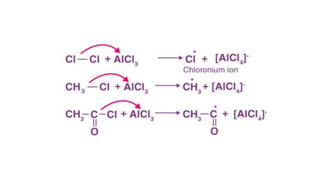
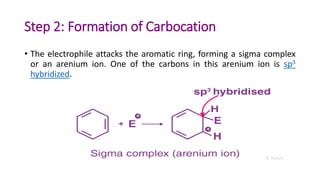
Electrophilic substitution reactions involve an electrophile replacing a functional group, typically a hydrogen atom, on an organic compound. There are two main types: electrophilic aromatic substitutions, where the electrophile replaces an atom in an aromatic ring, and electrophilic aliphatic substitutions, where the electrophile replaces a group on an aliphatic compound. Both proceed by a three step mechanism of electrophile generation, carbocation formation, and proton removal to restore aromaticity.








![• Step 3: Removal of Proton
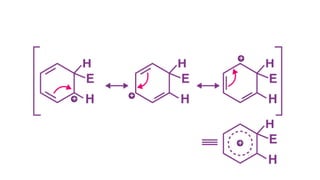
• In order to restore the aromatic character, the sigma complex
releases a proton from the sp3 hybridized carbon when it is attacked
by the [AlCl4]–. The reaction describing the removal of a proton from
the sigma complex is given below:](https://image.slidesharecdn.com/electrophilicsubstitutionreaction-230225082135-4a03f9b7/85/Electrophilic-substitution-reaction-pptx-12-320.jpg)

