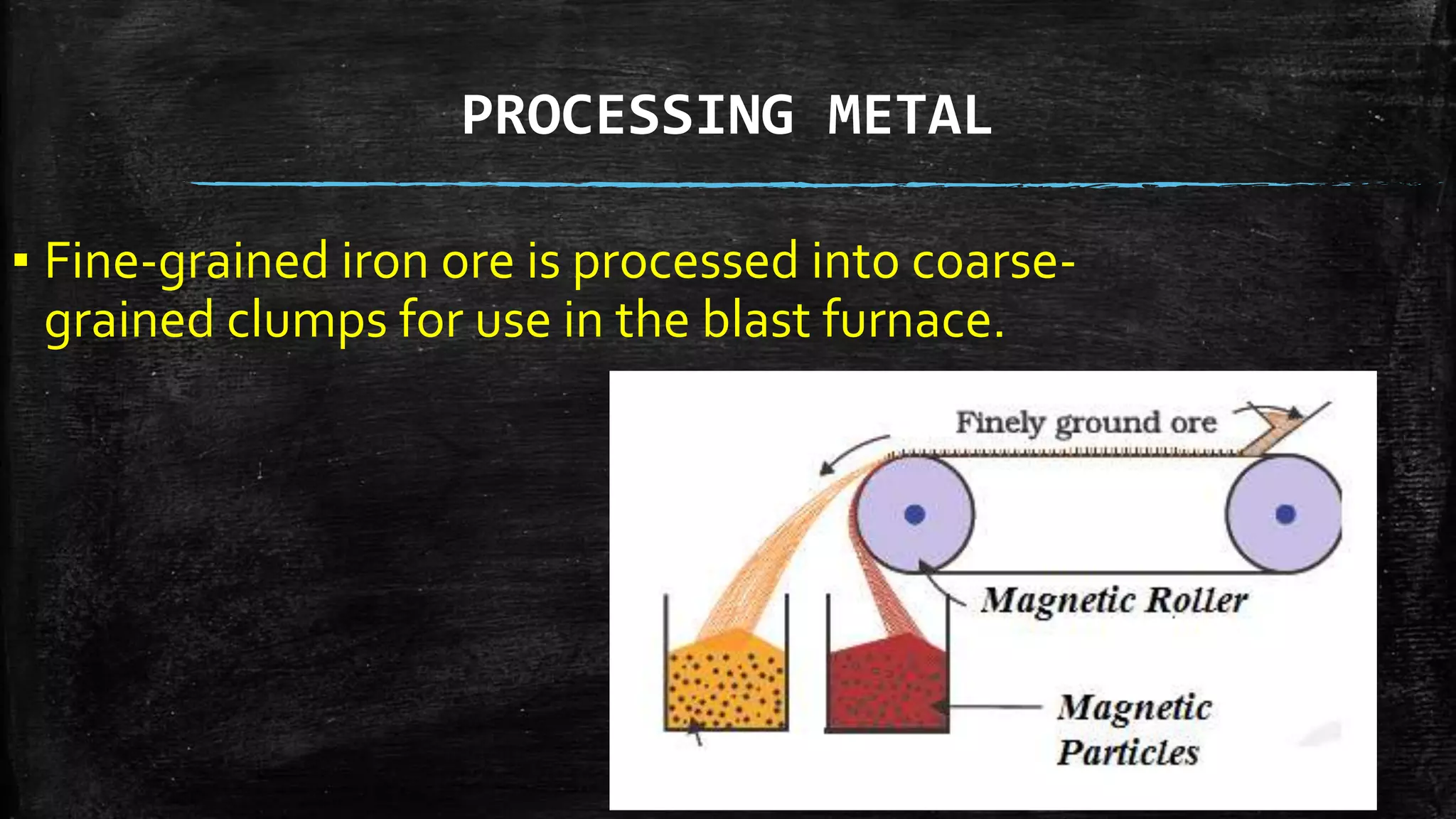
Metals occur naturally in the Earth's crust and most are found as ores, which are metal compounds. The 6 metals first used by prehistoric humans were gold, silver, lead, copper, tin, and iron. Metals are extracted from ores through smelting and can be further refined. Steel is an alloy of iron with 0.5-1.5% carbon, making it not a pure metal. Metals are strong, conductive materials that are important for tools, infrastructure, and more due to their properties.


























![WHAT IS THE DIFFERENCE BETWEEN
METAL AND STEEL?
▪ A metal is a chemical element of various opaque,
fusible, ductile and lustrous substances [source:
Merriam-Webster].
▪ The elements referred to in that definition are the
same ones you studied when you learned about the
periodic table of the elements in high school
chemistry. Some common metals are titanium,
copper and nickel.](https://image.slidesharecdn.com/howmetalsaremade-160221081724/75/How-metals-are-made-27-2048.jpg)