1. The document discusses the history, principles, anatomy, physiology, indications, and complications of intra-aortic balloon pumps (IABPs).
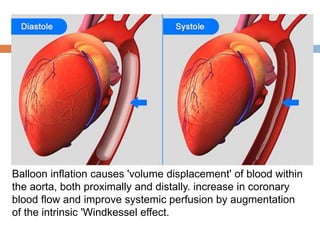
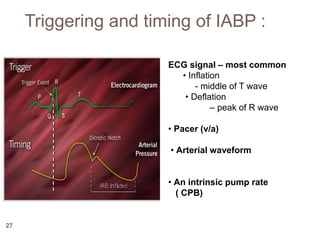
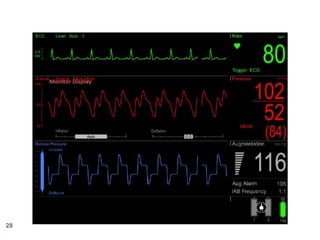
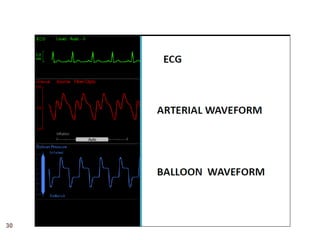
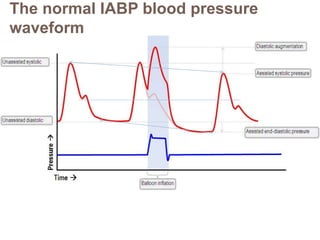
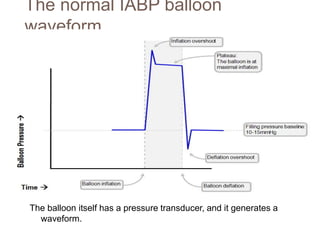
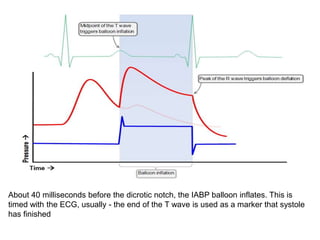
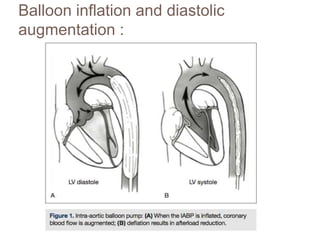
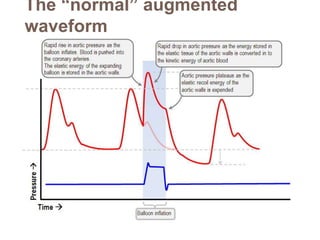
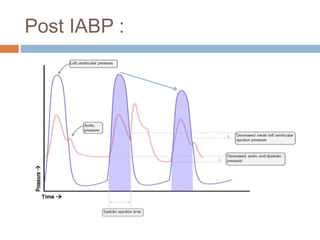
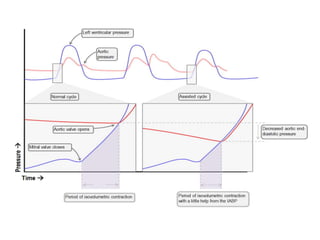
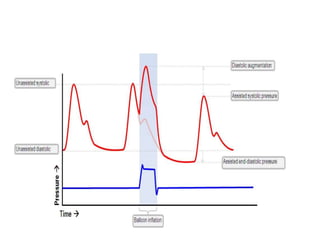
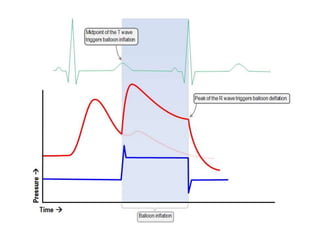
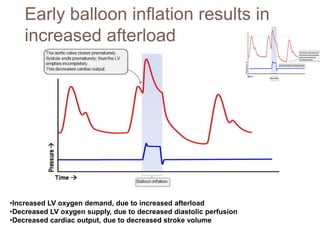
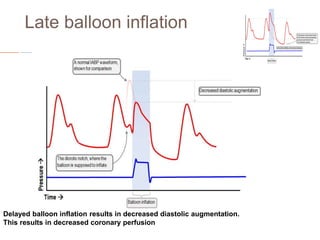
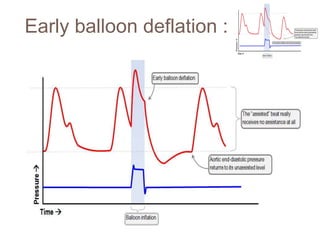
