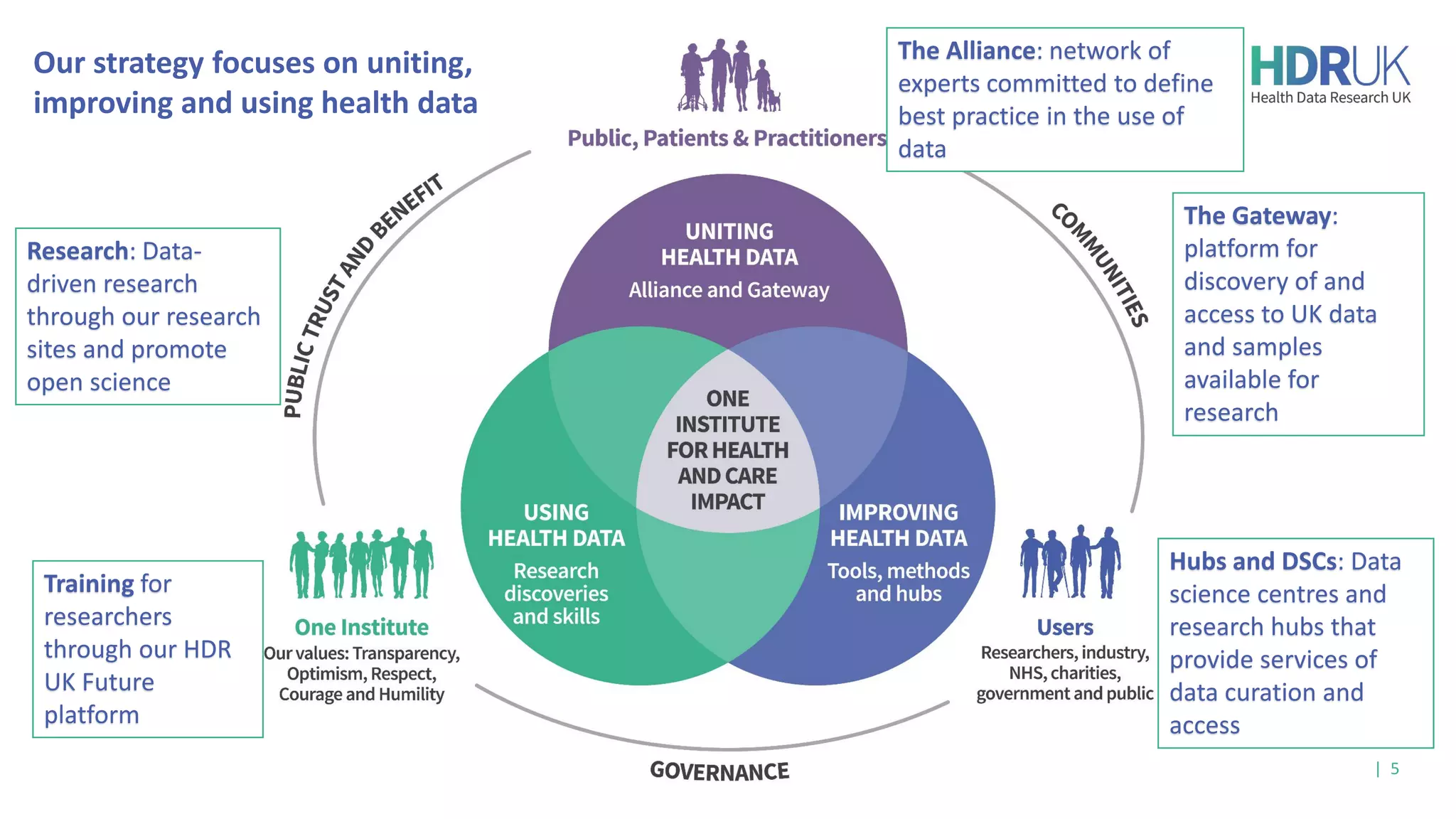
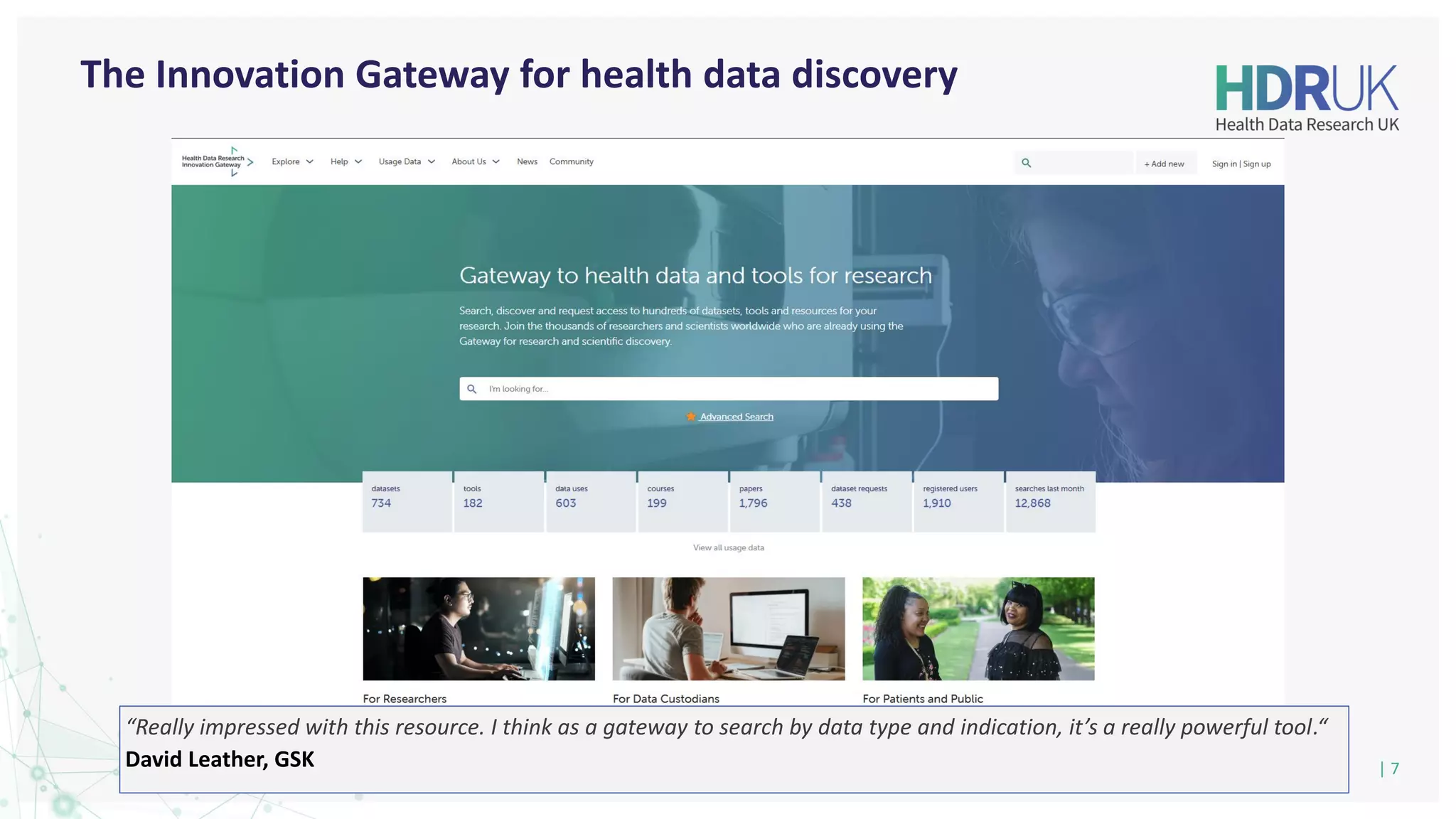
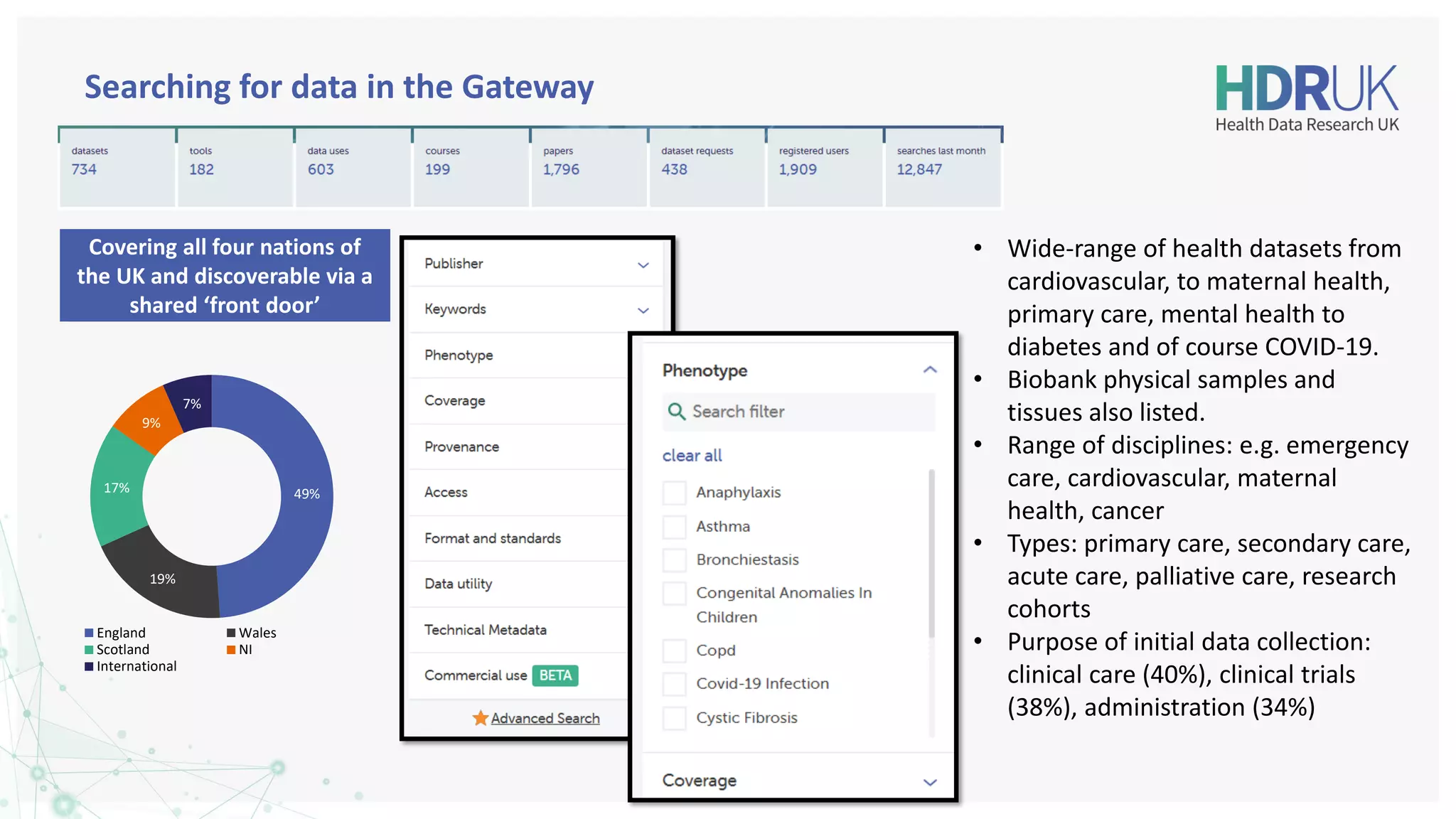
