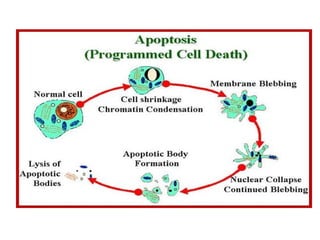
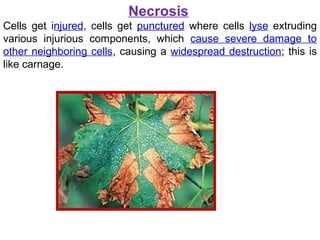
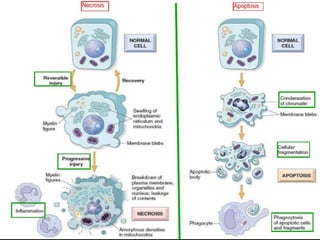
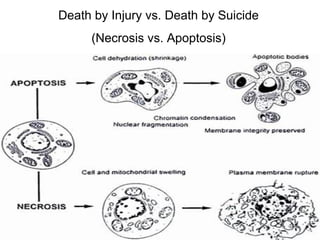
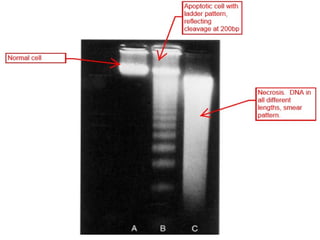
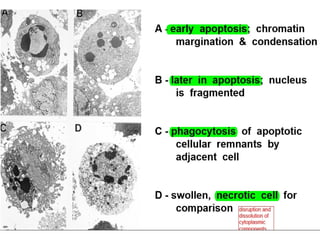
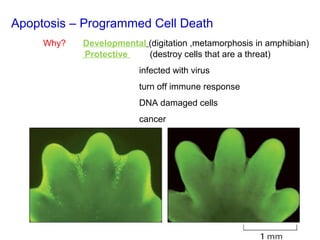
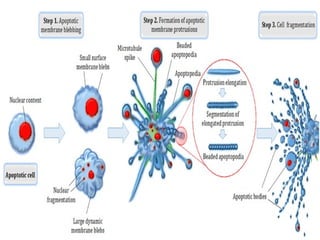
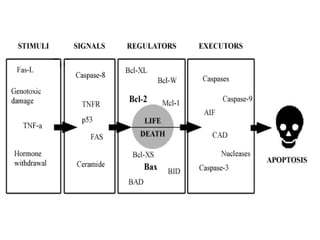
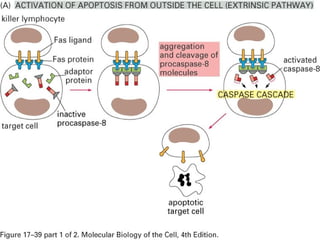
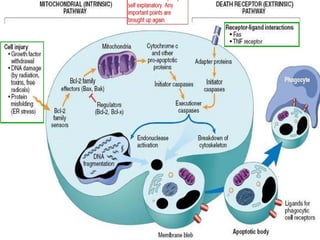
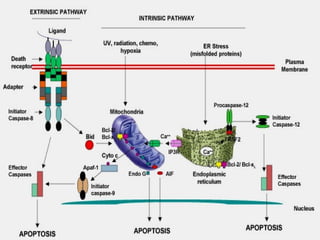
The document discusses various types of programmed cell death (PCD), including apoptosis, autophagy, paraptosis, autoschizis, oncosis, and necrosis. It provides details on the characteristics and mechanisms of apoptosis and autophagy. Apoptosis involves blebbing, cell shrinkage, nuclear fragmentation, and is mediated by caspases through the intrinsic and extrinsic pathways. Autophagy results in autophagosomic-lysosomal degradation of cytoplasmic contents and organelles. The document also discusses some plant-specific features of apoptosis and its role in pollen self-incompatibility.