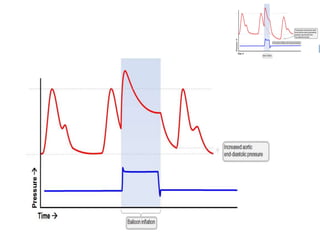
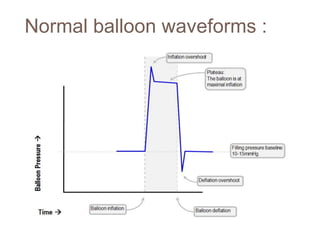
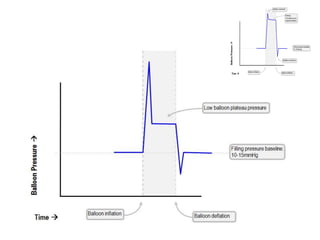
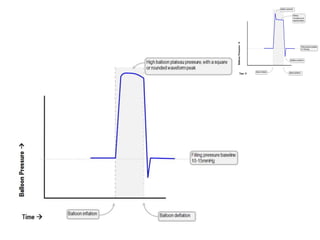
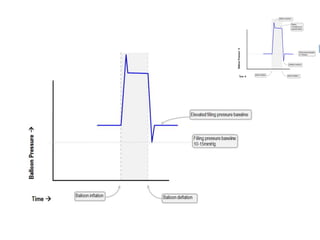
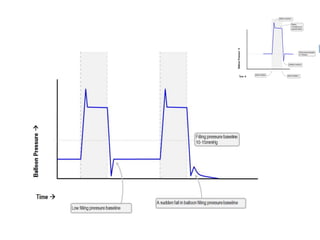
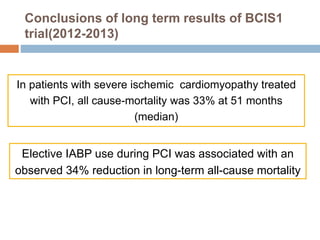
2. IABPs were developed in the 1950s and work by inflating during diastole to increase coronary blood flow and deflating during systole to reduce workload on the heart.
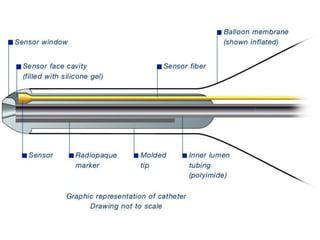
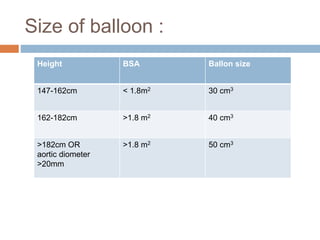
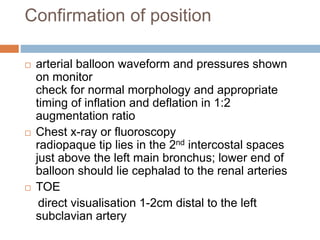
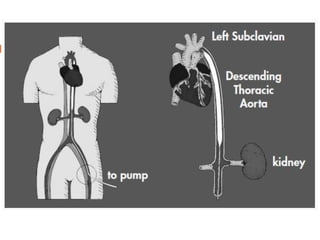
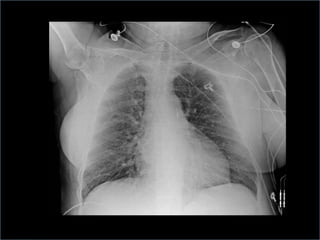
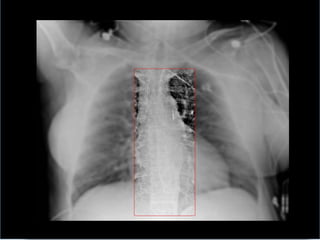
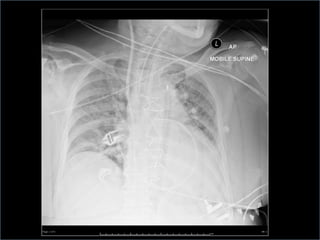
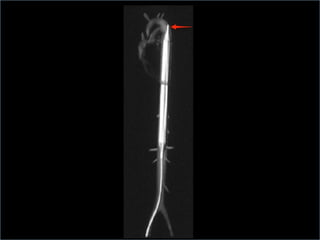
