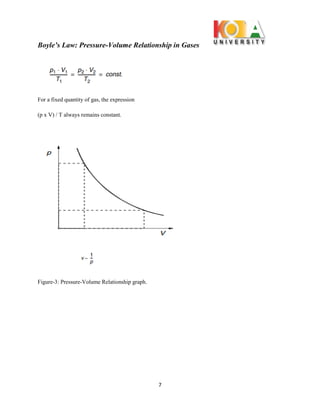
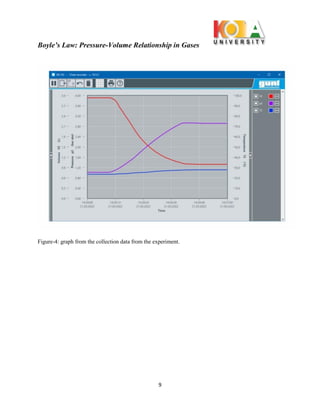
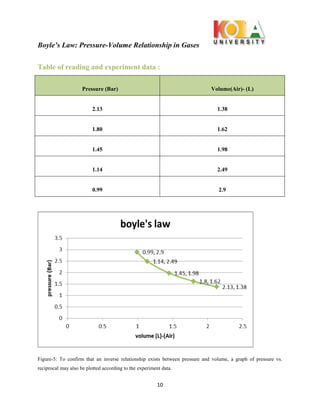
The document outlines an experiment conducted to demonstrate Boyle’s Law, which describes the inverse relationship between the pressure and volume of a confined gas, specifically air, at constant temperature. Through the use of the WL 102 experimental setup, data on pressure and volume changes are collected during isothermic expansions, supporting the theoretical premise that PV = constant. The findings affirm the significance of Boyle's Law in understanding gas behavior and its practical applications.









![Boyle’s Law: Pressure-Volume Relationship in Gases
13
References :
1. Wengert, H., 1969. A Different Approach to the Study of Pressure-Volume Relationship of
Gases. Iowa Science Teachers Journal, 6(4), pp.19-22. [accessed : 27/oct/2022]
2. Levitt, L.S., 1954. Extreme Pressures. I. A New Pressure–Volume Relationship. The Journal
of Physical Chemistry, 58(7), pp.573-576. [accessed : 27/oct/2022]
3. Potter, E., 2001. Gender and Boyle's law of gases. Indiana University Press.
4. Learn, W.Y.L., 2009. Gases. [accessed : 27/oct/2022]
5. Harris, D.J., O'Boyle, M., Bates, E. and Buckley, C., 2014. Harris, O'Boyle & Warbrick: Law
of the European convention on human rights. Oxford University Press, USA. [accessed :
27/oct/2022]
6. McGregor, D., Sweeney, W.V. and Mills, P., 2012. A Simple Mercury-Free Laboratory
Apparatus To Study the Relationship between Pressure, Volume, and Temperature in a
Gas. Journal of Chemical Education, 89(4), pp.509-512. [accessed :27/oct/2022]
7. Laugier, A. and Garai, J., 2007. Derivation of the ideal gas law. Journal of Chemical
Education, 84(11), p.1832. . [accessed :27/oct/2022]
8. Kenny, B.J. and Ponichtera, K., 2021. Physiology, Boyle's Law. In StatPearls [Internet].
StatPearls Publishing. [accessed :27/oct/2022]](https://image.slidesharecdn.com/00dima-lab-report-boyleslaw00-230417073955-52d02910/85/boyle-s-law-13-320.jpg)