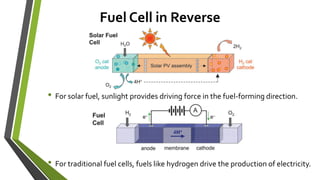
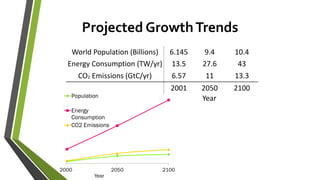
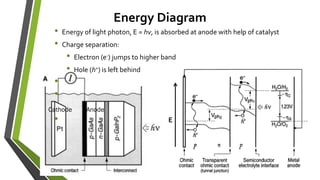
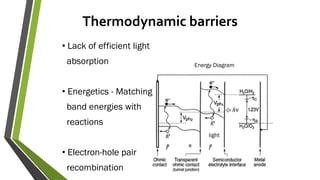
The document discusses the potential of solar fuels as a sustainable energy source that can address energy security and carbon emissions through artificial photosynthesis. It highlights key issues such as the cost of catalysts, thermodynamic barriers, and the need for scalable technology. Current research from notable scientists aims to improve efficiency and affordability to make solar fuels viable for widespread use.