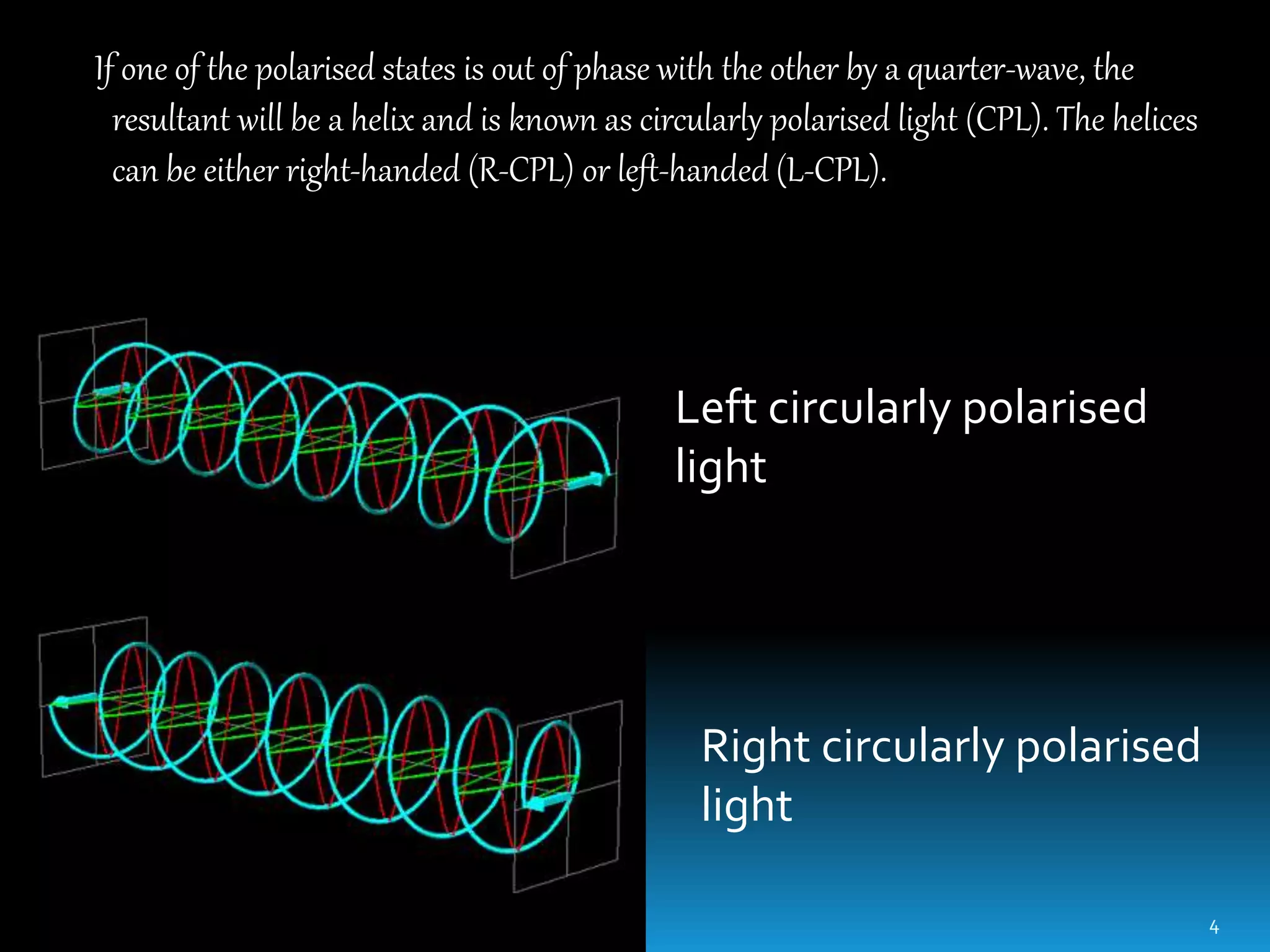
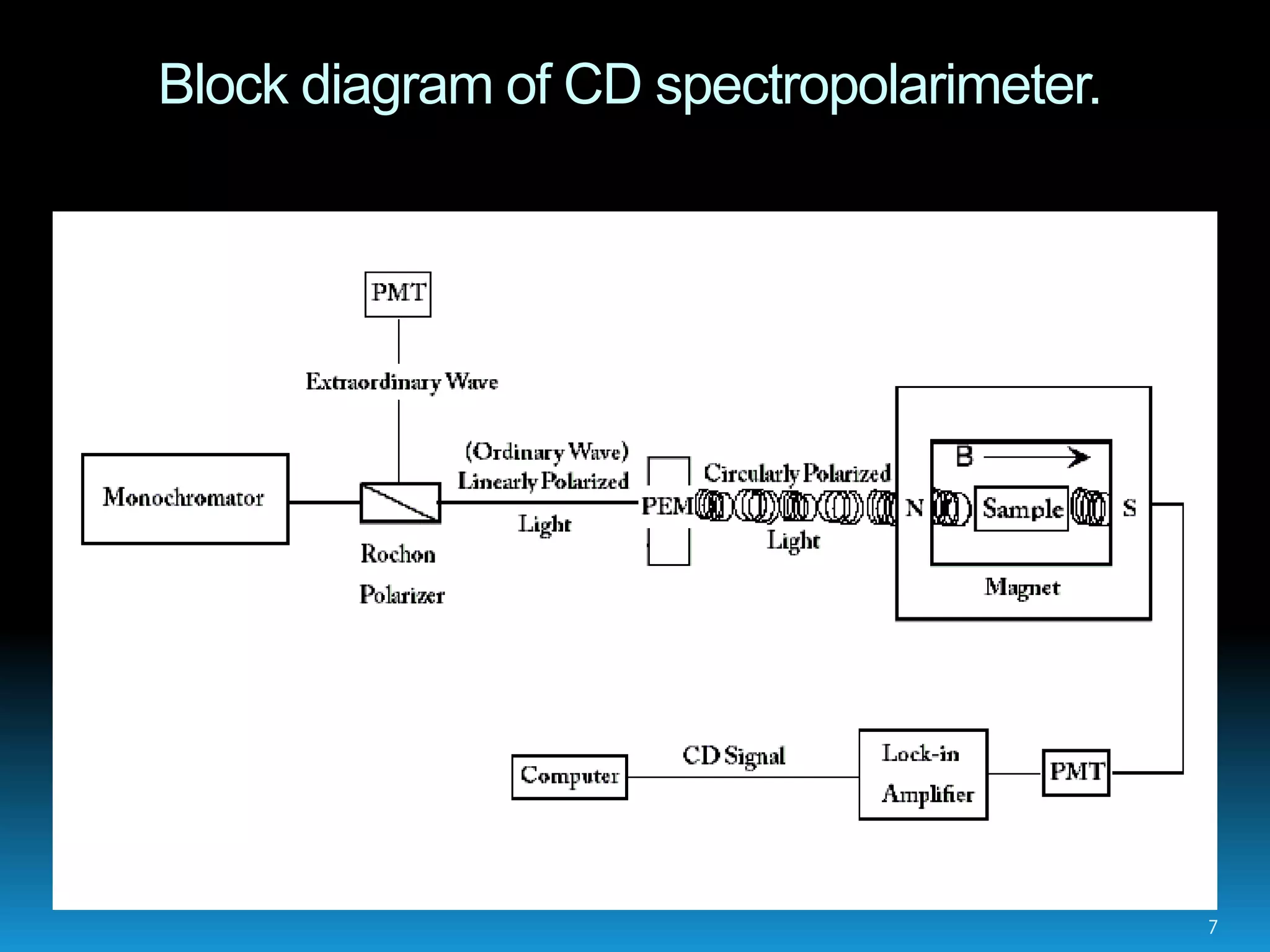
Circular dichroism spectroscopy measures the differential absorption of left and right circularly polarized light by chiral molecules. When light passes through an optically active substance, the left and right circular polarizations are absorbed to different extents. A CD spectrometer contains a light source, monochromator, polarizer, photoelastic modulator and detector. It measures the CD signal as a function of wavelength, providing information about secondary structure of proteins and nucleic acids. CD spectroscopy requires minimal sample amounts and can quickly analyze secondary structure without crystallization. It is useful for studying protein folding, ligand binding and environmental effects on structure.