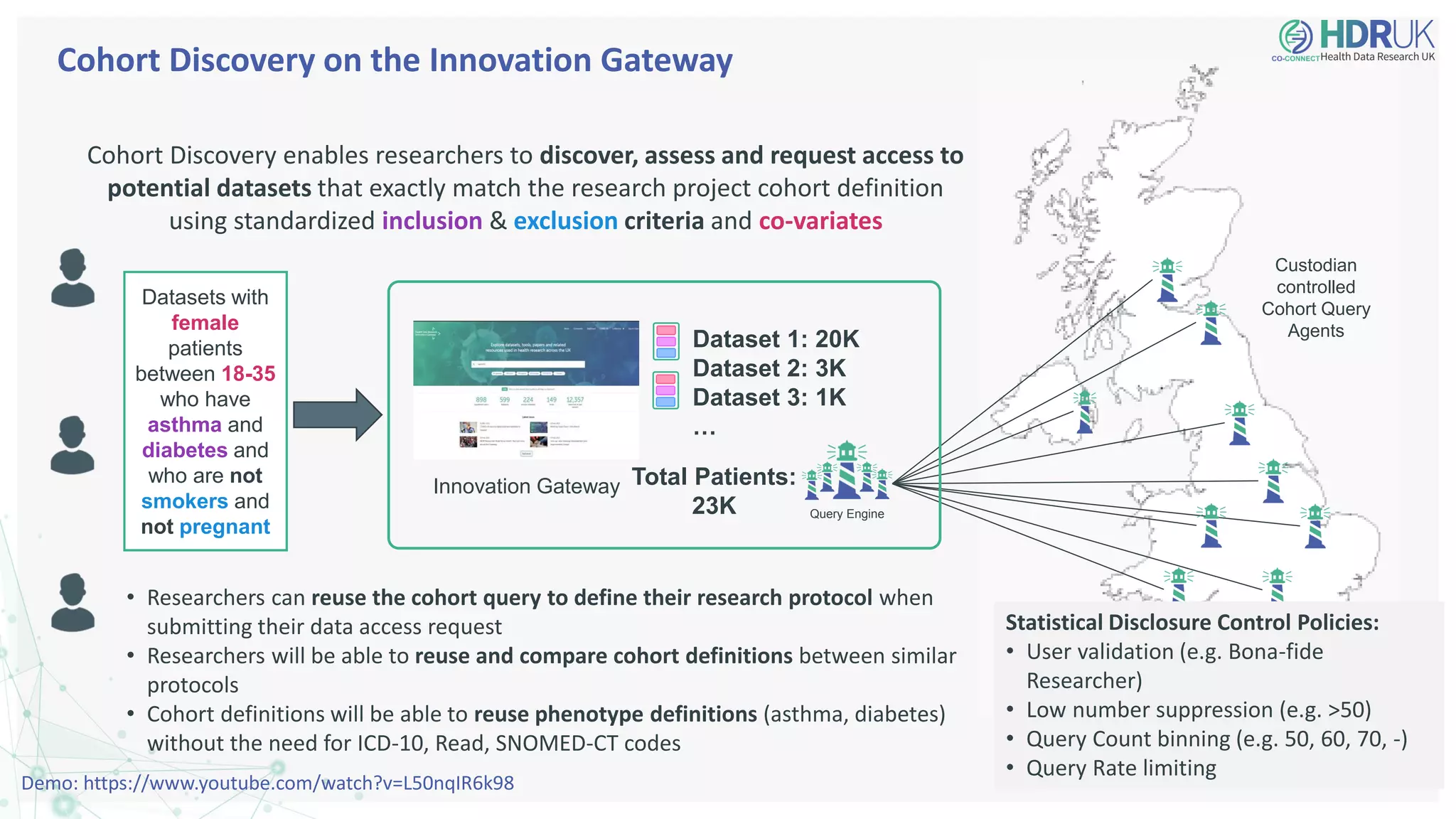
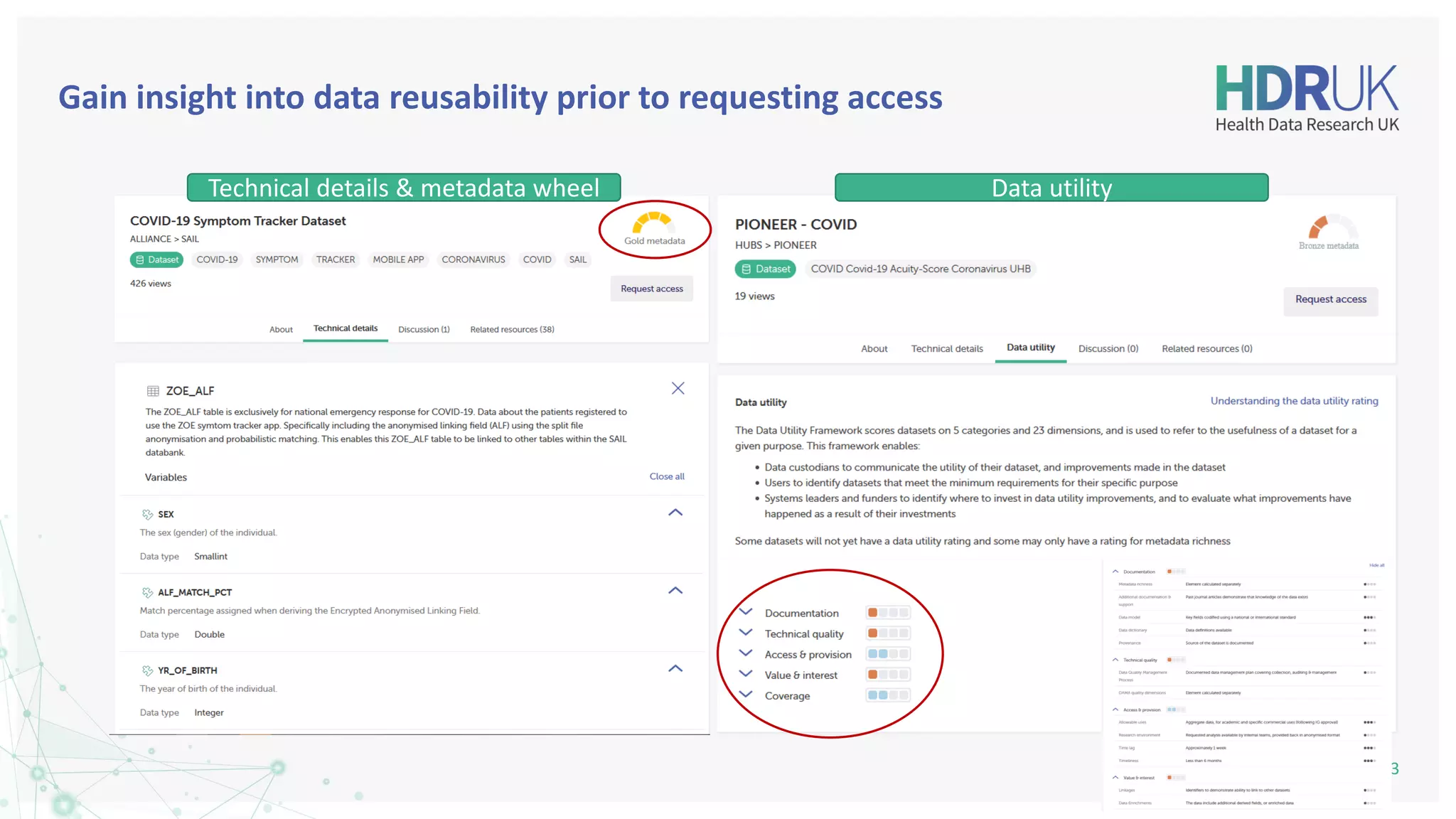
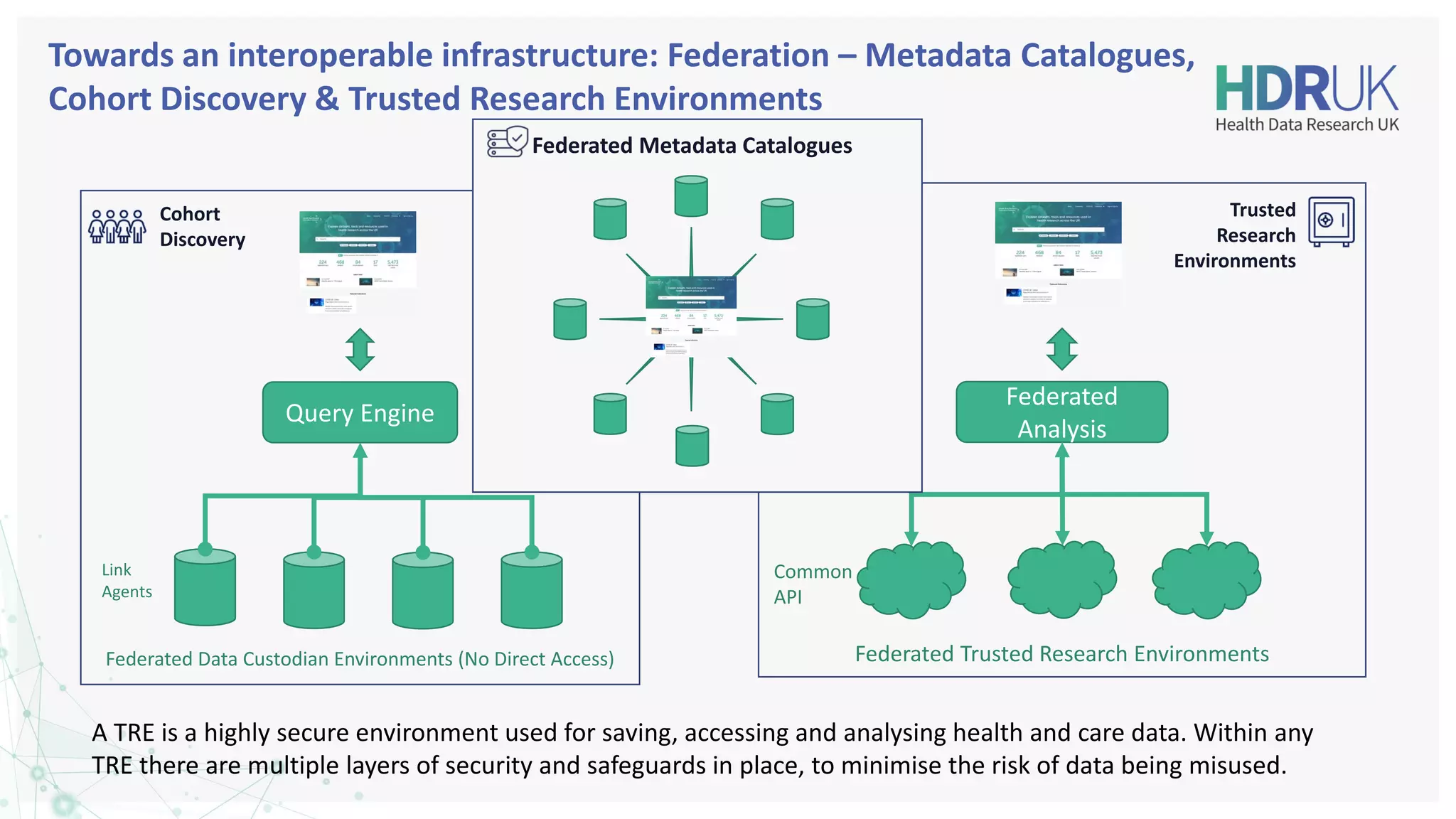
The document outlines the efforts of Health Data Research UK to unite the UK's health data for better access and discovery in health data research. It emphasizes the importance of improving findability, accessibility, interoperability, and reusability of health data, while actively involving the public and patients in the process. The Innovation Gateway is highlighted as a key platform for researchers to discover and access diverse health datasets, supporting data-driven research for improved health outcomes.