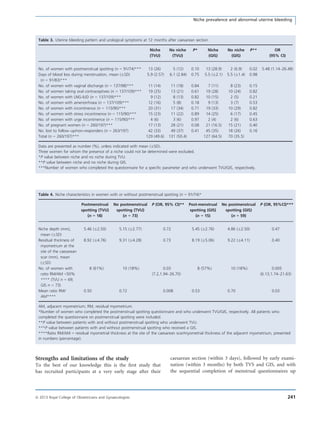
This study examined the prevalence of niches in the caesarean section scar and their relationship to abnormal uterine bleeding and urinary incontinence. The study used transvaginal ultrasound and gel instillation sonohysterography to examine 263 women 6-12 weeks after cesarean section. It found that niches were present in 49.6% of women when examined by ultrasound and 64.5% of women when examined by sonohysterography. Women with niches detected by sonohysterography reported more postmenstrual spotting. Women with thinner residual myometrium at the scar site also reported more postmenstrual spotting. No relationship was found between niches and urinary incontinence.






![References
1 Stichting Perinatale Registratie Nederland. Perinatale zorg in
Nederland 2008. Utrecht: Stichting Perinatale Registratie Nederland.
2011 [www.perinatreg.nl]. Accessed 25 April 2013.
2 [http://www.cdc.gov/nchs/data/nvsr/nvsr61/nvsr61_05.pdf] Accessed
29 April 2013.
3 Silver RM. Implications of the first cesarean: perinatal and future
reproductive health and subsequent cesareans, placentation issues,
uterine rupture risk, morbidity, and mortality. Semin Perinatol
2012;36:315–23.
4 Miller ES, Hahn K, Grobman A. Consequences of a primary elective
Cesarean Delivery Across the Reproductive Life. Obstet Gynecol
2013;121:789–97.
5 Naji O, Abdallah Y, Bij de Vaate AJ, Smith A, Pexsters A, Stalder C,
et al. Standardised approach for imaging and measuring Cesarean
section scars using ultrasonography. Ultrasound Obstet Gynecol
2012;39:252–9.
6 Roberghe S, Boutin A, Chaillet N, Moore L, Jastrow N, Demers S,
et al. Systematic review of cesarean scar assessment in the
nonpregnant state: imaging techniques and uterine scar defect. Am
J Perinatol 2012;29:465–72.
7 Monteagudo A, Carreno C, Timor-Tritsch IJ. Saline infusion
sonohysterography in nonpregnant women with previous caesarean
delivery: the “niche” in the scar. J Ultrasound Med 2001;20:1105–
15.
8 Thurmond AS, Harvey WJ, Smith SA. Cesarean section scar as a
cause of abnormal vaginal bleeding: diagnosis by
sonohysterography. J Ultrasound Med 1999;18:13–6.
9 Armstrong V, Hansen WF, Van Voorhis BJ, Syrop CH. Detection of
cesarean scars by transvaginal ultrasound. Obstet Gynecol
2003;101:61–5.
10 Valenzano M, Lijoi D, Mistrangelo E, Constantini S, Ragni N. Vaginal
ultrasound and hysterosonographic evaluation after caesarean
section: correlation with gynaecological symptoms. Gynecol Obstet
Invest 2006;61:216–22.
11 Bij de Vaate AJM, Br€olmann HAM, van der Voet LF, van der Slikke
JW, Veersema S, Huirne JAF. Ultrasound evaluation of the caeserean
scar: relation between a niche and postmenstrual spotting.
Ultrasound Obstet Gynecol 2011;37:93–9.
12 Uppal T, Lanzarone V, Mongelli M. Sonographically dtected
caesarean section scar defects and menstrual irregularity. J Obstet
Gynaecol 2011;31:413–6.
13 Vikhareva Osser O, Jokubkiene L, Valentin L. High prevalence of
defects in Cesarean section scars at transvaginal ultrasound
examination. Ultrasound Obstet Gynecol 2009;34:90–7.
14 Regnard C, Nobusch M, Fellemans C, Benali N, Van Rijsselberghe M,
Rozenberg S. Caesarean section scar evaluation by saline contrast
sonohysterography. Ultrasound Obstet Gynecol 2004;3:289–92.
15 Ofilli-Yebovi D, Ben-Nagi J, Swayer E, Lee C, Gonzalez J, Jurkovic D.
Deficient lower-segment Cesarean section scars: prevalence and risk
factors. Ultrasound Obstet Gynecol 2008;31:72–7.
16 Wang CB, Chiu WW, Lee CY, Sun YL, Lin YH, Tseng CJ. Cesarean
scar defect: correlation between Cesarean section number, defect
size, clinical symptoms and uterine position. Ultrasound Obstet
Gynecol 2009;34:85–9.
17 Janssen CA, Scholten PC, Heintz AP. A simple visual assessment
technique to discriminate between menorrhagia and normal
menstrual blood loss. Obstet Gynecol 1995;85:977–82.
18 Vikhareva Osser O, Jakubkiene L, Valentin l. Cesarean section scar
defects: agreement between transvaginal sosnographic findings with
and without saline contrast enhancement. Ultrasound Obstet
Gynecol 2010;35:75–83.
19 Chen HY, Chen SJ, Hsieh FJ. Observation of caesarean section scar
by transvaginal ultrasonography. Ultrasound Med Biol 1990;16:443–
7. Ultrasound 447, 1990.
20 Chang Y, Tsai EM, Long CY, Lee CL, Kay N. Resectoscopic
treatment combined with sonohysterographic evaluation of women
with postmenstrualbleeding as a result of previous cesarean delivery
scar defects. Am J Obstet Gynecol 2009;200:370.e1–4.
244 ª 2013 Royal College of Obstetricians and Gynaecologists
van der Voet et al.](https://image.slidesharecdn.com/c-150927210940-lva1-app6892/85/C-s-scar-nich-9-320.jpg)