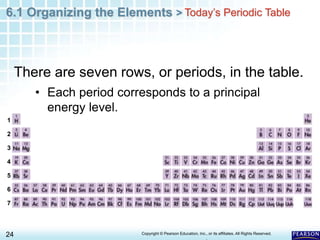
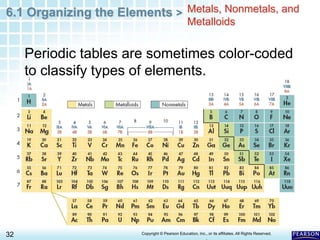
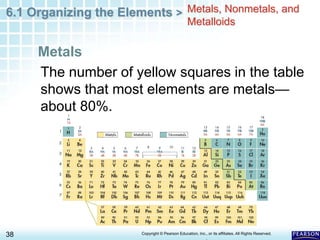
The document discusses the organization of elements in the periodic table. It describes how early chemists like Dobereiner began organizing elements into groups based on their properties. Mendeleev later developed the periodic table by arranging elements in order of increasing atomic mass and placing those with similar properties in the same rows. The modern periodic table is organized by atomic number instead of mass and places elements with similar properties in the same columns. Elements are classified as metals, nonmetals or metalloids based on their general physical and chemical properties.





![6.1 Organizing the Elements >
6 Copyright © Pearson Education, Inc., or its affiliates. All Rights Reserved.
.
Dobereiner noted a pattern in his triads.
One element in each triad tended to have
properties with values that fell midway
between those of the other two elements.
• For example, the average of the atomic
masses of chlorine and iodine is [(35.453 +
126.90)/2], or 81.18 amu.
• This value is close to the atomic mass of
bromine, which is 79.904 amu.
Searching for an
Organizing Principle](https://image.slidesharecdn.com/chem12c06l1lo-211020192056/85/Chem12-c06-l1_lo-6-320.jpg)