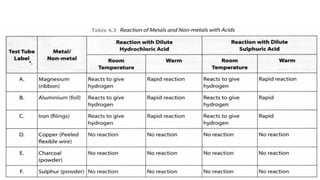
The document discusses the physical and chemical properties of metals and non-metals. Physically, metals are typically solid, malleable, ductile, and good conductors of heat and electricity, while non-metals can be solid, liquid, or gas and are not malleable, ductile, or good conductors. Chemically, metals react with oxygen to form metal oxides, react with water or acids, and can corrode in air/moisture. Non-metals typically burn when reacting with oxygen to form non-metal oxides. The reactivity series lists metals in order of their reactivity from most (potassium) to least (gold, platinum) reactive.













![Reaction with oxygen :-
Copper articles exposed to moist air for long, they develop a dull greenish coating.
The green material is a mixture of copper hydroxide {Cu(OH)2} and copper
carbonate[ CuCO3]
Silver objects tarnish with time and get a black coating of silver sulphide when they
react with sulphur containing substances in the air.
Gold and Platinum do not react with oxygen in the air.
2 Cu+H2O+CO2+O2-------> Cu[OH]2+CuCO3](https://image.slidesharecdn.com/ch-4materialsmetalsandnonmetals-220715085720-0cef9e9a/85/CH-4-MATERIALS_-METALS-AND-NON-METALS-pdf-14-320.jpg)