1. The document discusses the electron theory of solids, which aims to explain the structures and properties of solids through their electronic structure.
2. It describes three main stages in the development of the electron theory of solids: the classical free electron theory developed in 1900, the quantum free electron theory developed in 1928, and Bloch's zone theory also developed in 1928, known as the band theory of solids.
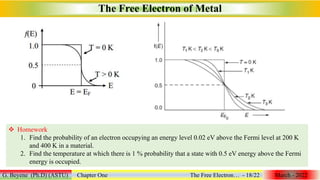
3. The quantum free electron theory overcomes many drawbacks of the classical theory by applying quantum mechanics and using Fermi-Dirac statistics instead of Maxwell-Boltzmann statistics.