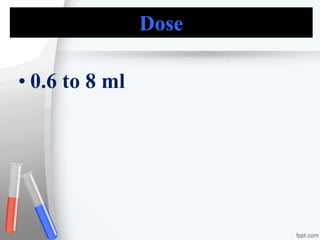
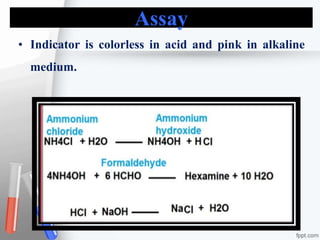
The document provides an overview of acidifiers used in pharmaceutical inorganic chemistry, detailing their types, preparation methods, properties, and applications. It categorizes acidifiers into gastric, urinary, and systemic types, describing their role in adjusting acidity levels in various body fluids. Additionally, it includes specific preparations for dilute hydrochloric acid and ammonium chloride, including their assays, uses, and storage conditions.