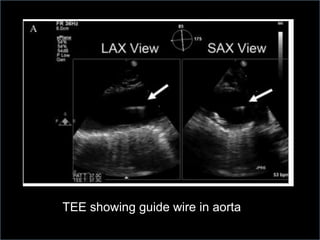
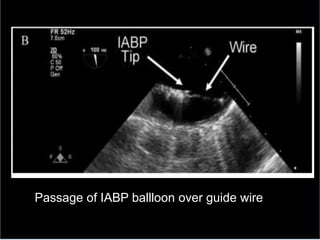
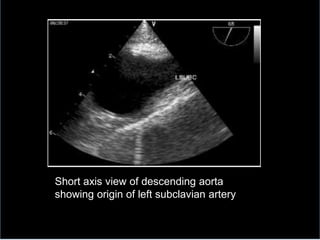
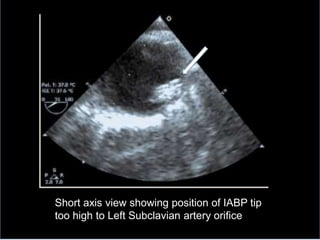
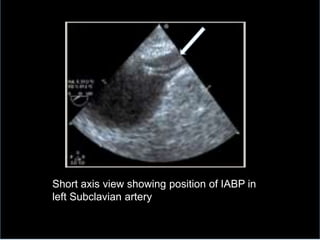
3. Proper positioning of the IABP catheter is important for effectiveness with tips typically placed just above the left main bronchus.
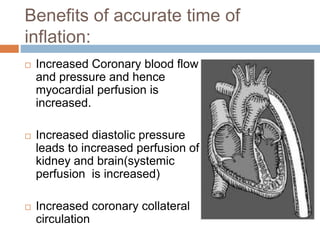
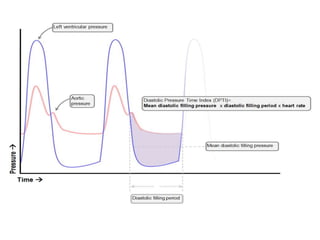
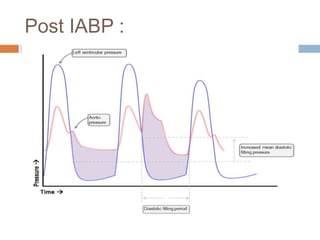
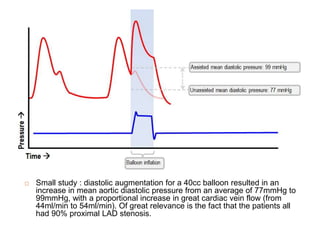
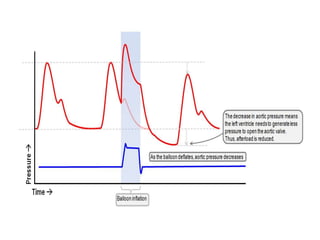
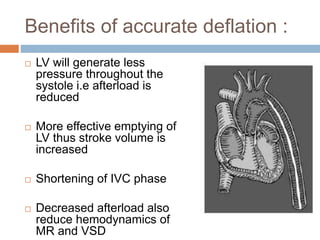
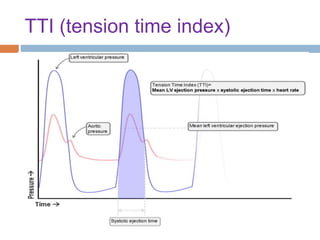
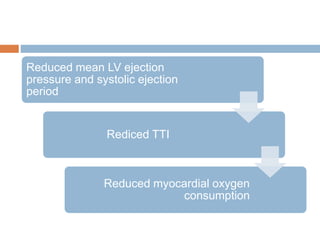
4. IABPs reduce myocardial oxygen demand and increase coronary blood flow by lowering systolic pressure and increasing diastolic pressure and flow.