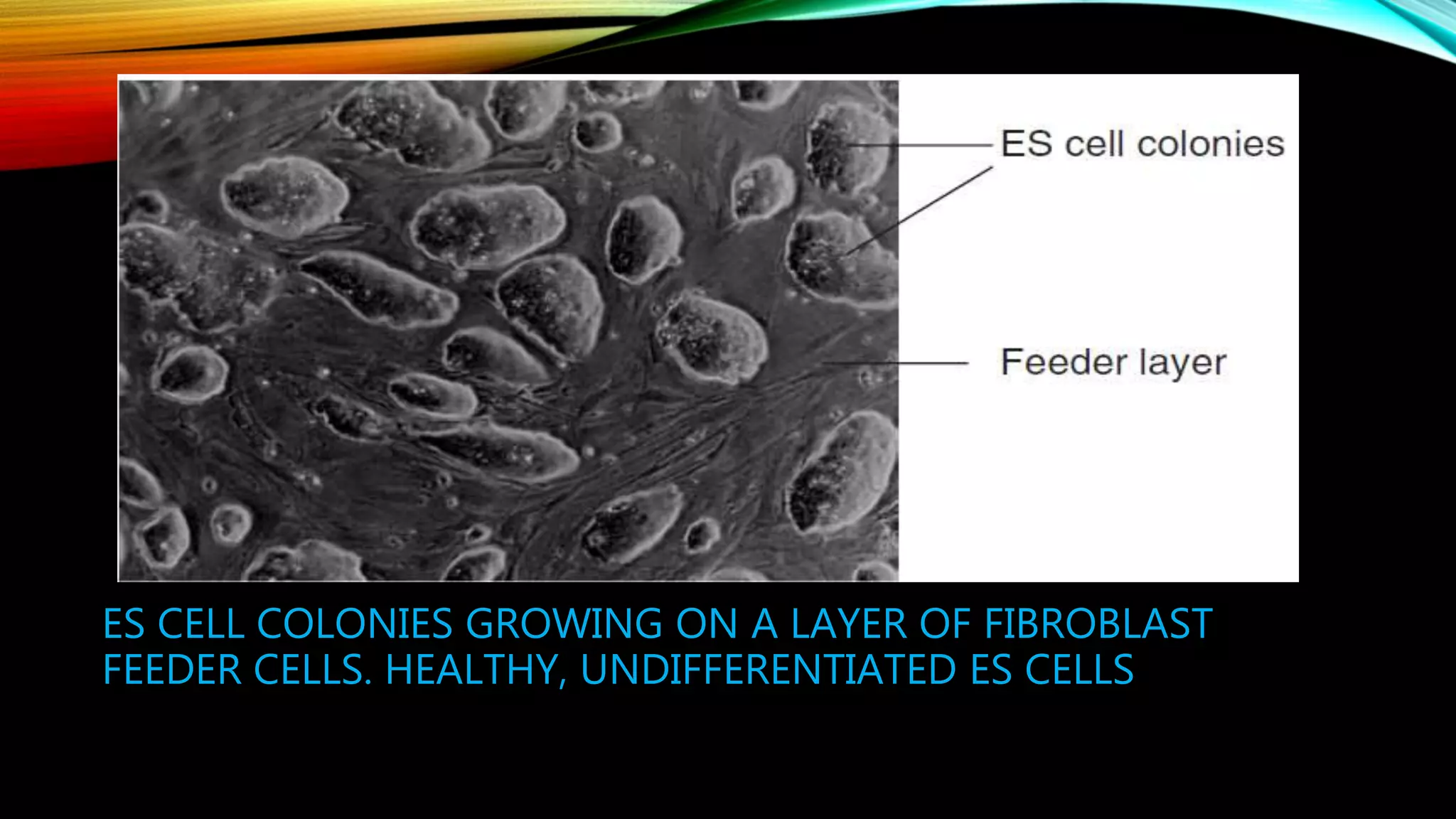
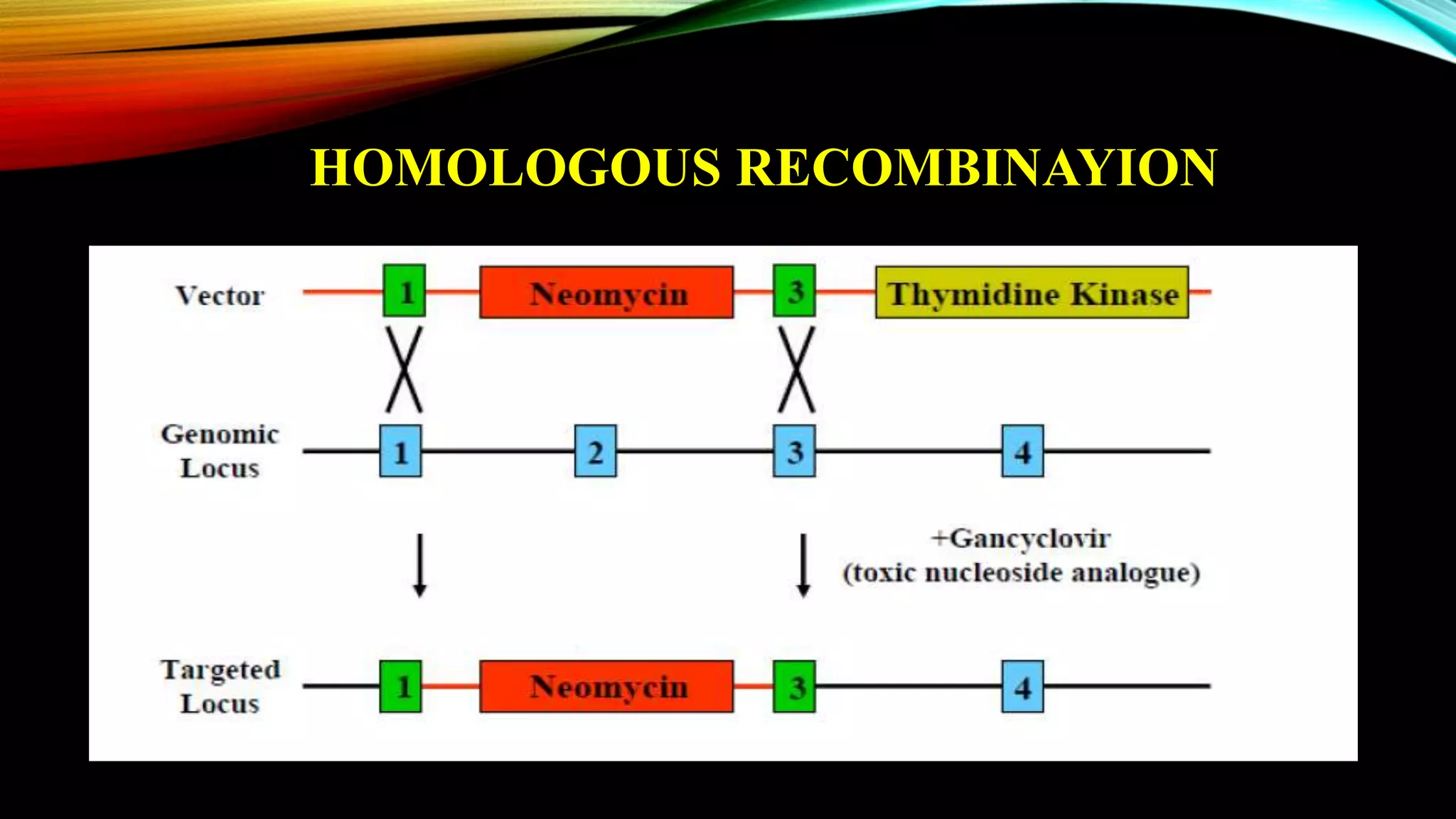
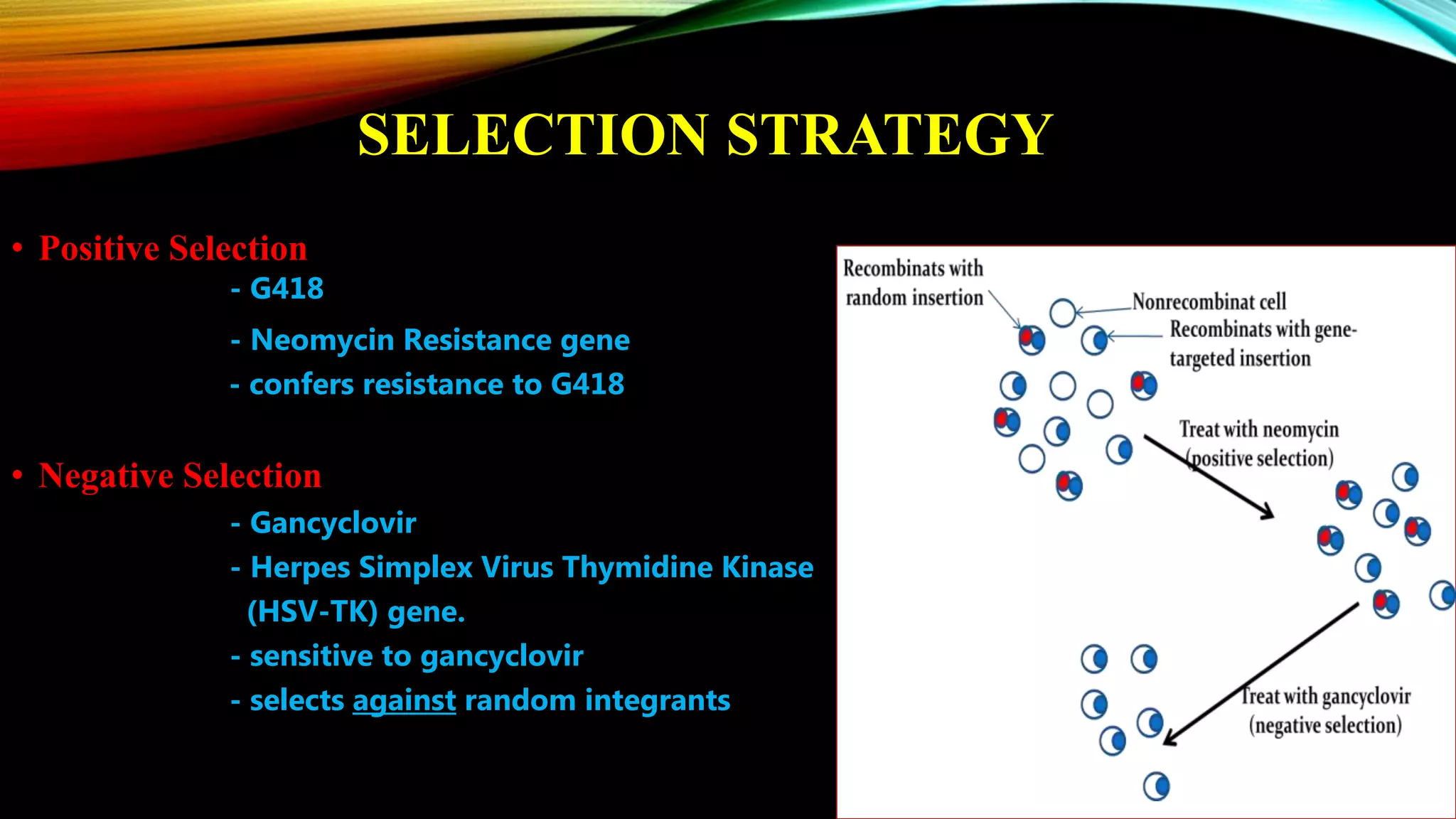
