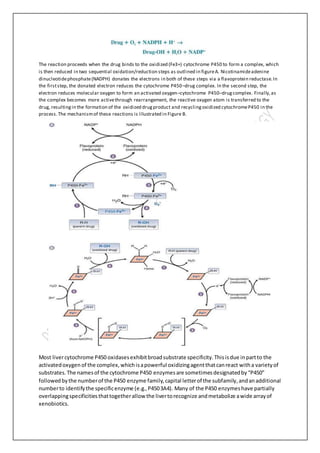
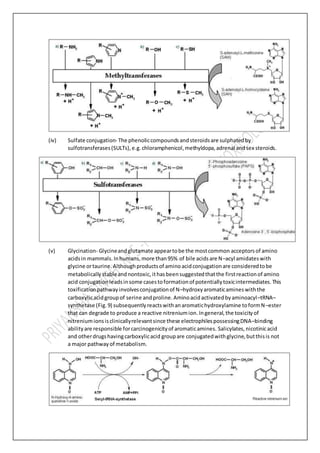
Biotransformation involves chemical alteration of drugs in the body through two phases. Phase I makes the drug more polar through reactions like oxidation, reduction and hydrolysis. This is mainly done by cytochrome P450 enzymes in the liver. Phase II further increases polarity through conjugation reactions like glucuronidation and sulfation. This makes the drug water-soluble and ready for excretion to remove it from the body. Biotransformation is an important mechanism for maintaining homeostasis after exposure to various xenobiotics.