1) There are two methods for analyzing kinetic data - the integral method and the differential method.
2) The integral method involves guessing a rate equation and integrating to predict a linear concentration-time relationship. The differential method analyzes rate directly from concentration-time data without integration.
3) An example reaction is analyzed using both methods. The integral method indicates first-order kinetics while the differential method derives a rate constant.

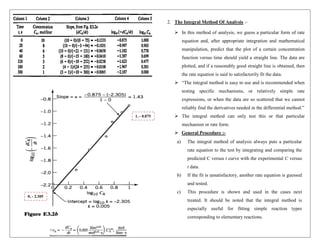


![To evaluate the rate constant, take any point on the CA vs. t
curve. Pick CAo = 10, for which tf = 18.5 s.
Replacing all values into Eq. (i) gives -
2. Rearranging above, we get –
𝐥𝐧([𝐀]𝟏𝐭)/𝟎. 𝟐) = −𝐊𝟏𝐭 (Straight line eqn : - y = mx + c)
Time (min) [A]1t (mol/L)
ln([A]1t)/[A10])
Or
ln([A]1t)/0.2)
0 0.2 0
10 0.15 -0.124938737
20 0.1 -0.301029996
30 0.07 -0.455931956
40 0.05 -0.602059991
DATA TABLE ASSUMING REACTION 1 IS A FIRST ORDER
REACTION
18.5, 8](https://image.slidesharecdn.com/integralmethod-240223033629-e9a255e5/85/Integral-method-to-analyze-reaction-kinetics-6-320.jpg)
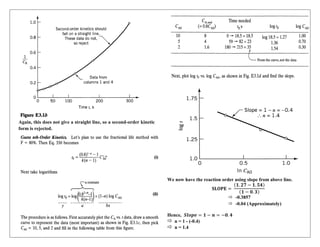
![Analysis for Reaction 1:
The plot shows a clear linear relationship, confirming first-order
kinetics. Therefore, the rate expression for Reaction 1 is:
But, the value of K1 will be –
𝐾1 = (− 𝑆𝑙𝑜𝑝𝑒)
(𝐅𝐨𝐫 𝐞𝐪𝐮𝐚𝐭𝐢𝐨𝐧 𝐨𝐟 𝐬𝐭𝐫𝐚𝐢𝐠𝐡𝐭 𝐥𝐢𝐧𝐞 𝐰𝐢𝐭𝐡 𝟐 𝐩𝐨𝐧𝐭 𝐤𝐧𝐨𝐰𝐧) −
𝐬𝐥𝐨𝐩𝐞 = [
(𝐲𝟐−𝐲𝟏)
(𝐱𝟐−𝐱𝟏)
]
[
(−𝟎.𝟔−𝟎)
(𝟒𝟎−𝟎)
] = -0.015
Hence, the value of K1 will be –
𝐾1 = (− 𝑆𝑙𝑜𝑝𝑒) = −(−0.015)
0.015
Therefore, the rate expression for Reaction 1 is:
(−𝐫𝐀) = 𝟎. 𝟎𝟏𝟓 𝐂𝐀
𝟏
ln([A]1t)/0.2)
ln([A]1t)/0.2)](https://image.slidesharecdn.com/integralmethod-240223033629-e9a255e5/85/Integral-method-to-analyze-reaction-kinetics-7-320.jpg)