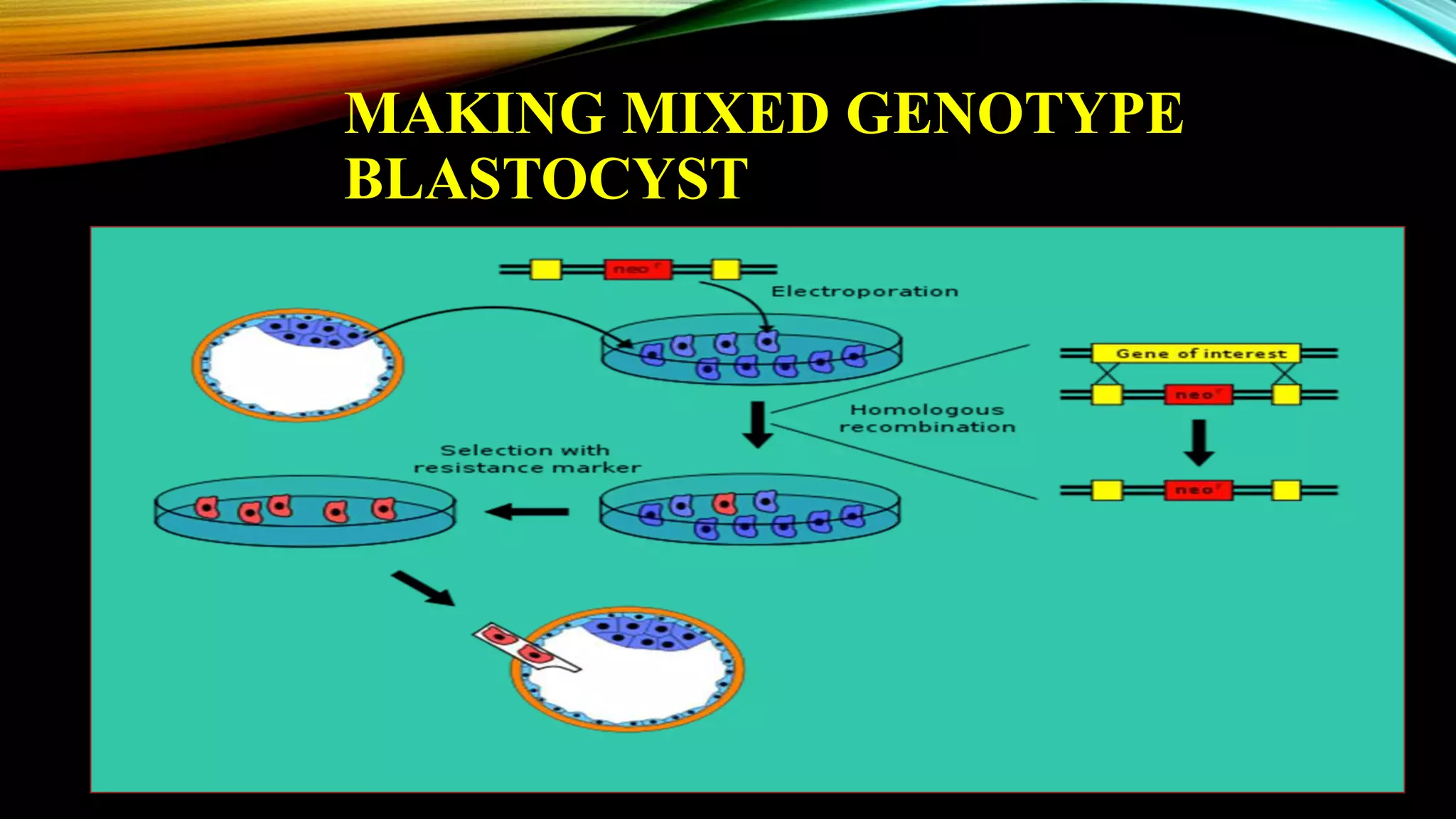
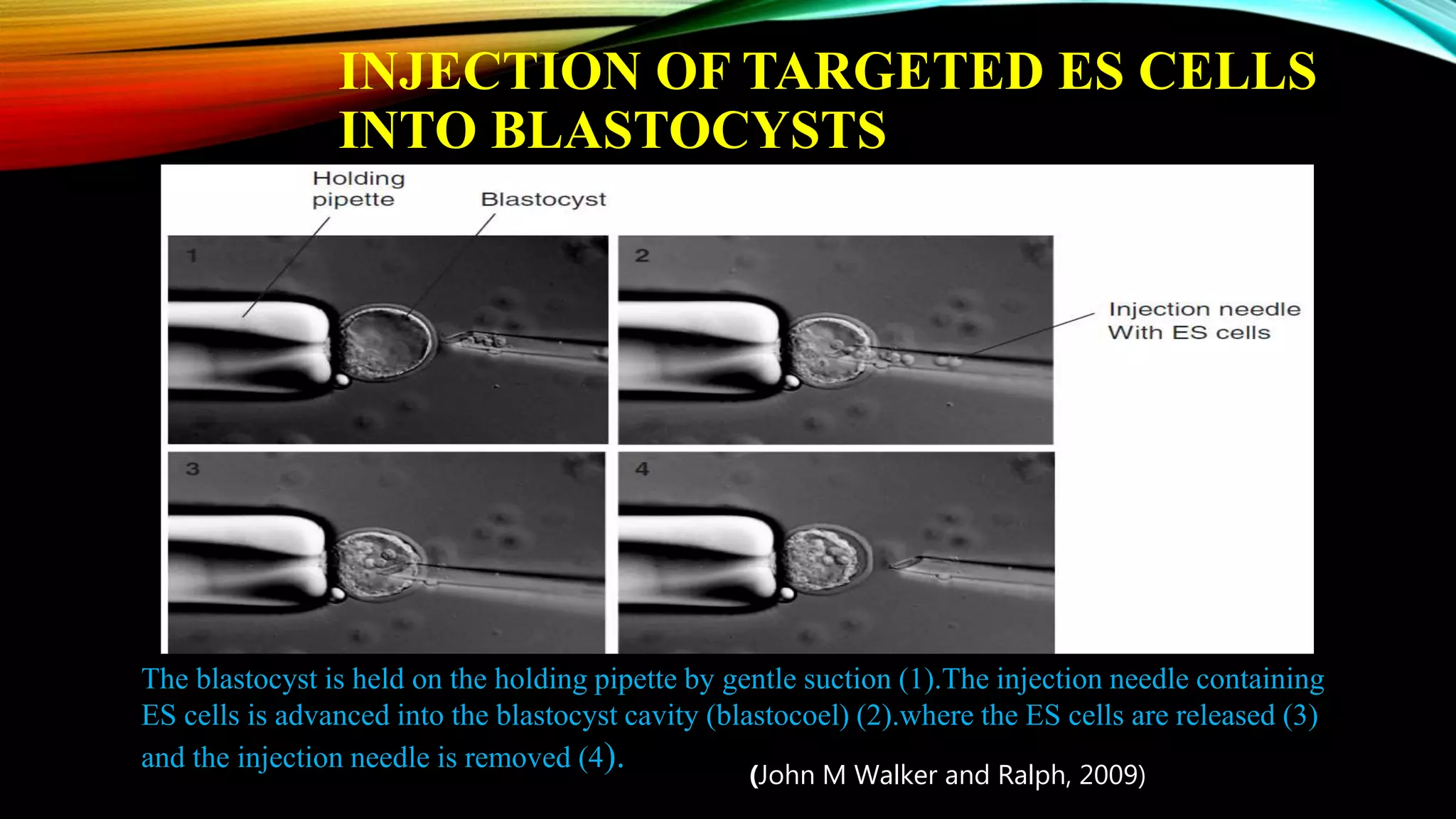
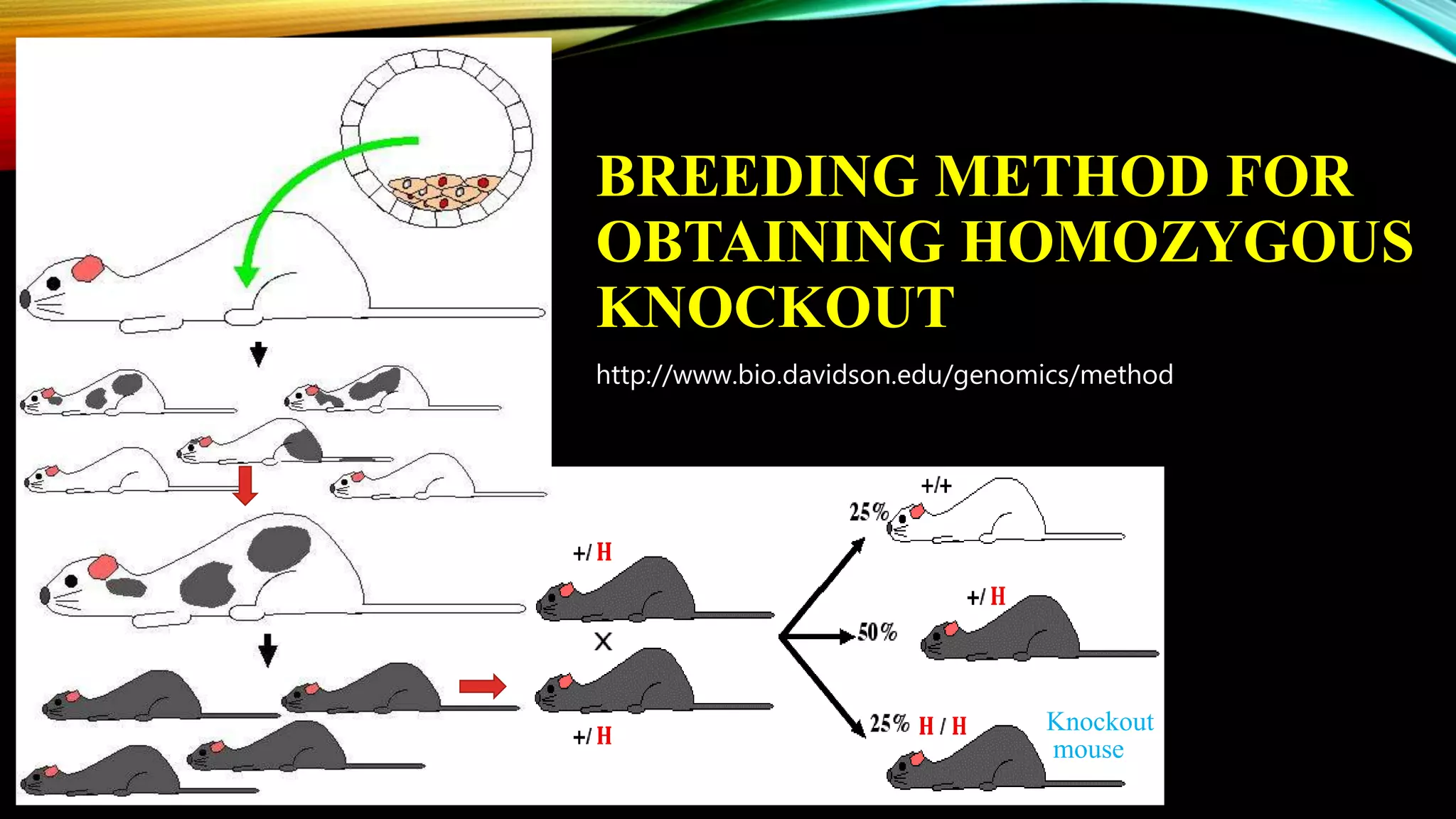
