Embed presentation
Download to read offline




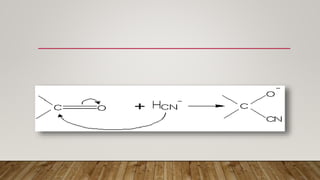

The electromeric effect refers to the temporary polarity produced in a multiple bonded compound when approached by a reagent, and only occurs in the presence of that reagent. For example, in ethylene the pi electrons of the carbon-carbon double bond completely transfer to one carbon atom when attacked by an electrophile, disturbing the molecular orbital symmetry. Addition then follows Markownikoff's rule, with the negative portion of the reagent adding to the carbon with fewer hydrogen atoms.





