Tutorial - Unit conversions
•Download as PPTX, PDF•
0 likes•207 views
The slide show presents most common unit conversions that are necessary for success in high school Chemistry and Physics classes.
Report
Share
Report
Share
Recommended
More Related Content
Similar to Tutorial - Unit conversions
Similar to Tutorial - Unit conversions (20)
PPT-MST (2).pptxgsggggggggggggggggggggggggggggggggghhhs

PPT-MST (2).pptxgsggggggggggggggggggggggggggggggggghhhs
Units of measurements( standard system for weight, length and volume)

Units of measurements( standard system for weight, length and volume)
More from Tatiana Dyakina
More from Tatiana Dyakina (13)
Another Solution Will Present Itself - Qui-Gon Jinn

Another Solution Will Present Itself - Qui-Gon Jinn
End of the Unit Quiz (Physics) - Sig Figs and Unit Conversion

End of the Unit Quiz (Physics) - Sig Figs and Unit Conversion
Recently uploaded
Antibiotics are medicines that fight infections caused by bacteria in humans and animals by either killing the bacteria or making it difficult for the bacteria to grow and multiply. Bacteria are germsABHISHEK ANTIBIOTICS PPT MICROBIOLOGY // USES OF ANTIOBIOTICS TYPES OF ANTIB...

ABHISHEK ANTIBIOTICS PPT MICROBIOLOGY // USES OF ANTIOBIOTICS TYPES OF ANTIB...ABHISHEK SONI NIMT INSTITUTE OF MEDICAL AND PARAMEDCIAL SCIENCES , GOVT PG COLLEGE NOIDA
HIV (human immunodeficiency virus) is a virus that attacks cells that help the body fight infection, making a person more vulnerable to other infections and diseases. It is spread by contact with certain bodily fluids of a person with HIV, most commonly during unprotected sex (sex without a condom or HIV medicine to prevent or treat HIV), or through sharing injection drug equipment.
Seasonal influenza (the flu) is an acute respiratory infection caused by influenza viruses. It is common in all parts of the world. Most people recover without treatment.
Influenza spreads easily between people when they cough or sneeze. Vaccination is the best way to prevent the disease.
Symptoms of influenza include acute onset of fever, cough, sore throat, body aches and fatigue.
Treatment should aim to relieve symptoms. People with the flu should rest and drink plenty of liquids. Most people will recover on their own within a week. Medical care may be needed in severe cases and for people with risk factors.
There are 4 types of influenza viruses, types A, B, C and D. Influenza A and B viruses circulate and cause seasonal epidemics of disease.
Influenza A viruses are further classified into subtypes according to the combinations of the proteins on the surface of the virus. Currently circulating in humans are subtype A(H1N1) and A(H3N2) influenza viruses. The A(H1N1) is also written as A(H1N1)pdm09 as it caused the pandemic in 2009 and replaced the previous A(H1N1) virus which had circulated prior to 2009. Only influenza type A viruses are known to have caused pandemics.
Influenza B viruses are not classified into subtypes but can be broken down into lineages. Influenza type B viruses belong to either B/Yamagata or B/Victoria lineage.
Influenza C virus is detected less frequently and usually causes mild infections, thus does not present public health importance.
Influenza D viruses primarily affect cattle and are not known to infect or cause illness in people.HIV AND INFULENZA VIRUS PPT HIV PPT INFULENZA VIRUS PPT

HIV AND INFULENZA VIRUS PPT HIV PPT INFULENZA VIRUS PPTABHISHEK SONI NIMT INSTITUTE OF MEDICAL AND PARAMEDCIAL SCIENCES , GOVT PG COLLEGE NOIDA
Molecular and Cellular Mechanism of Action of Hormones such as Growth Hormone, Prolactin, InsulinMolecular and Cellular Mechanism of Action of Hormones such as Growth Hormone...

Molecular and Cellular Mechanism of Action of Hormones such as Growth Hormone...Ansari Aashif Raza Mohd Imtiyaz
Recently uploaded (20)
ANITINUTRITION FACTOR GYLCOSIDES SAPONINS CYANODENS

ANITINUTRITION FACTOR GYLCOSIDES SAPONINS CYANODENS
ABHISHEK ANTIBIOTICS PPT MICROBIOLOGY // USES OF ANTIOBIOTICS TYPES OF ANTIB...

ABHISHEK ANTIBIOTICS PPT MICROBIOLOGY // USES OF ANTIOBIOTICS TYPES OF ANTIB...
Harry Coumnas Thinks That Human Teleportation is Possible in Quantum Mechanic...

Harry Coumnas Thinks That Human Teleportation is Possible in Quantum Mechanic...
X-rays from a Central “Exhaust Vent” of the Galactic Center Chimney

X-rays from a Central “Exhaust Vent” of the Galactic Center Chimney
Vital Signs of Animals Presentation By Aftab Ahmed Rahimoon

Vital Signs of Animals Presentation By Aftab Ahmed Rahimoon
HIV AND INFULENZA VIRUS PPT HIV PPT INFULENZA VIRUS PPT

HIV AND INFULENZA VIRUS PPT HIV PPT INFULENZA VIRUS PPT
Molecular and Cellular Mechanism of Action of Hormones such as Growth Hormone...

