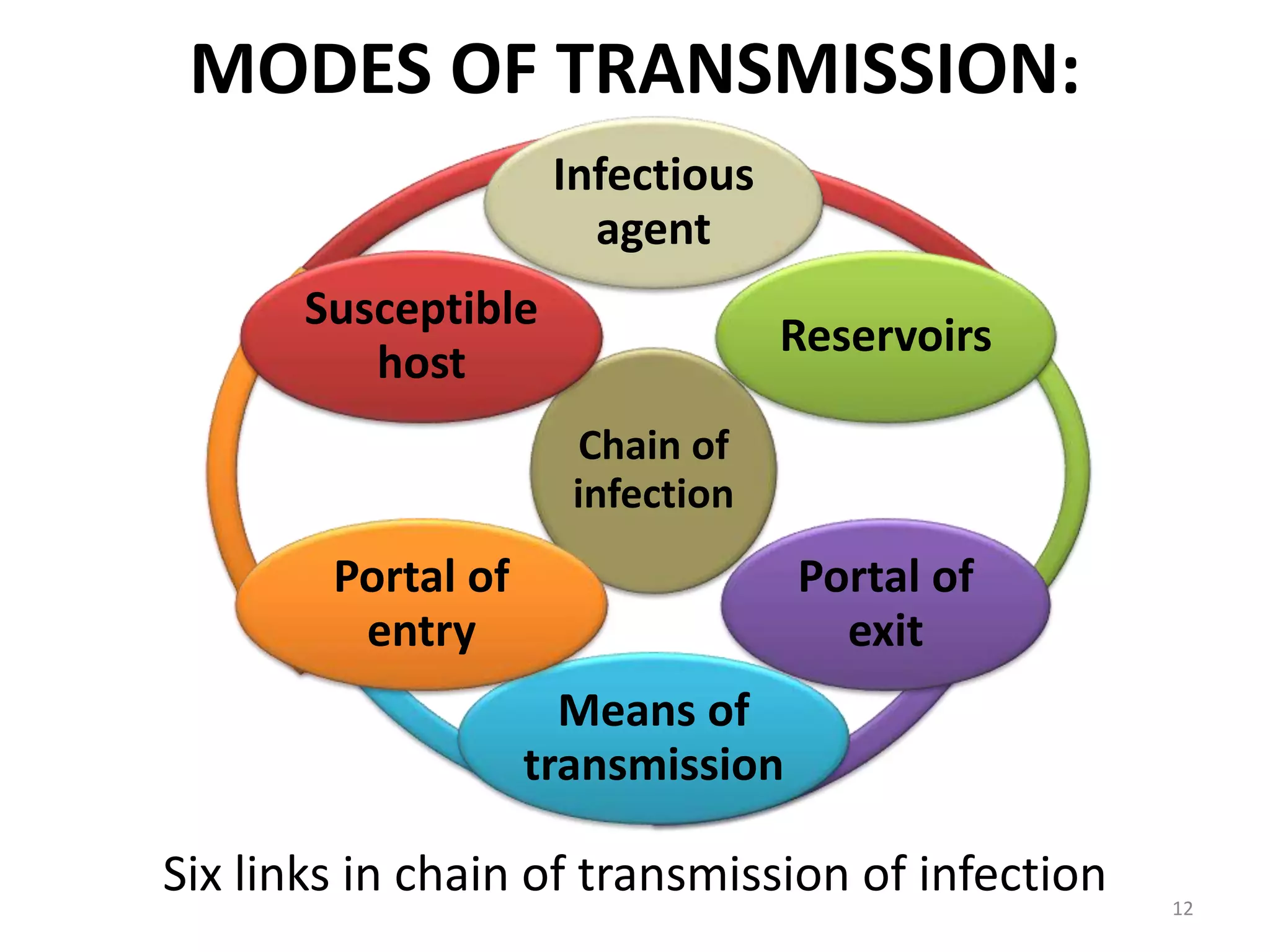
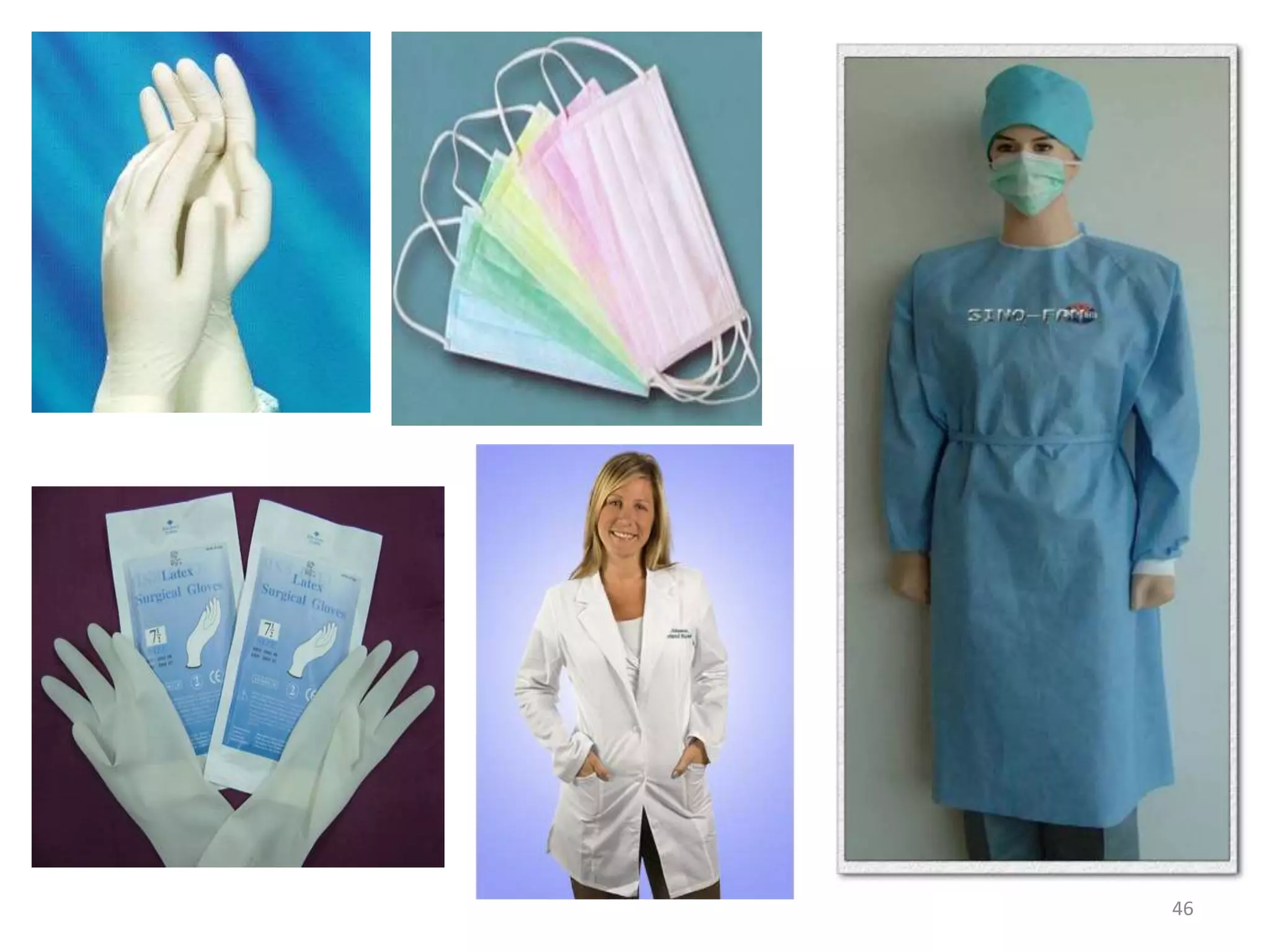
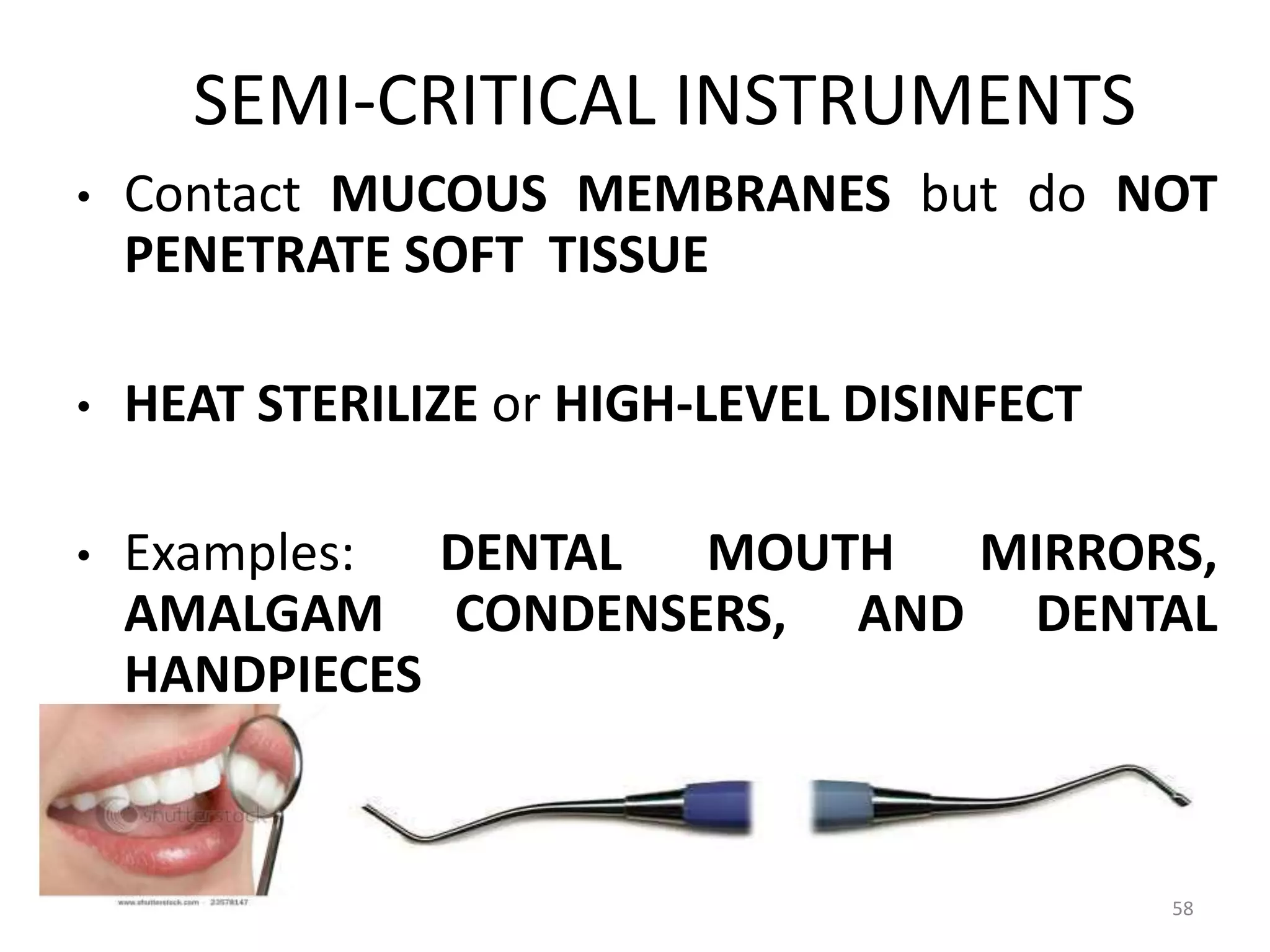
This document discusses infection control in dentistry. It defines key terms like sterilization, disinfection, asepsis, and provides the objectives and importance of infection control. The modes of disease transmission are described. Infection control involves proper sterilization and disinfection of instruments, protective barriers, immunization, and waste disposal. Proper hand hygiene and use of PPE is essential. Infection control guidelines help protect dental staff and patients from exposure to pathogens.