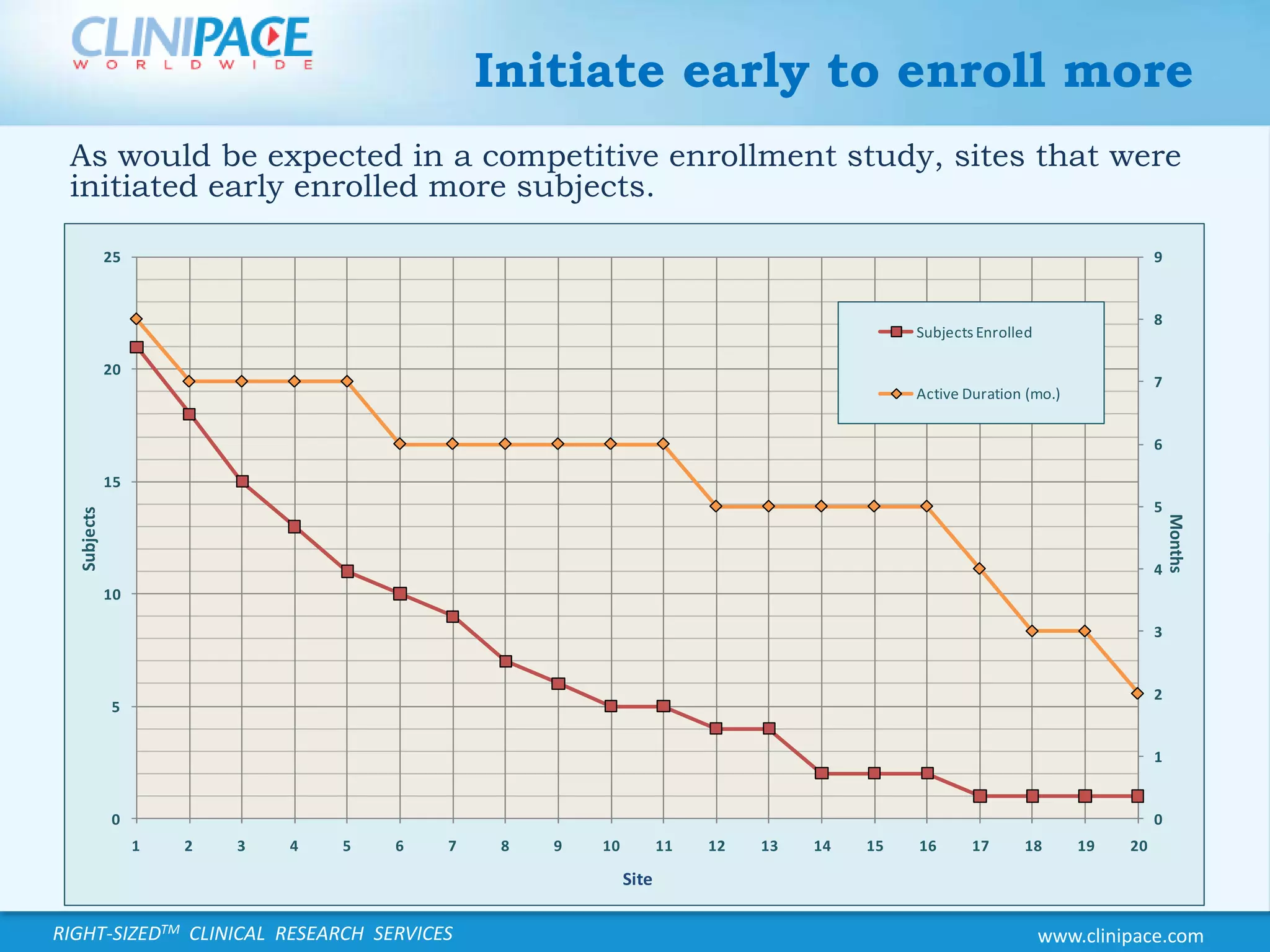
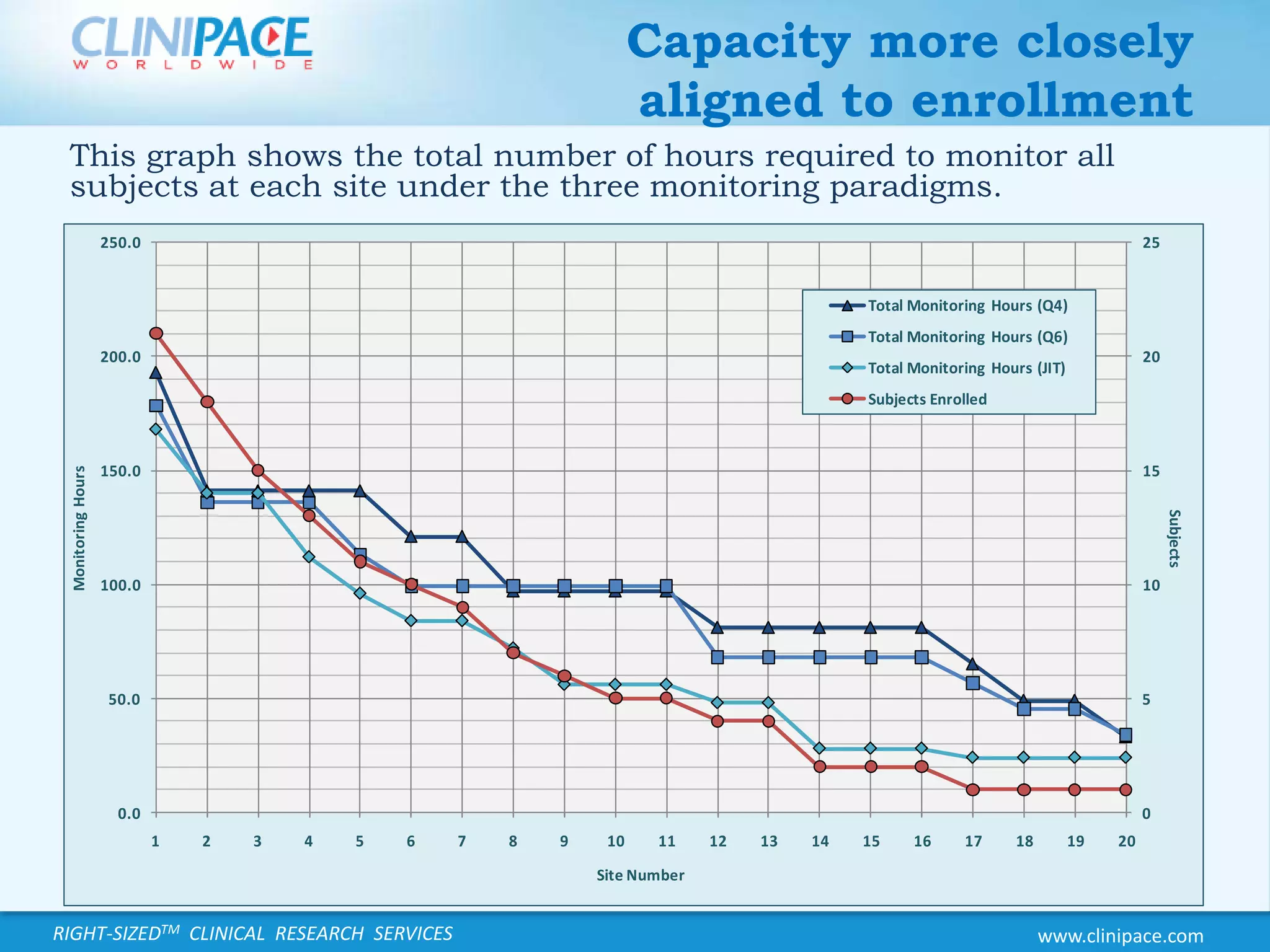
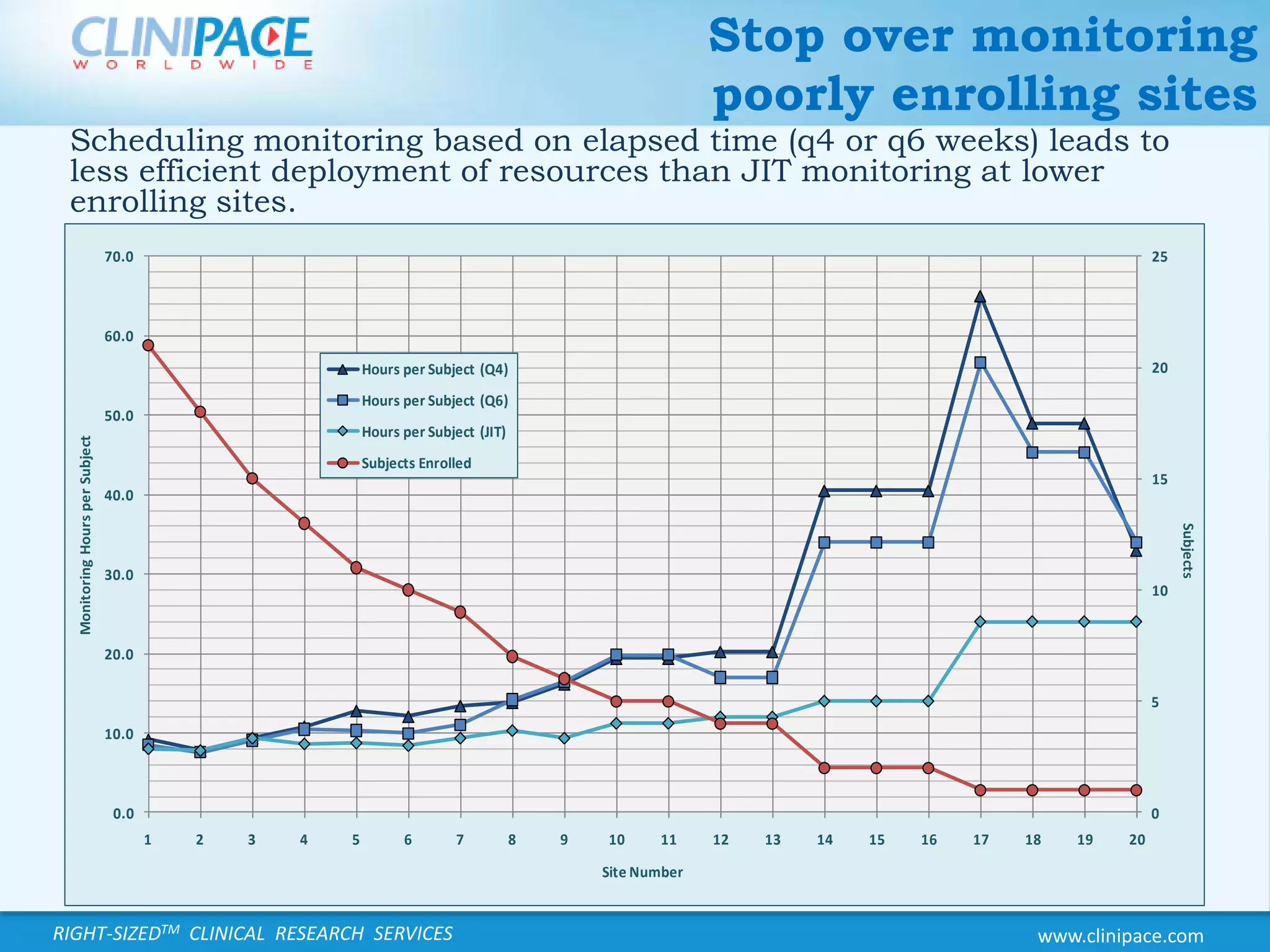
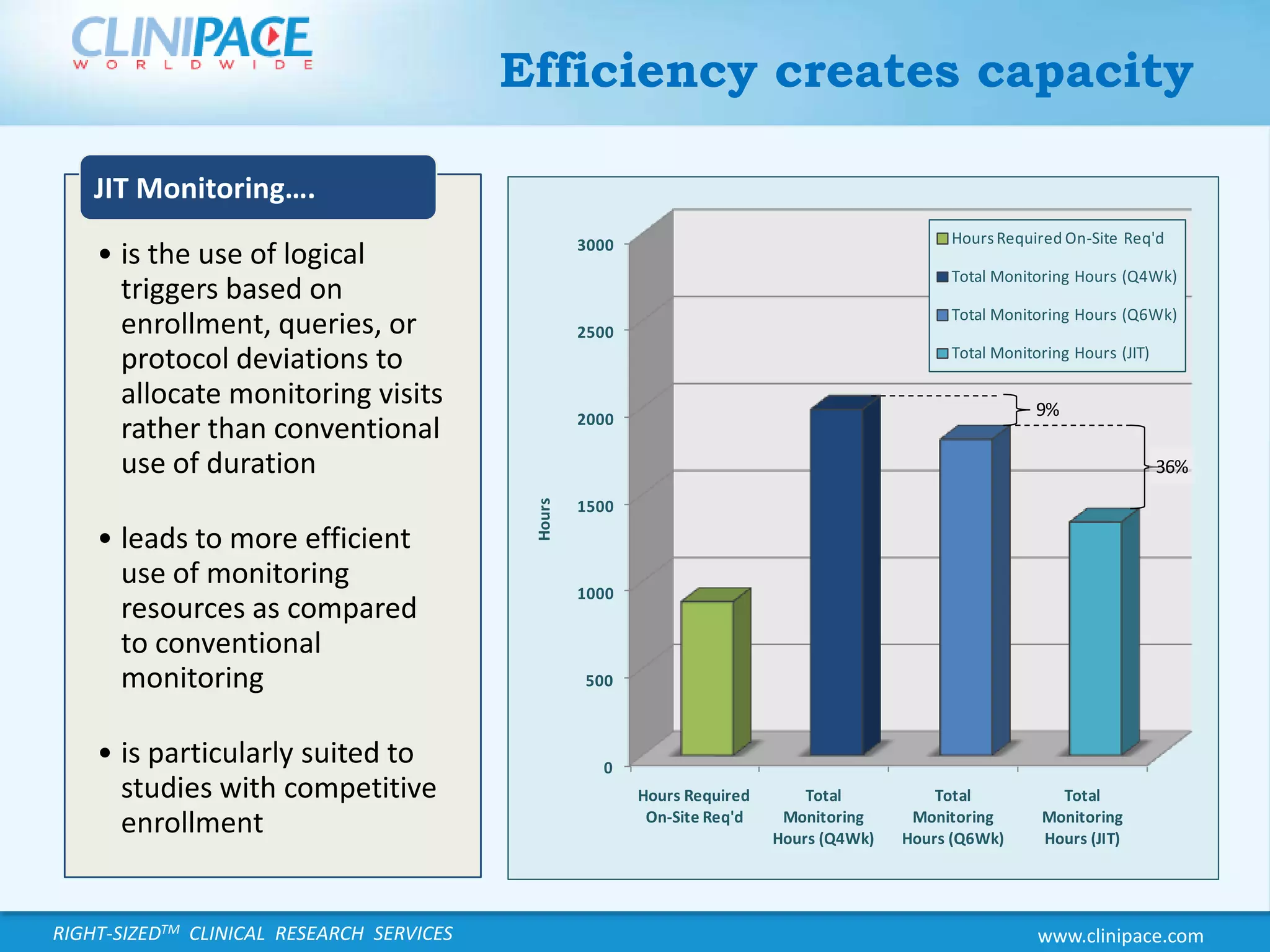
The document discusses the benefits of just-in-time monitoring in clinical trials, emphasizing its potential to enhance efficiency, reduce costs, and improve site relationships. By shifting from a fixed schedule to an event-driven approach, trials can identify issues early and maintain better compliance with ethical standards. Additionally, this method promotes remote monitoring, reducing travel for clinical research associates and ultimately leading to higher job satisfaction.


![Why are we here?Clinical trials are complex [and expensive], and good monitoring can positively impact on site performance, timeline, and budget.Study Start-upStudy AnalysisStudy PlanningStudy ReportingStudy Execution!!!!!!!!!!Moving to Just-in-Time Monitoring can make a significant impact on study execution](https://image.slidesharecdn.com/just-in-time-clinical-trial-monitoring-100421140315-phpapp02/75/Just-in-time-clinical-trial-monitoring-3-2048.jpg)