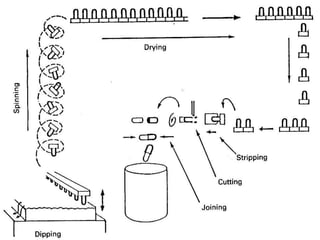
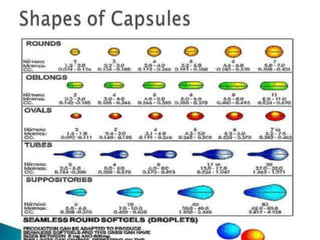
The document provides an overview of capsules as solid dosage forms, detailing hard-shelled and soft-shelled capsules, their composition, manufacturing processes, and filling methods. It outlines key aspects such as storage conditions, characteristics, and the machinery used in capsule production, including descriptions of various companies that manufacture capsule filling machines. Additionally, it covers the formulation of gelatin and soft gelatin capsules, including the ingredients and processes involved in their production.