Molecular and Cellular Mechanism of Action of Hormones such as Growth Hormone...
Fun for mover student's book- English book for teaching.pdf

Fun for mover student's book- English book for teaching.pdf
Mining Activity and Investment Opportunity in Myanmar.pptx

Mining Activity and Investment Opportunity in Myanmar.pptx
MSC IV_Forensic medicine - Mechanical injuries.pdf

MSC IV_Forensic medicine - Mechanical injuries.pdf
Factor Causing low production and physiology of mamary Gland

Factor Causing low production and physiology of mamary Gland
Tutorial - Unit conversions
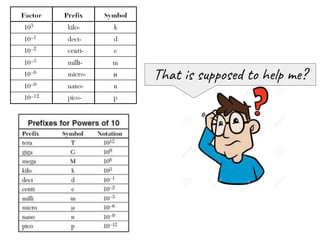
- 1. That is supposed to help me?
- 2. Let’s deal with prefix milli- , since we see it quite often in Chemistry. (unit of mass) gram g (unit of volume) liter L (unit of distance) meter m 1 gram contains 1000 milligrams → 1g = 1000mg 1 liter contains 1000 milliliters → 1L = 1000mL 1 meter contains 1000 millimeters → 1m = 1000mm
- 3. And 1 milliwhatever is one thousands part of whatever it is :) Too bad, something and whatever are not basic units of metric system. So, technically: 1 something contains 1000 millisomethings? 1 something = 1000 msomethings If I collect and put on a scale 1000 milligrams of something. What would be the reading on the scale? _____________1 gram
- 4. If I collect and pour together 1000 milliliters of liquid in one big measuring cylinder, what would be the overall volume of that liquid? _____________ My pet caterpillar crawls 1 millimeter every minute. It travels in a straight line. How far will it go if it will crawl for a 1000 minutes without rest? _____________ He will never catch up. Freedom!!! 1 Liter 1 meter
- 5. What if my pet caterpillar was crawling for 3 hours and then went to sleep? 3 hours → 60 x 3 = 180 min Speed : 1 mm/min ⇒ 180mm What is it in meters? Oh, I know, I know… I have to multiply it by a thousand or divide? Which one?!?
- 6. 1 m = 1000 mm
- 7. I have to multiply it by a thousand or divide? Which one?!? Any number can be multiplied by 1 without any change to it. You want to get rid of millimeters. So, make sure, they are in denominator in the conversion factor, so they would get canceled out.
- 8. You have 10875.56mg of gold. Are you rich? Convert it into grams... I know: 1g = 1000mg I want mg to cancel out… About 11g of gold. I guess, I’m not that rich.
- 9. I have 3.75 L of water. What that would be in mL? I know: 1L = 1000mL I want L to cancel out, and L in numerator. So, I must choose conversion factor with L in denominator!
- 10. Density of gold is 19.3 g/mL. What would be this density in g/L? I know: 1L = 1000mL I want mL to cancel out, and mL in denominator. So, I must choose conversion factor with mL in numerator!
- 11. Another unit of volume that Chem students get to deal with: cm3 Great! (deep sarcasm) What do I do with it? Nothing special: 1mL = 1 cm3 What’s the trick? The trick is not to overthink it. When converting mL into cm3 and back, don’t cube anything, don’t take cubic routes. Just change the unit - it is 1:1 ratio.
- 12. Convert 2.35 mL into cm3 2.35 cm3 Convert 28.77 cm3 into mL 28.77 mL Convert 7.23 L into cm3 7.23 L → 7230 mL 7230 ml → 7230 cm3 Convert 252 cm3 into L 252 cm3 → 252 mL 252 ml → 0,252 L
- 13. My average stride is 58 cm. What is it in meters? 1 gram contains 100 centigrams → 1g = 100cg 1 liter contains 100 centiliters → 1L = 100cL 1 meter contains 100 centimeters → 1m = 100cm
- 14. 1 m = 100 cm
- 15. I want cm to cancel out and cm in numerator, I must choose a conversion factor with cm in denominator . Wow! And what would that be in good old inches? Do you know conversion factor for meters and inches?
- 16. No. But I know that 1 inch is 2.54 cm. First, I want to get rid of meters… And now, let’s get rid of centimeters... I could have done it in one go...
- 17. Kilo- is another prefix that you frequently get to deal with in Chemistry and in Physics. 1000 grams make up 1 kilogram → 1000g = 1kg 1000 liters make up 1 kiloliter → 1000L = 1kL 1000 meters make up 1 kilometer → 1000m = 1km
- 18. Hedwig weighs 4.5lbs. How much that is in grams? (1 lb = 0.45kg) 1000g = 1kg
- 19. Dimensional analysis is especially important in Physics where you need to convert complex units… Car is moving at 60 mph (mph → miles/hour) What would it be in m/sec? (1 mile = 1.6km)
- 20. 1 mi = 1.6 km 1 km = 1000m
- 21. 1 hour = 60 min 1 min = 60 sec
- 22. Why not? Can we do all of it in one go?
- 23. My speed is 5 cm/min. Convert it into m/sec, km/hour and mph (mi/hour). 1 mi = 1.6 km