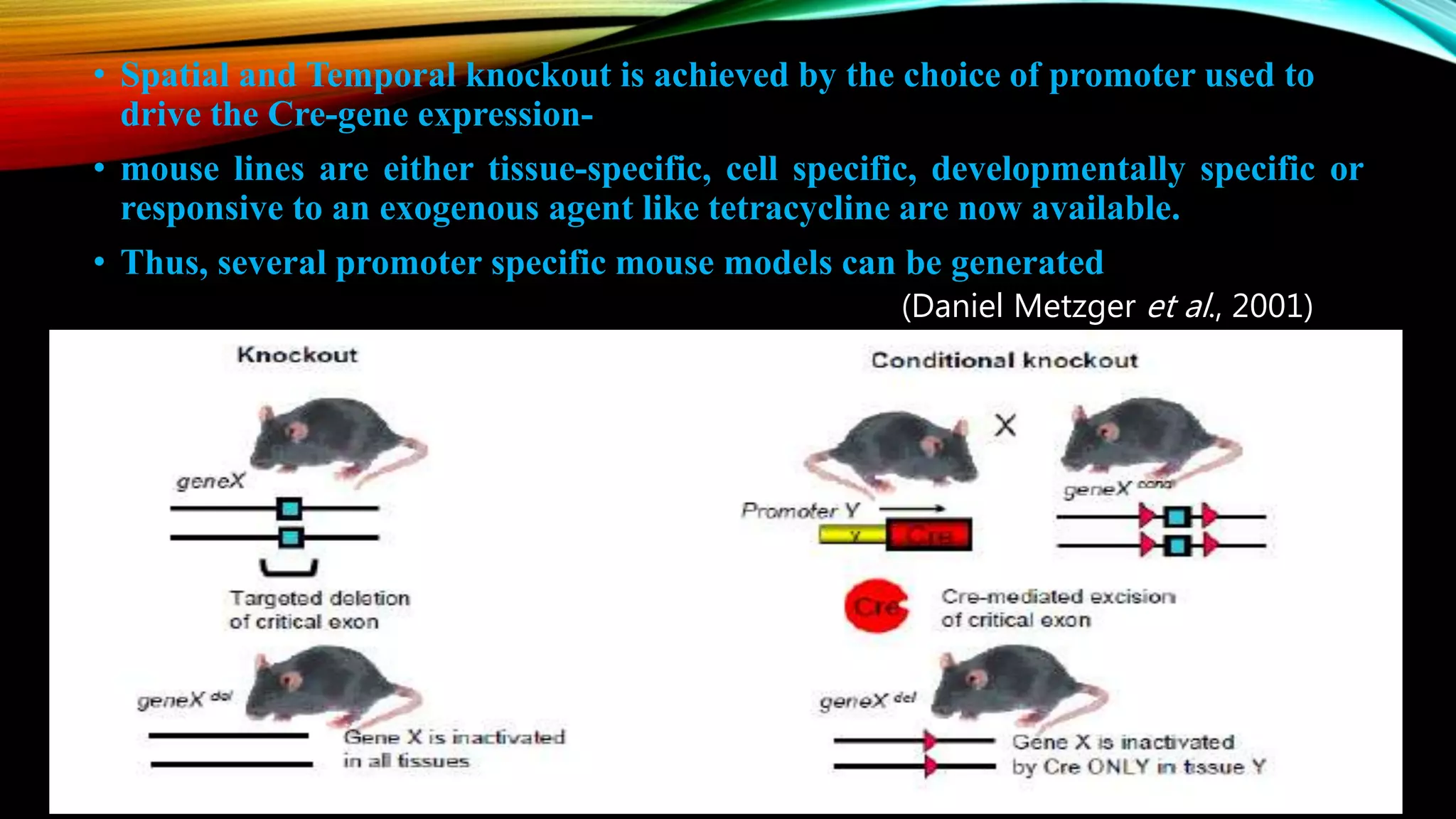
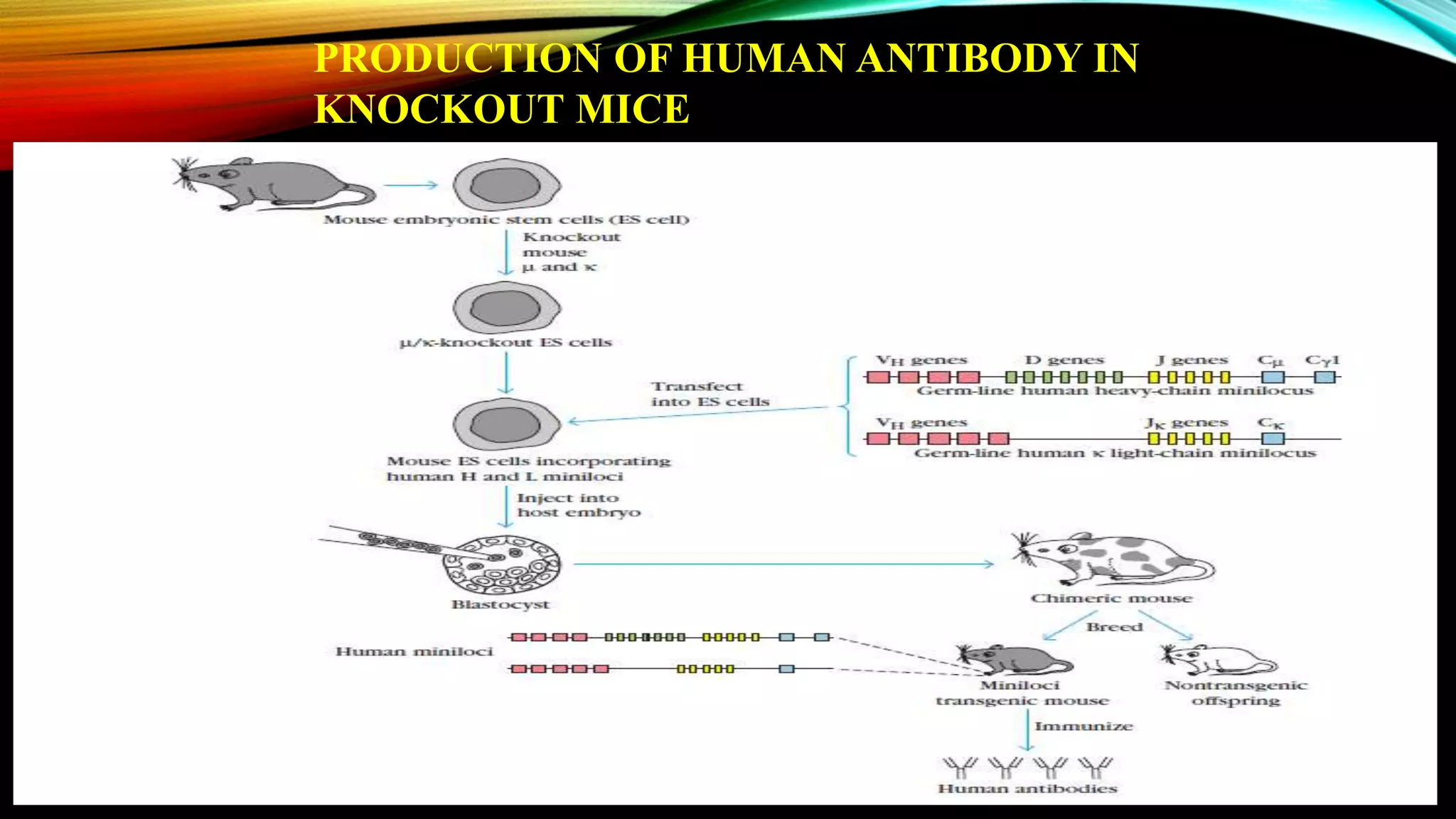
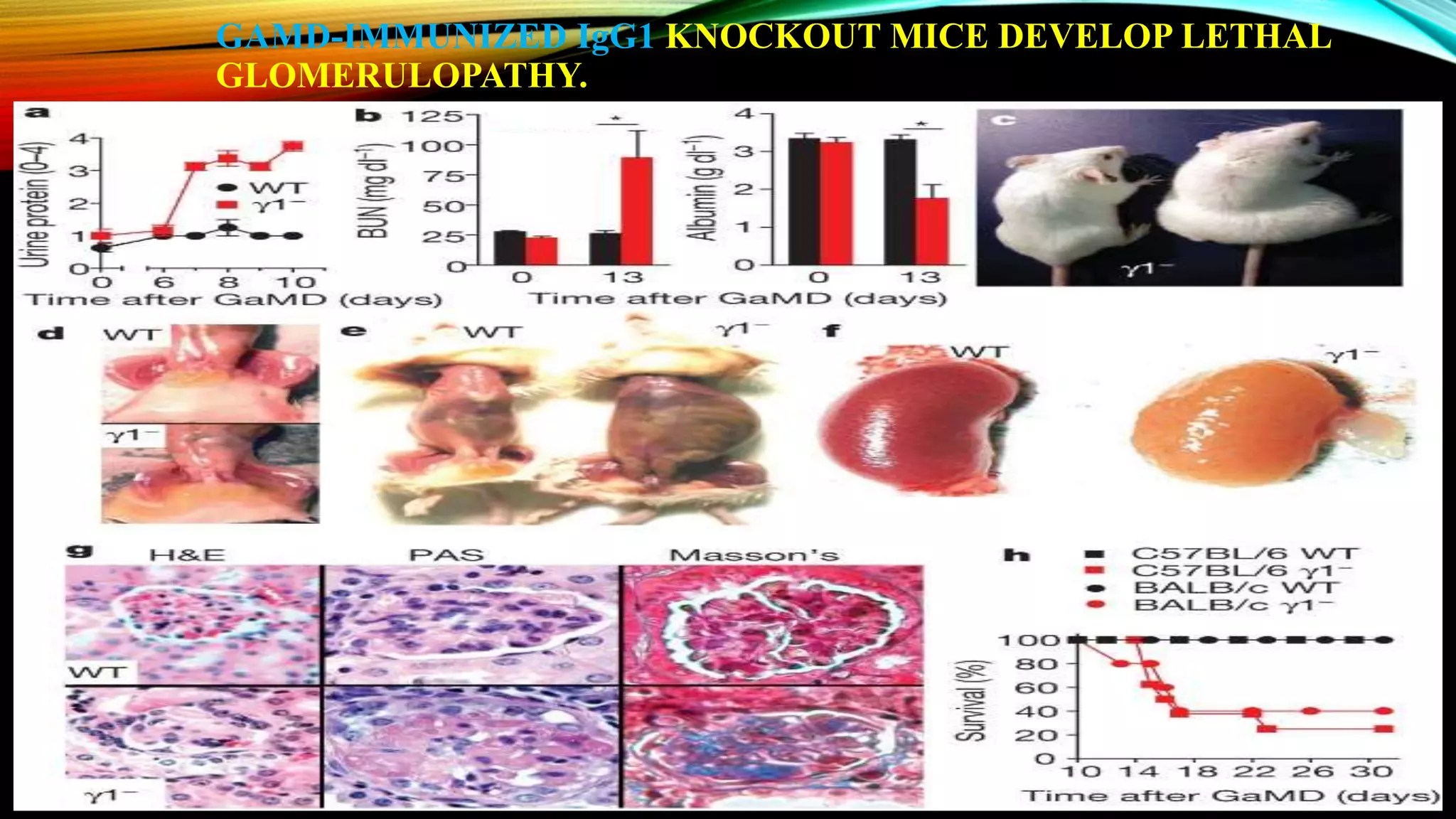
