Chem
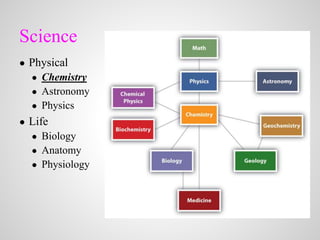
- 1. Science ● Physical ● Chemistry ● Astronomy ● Physics ● Life ● Biology ● Anatomy ● Physiology
- 2. Chemistry ● Chemistry studies the structure and properties of matter and the changes it undergoes. ● Chemistry focuses on chemical rather than physical change.
- 4. Change ● Physical Change ● a usually reversible change, chemical properties do not change ● H20 H20 H2O ● Chemical Change ● A reaction involving the rearrangement of the atoms of one or more substances ● There is a formation of at least one new substance
- 5. Matter scientific word for “stuff”
- 6. Classifying Matter Substances ● Pure ● Element ● A substance is an element if you can find it on the periodic table ● Compound ● A substance is a compound if it has a chemical formula ● Not Pure ● Homogeneous Mixture ● A homogeneous mixture is uniform, also called a solution ● Heterogeneous Mixture ● A heterogeneous mixture is not uniform
- 7. Substances ● Pure Substances ● Elements ● K Na Ne Ag ● Compounds ● O2 NaCl H20 Fe2O3 ● Substances ● Homogeneous Mixture ● Water with salt ● Tylenol ● Apple Juice ● Heterogeneous Mixture ● Pozole ● Lago de Chapala ● Pizza
- 8. Substances States of Matter Solid Liquid Gas Plasma
- 12. Solids ● Definite shape & volume ● Do not take the shape of a container ● Incompressible ● *Do not confuse with rigidity ● Expands with heat
- 13. (Chemical Bonds Influence Rigidity)
- 14. Liquids ● Constant volume ● Flow ● Take shape of container ● Incompressible ● Expand with heat
- 15. Gases ● Do not have constant volume ● Compressible ● Do not have a definite shape ● Move freely ● Fill the space of a container 482 m/s km/hr??
- 16. (Gas = Vapor) 25o C 120oC A substance is a gas if it is gaseous at room temperature (25oC).
- 17. Physical Properties of Matter ● Physical Property ● a characteristic that can be observed or measured without changing the objects composition. ● Density ● color ● odor ● hardness ● melting point and boiling point ● Extensive (Exterior) ● Depend on the amount present ● Intensive (Interior) ● Do not depend on the amount present
- 18. Extensive How much does the copper wire/plastic weigh? Intensive Does the copper wire/plastic conduct electricity?
- 19. Chemical Properties of Matter Chemical Property ● Ability to: ● Combine with another substance ● Change into another substance ● Examples ● Flammability ● Gasoline vs Water ● Toxicity ● Chlorine vs Water ● Ability to oxidize ● Iron vs Aluminum
- 22. Changes in Matter ● Physical Change ● Phase Change - Requires changes in pressure and/or temperature ● A transition from one state of matter to another ● boil, freeze, condense, vaporize, or melt Physical Changes depend on Physical Properties or Chemical Properties? At what temperature does copper change state from a solid to a liquid? What type of property determines this change?
- 23. Changes in Matter ● Chemical Change ● sdfjkl **complete the slide
- 24. Changes in Matter The Law of Conservation of Mass Physical Change Chemical Change **write your understanding here
- 26. Elements
- 27. Atoms & Periodic Table
- 28. Vocabulary ● Atom ● Proton ● Neutron ● Electron ● Isotope ● Atomic Number ● Atomic Mass ● Period ● Group ● Family ● Valence Electrons ● Electron-dot Structure
- 31. http://home.web.cern.ch/about/how-accelerator- works http://wordlesstech.com/2012/05/03/hi ggs-boson-explained/
- 34. Periodic Table ● Organized according to: ● Atomic Number ● The number of protons in an atom ● Electron Structure ●Divided into: ●Periods ●Groups
- 35. Periods = Rows 1 2 3 4 5 6 7
- 36. Groups = Columns 8 ****Group number = number of Valence Electrons
- 37. Valence Electrons ● The number of electrons in the outermost electron orbital ☞ **Determines behaviour
- 38. Valence Electrons Electron-dot Structure / Lewis-dot
- 40. Element Atomic Number Protons Neutrons Electrons Period Group Valence Electrons Family Ba 33 2 7 87 6 3
- 41. All elements want to have a complete set of valence electrons; elements will react until they have a complete set. A complete set consists of eight electrons, this explains why Noble gases do not react. Helium is a Noble gas, even though its outer orbital only has two electrons. Helium’s outer orbital only accepts two electrons, therefore its set of valence electrons is complete; it will not react.
- 43. Alkali Metals!
- 44. Reactivity The attraction that a bonding pair of electrons feels for a particular nucleus. Depends on: ● the number of protons in the nucleus; ● the distance from the nucleus; ● the amount of screening by inner electrons.
- 46. At B
- 47. 1 2 3 7 8 Transitional Metals Groups 4, 5, 6 Row 3, 4, 5 Cs Ba Row 7 At Rn Is Cs more reactive than Li? Why?
