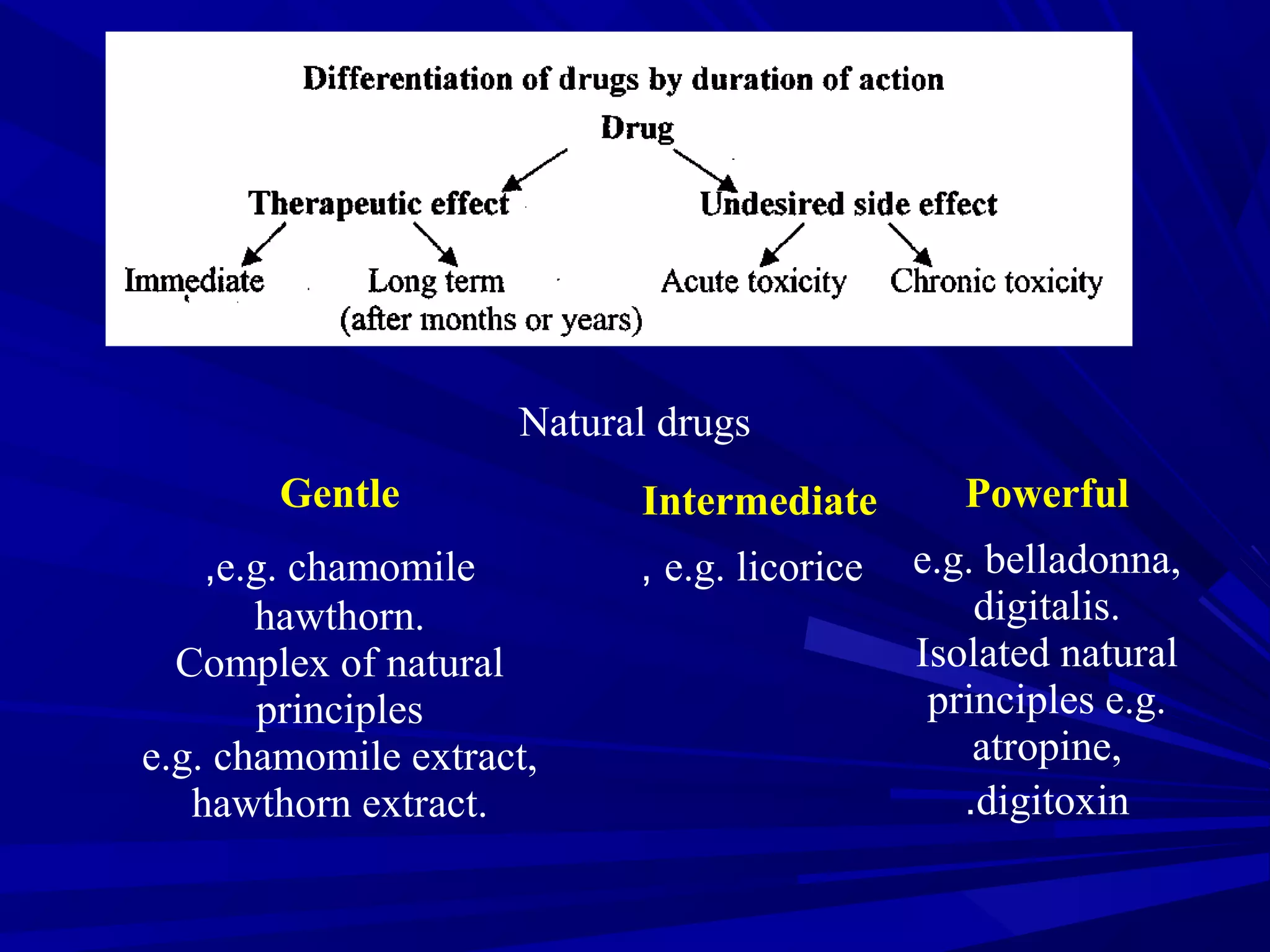
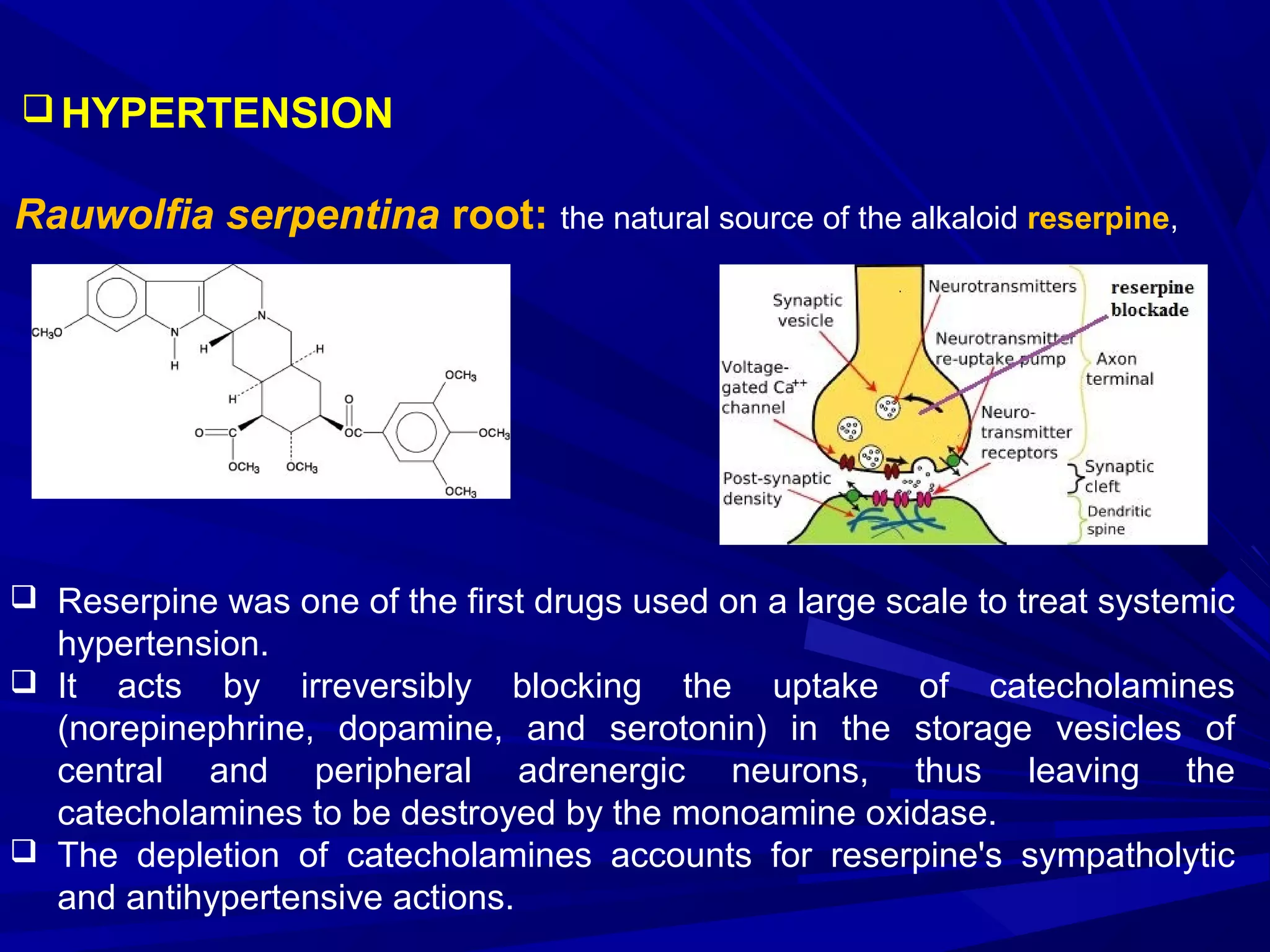
Herbal medicine, a type of alternative medicine, utilizes plants and their crude products for treating diseases, differing from conventional medicine which relies on pure chemicals. It encompasses various practices and concepts such as phytochemistry, standardized extracts, and the therapeutic categories for conditions like cardiovascular and gastrointestinal diseases. Quality control is crucial in herbal medicine, focusing on identity, purity, efficacy, and safety, while many conventional drugs have origins in plant-derived substances.