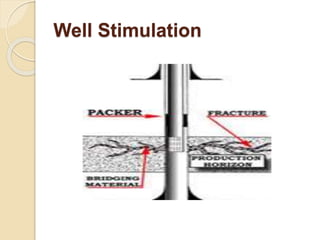
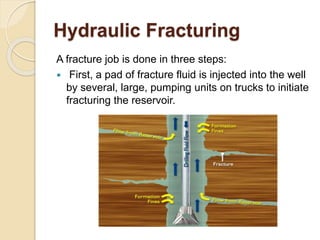
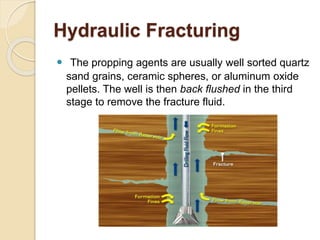
The document discusses well stimulation techniques in petroleum engineering, focusing on methods to enhance oil production from wells with low permeability, such as acidizing and hydraulic fracturing. It emphasizes the importance of various acid types and additives to mitigate common problems during acid treatments. Additionally, it details the processes and components involved in effectively executing fracture jobs to create conductive paths in reservoir rocks.