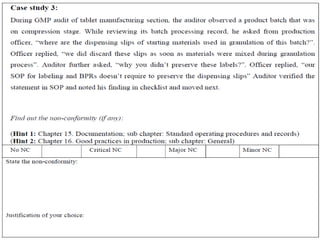
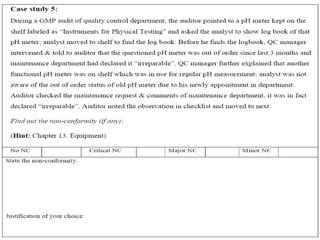
Here is a sample audit plan:
Day 1:
09:00-10:00: Opening meeting
10:00-12:00: Quality management system documentation review
13:00-15:00: Production area audit (warehousing, batching, mixing)
15:00-16:00: Non-conformance review meeting
Day 2:
09:00-11:00: Laboratory area audit (QC, QA labs)
11:00-13:00: Packaging and labeling area audit
13:00-14:00: Lunch
14:00-16:00: Regulatory affairs and change control audit
16:00-17:00