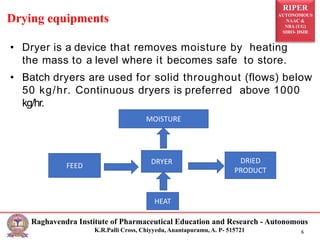
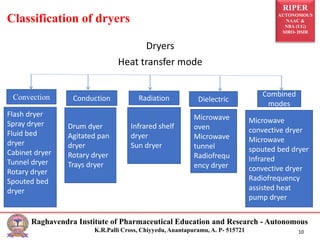
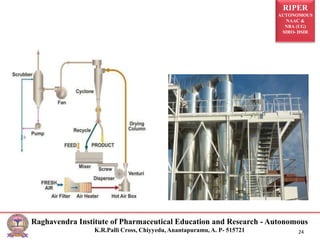
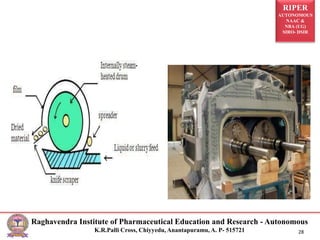
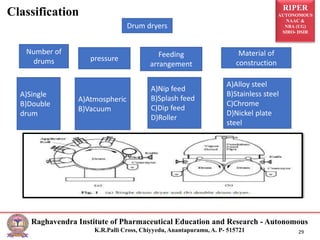
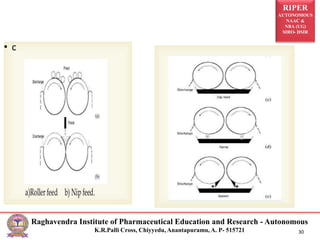
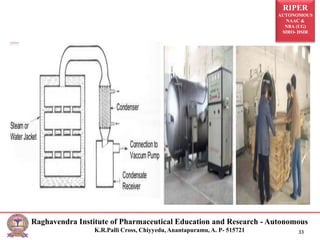
The document provides an overview of drying processes and equipment used in pharmaceutical and industrial applications. It details the purpose and benefits of drying, classification of dryers, and specific types like spray, tray, fluidized bed, flash, drum, and vacuum dryers, along with their advantages and disadvantages. Additionally, it discusses general considerations for choosing dryers and applications in various industries.