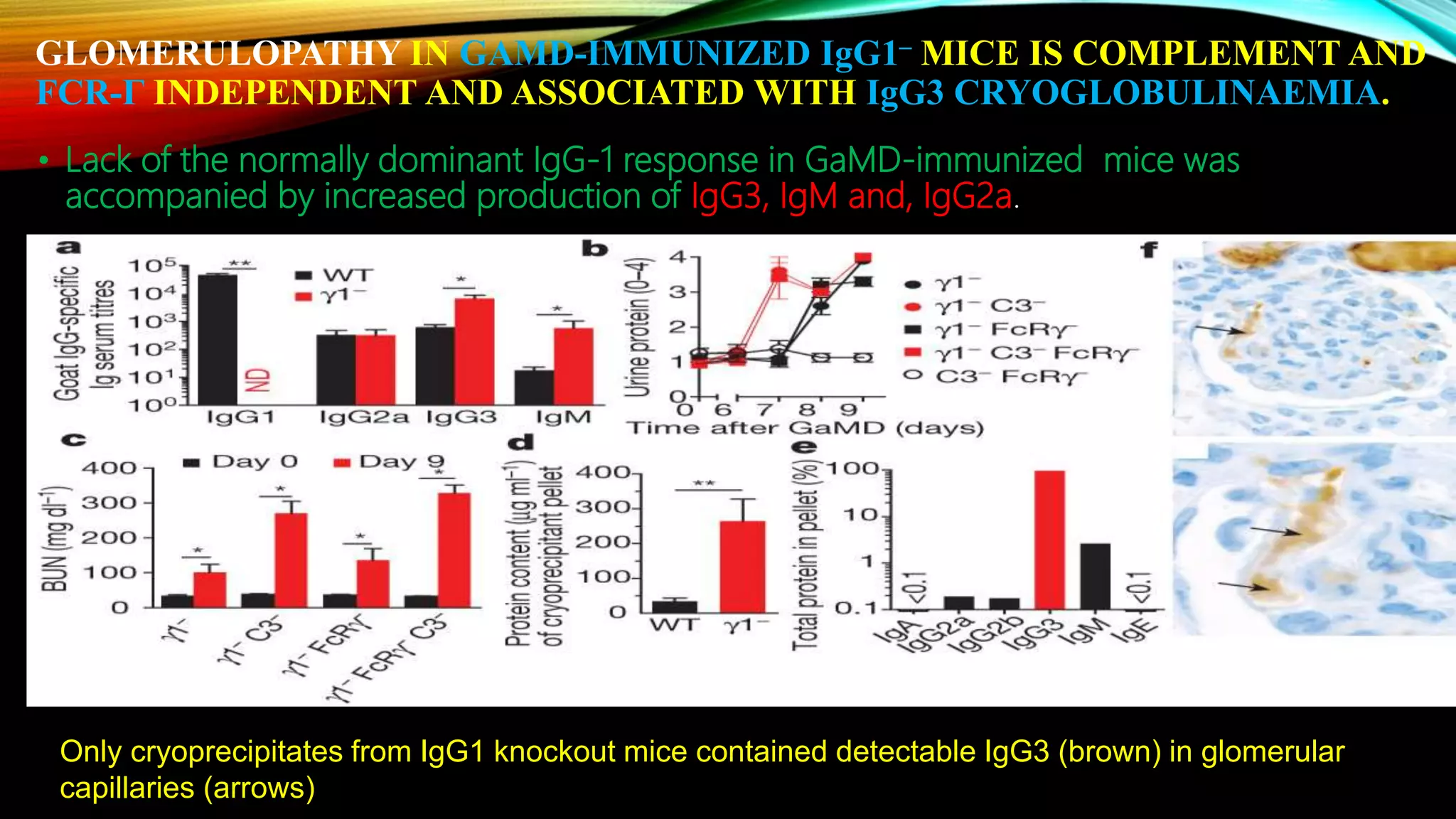
Knockout mice are genetically engineered mice that have had specific genes inactivated through gene targeting. This document provides an overview of knockout mice, including their history, generation process, and uses. It describes how embryonic stem cells are isolated and genetically modified through homologous recombination before being injected into blastocysts to generate chimeric mice. Knockout mice are valuable research tools for studying gene function and modeling human diseases. They have contributed significantly to our understanding of immunology and the development of humanized antibody therapies.