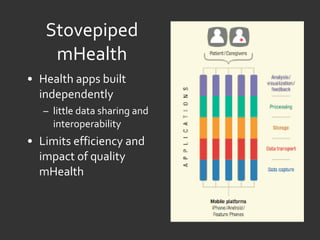
The document discusses evidence farming as an approach to evaluating mobile health (mHealth) applications and interventions. Evidence farming involves extracting evidence from care process data and manipulating care processes with flexible protocols. This allows generating hypotheses at the population level while maintaining some internal validity. The document proposes an open architecture called OpenmHealth to support evidence farming and crowdsourcing evidence through modules for usage analytics, randomized trials, individualized studies, and sharing findings. The goal is a learning community that can rapidly disseminate and iterate on evaluation methods that matter to improving mHealth.

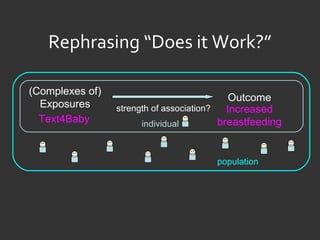
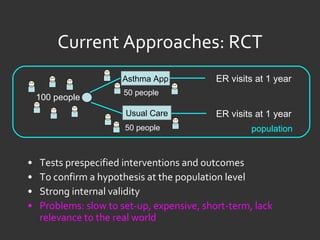
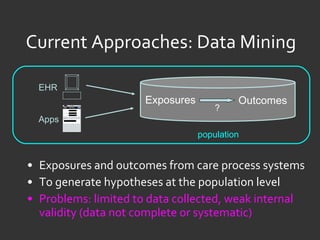
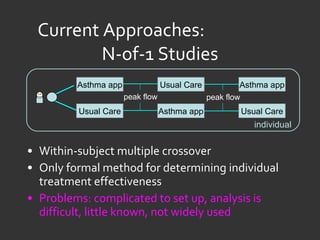

















![Ida Sim [email_address] Deborah Estrin [email_address] http://openmhealth.org/](https://image.slidesharecdn.com/11amsim-110505174810-phpapp01/85/11-am-sim-24-320.jpg)