The document discusses the history and challenges of clinical trial blinding, highlighting various definitions and interpretations of blinding terminology and the importance of proper implementation in randomized controlled trials. It reviews past studies, critiques the effectiveness of blinding, and outlines the evolving standards and ethical considerations in clinical trial designs, especially in the context of recent public health emergencies like HIV and Ebola. Additionally, it touches upon the 'Right to Try' laws that allow terminally ill patients to access experimental treatments.


![Blinding: Do we know it when we see it?
• Some suggest “masking” rather than blinding. [1]
• Some suggest blinding be specifically explained
in the study report. [1]
KF Schulz, I Chalmers, DG Altman. The landscape and lexicon of blinding in randomized trials. Annals of Internal Medicine.
2002;136:254-9](https://image.slidesharecdn.com/blindinghistorysocrartp01jun2015-150610113718-lva1-app6891/85/Blinding-History-and-Current-Issues-3-320.jpg)

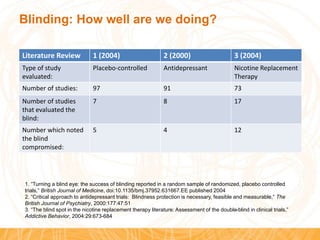
![Blinding: How well are we doing?
• Of 200 randomly chosen studies from 2001, 19%
stated they were “double blind” but had not
blinded either patients, health care providers, or
data collectors. [1]
M Haahr, A Hróbjartsson. Who is blinded in randomized clinical trials? A study of 200 trials and a survey of authors. Clin Trials.
2006;3:360-5](https://image.slidesharecdn.com/blindinghistorysocrartp01jun2015-150610113718-lva1-app6891/85/Blinding-History-and-Current-Issues-5-320.jpg)










![1920’s English Speaking
• Michigan Tuberculosis Trial (1926-1931) [1]
Single blind
Control for instability of disease state
• Cardiac Department of London Hospital Angina Study,
1930 [1]
Control for variations of symptoms
• Claude Bernard’s paper “An Introduction to the Study of
Experimental Medicine” translated and published in New
York, 1927 [2]
Used the term “comparative method” and described blinding
1. TJ Kaptchuk, "Intentional Ignorance: A History of Blind Assessment and Placebo Controls in Medicine", Bulletin of the
History of Medicine. 1998;72.3:389-433
2. Shapero AK, Shapero E. The powerful placebo: From Ancient Priest to Modern Physician. Baltimore: The Johns Hopkins
University Press. 1997](https://image.slidesharecdn.com/blindinghistorysocrartp01jun2015-150610113718-lva1-app6891/85/Blinding-History-and-Current-Issues-17-320.jpg)
![Paul Martini, Methodenlehre der
therapeutischen Untersuchung
• 1932
• “exclude suggestive or
other irrelevant factors in
the unwissentliche
Versuchsanordnung”
• Literally, “unwitting test” or
“test without knowing”
TJ Kaptchuk, "Intentional Ignorance: A History of Blind Assessment and Placebo Controls in Medicine", Bulletin of the History
of Medicine. 1998;72.3:389-433
Stoll S, Raspe H. Paul Martini and the development of clinical epidemiology in Germany. The Paul Martini Foundation. [poster]](https://image.slidesharecdn.com/blindinghistorysocrartp01jun2015-150610113718-lva1-app6891/85/Blinding-History-and-Current-Issues-18-320.jpg)


![Streptomycin Clinical Trial
• 1948
• Austin Bradford Hill
• “deliberately left out the words
‘randomization’ and ‘random
sampling numbers’ at the time
because… I might have scared
them [collaborating physicians]
off.”
TJ Kaptchuk, "Intentional Ignorance: A History of Blind Assessment and Placebo Controls in Medicine", Bulletin of the History
of Medicine. 1998;72.3:389-433](https://image.slidesharecdn.com/blindinghistorysocrartp01jun2015-150610113718-lva1-app6891/85/Blinding-History-and-Current-Issues-21-320.jpg)



![Some other thinking on blinding:
• Kolahi J. Abrishami M. Multiple-blind: Towards a new blinding protocol for
future generations of clinical trials. Med Hypotheses. 73(2009):843-5
Suggest expanding blinding, including requiring blinded creation of dual study reports and
blinding evaluation of study reports. (Some of these techniques were implemented by
Gold, but did not catch on for general use. [1])
• Bang H et al. Assessment of blinding in clinical trials. Controlled Clinical
Trials 2004;(25):143-56
Provides a method for arriving at a blinding index.
• JR Rees, TJ Wade, DA Levy, JM Colford Jr., JF Hilton. Changes in beliefs
identify unblinding in randomized controlled trials: a method to meet
CONSORT guidelines. Contemporary Clinical Trials. 2005;26:25-37
Suggest adding patient preference arms to studies.
• -D’Rozario MKD, Unblinded monitoring programs: design and education.
SoCRA Source. February 2011: 70-75
Discusses the need for unblinded monitoring and how to implement an unblinded
monitoring program.
1. Shapero AK, Shapero E. The powerful placebo: From Ancient Priest to Modern Physician.
Baltimore: The Johns Hopkins University Press. 1997](https://image.slidesharecdn.com/blindinghistorysocrartp01jun2015-150610113718-lva1-app6891/85/Blinding-History-and-Current-Issues-25-320.jpg)






