Production c hapter-two-engineering-materials
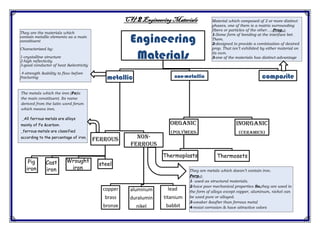
- 1. rialsEngineering Mate2CH. Engineering Materials metallic ferrous Pig iron Cast iron Wrought iron steel non- ferrous copper brass bronze aluminum duralumin nikel lead titanium babbit non-metallic Organic (polymers) Thermoplasts Thermosets inorganic (ceramics) composite They are the materials which contain metallic elements as a main constituent Characterized by: 1-crystalline structure 2-high reflectivity 3-good conductor of heat &electricity 4-strength &ability to flow before fracturing The metals which the iron (Fe)is the main constituent. Its name derived from the latin word ferum which means iron. _All ferrous metals are alloys mainly of Fe &carbon. _ferrous metals are classified according to the percentage of iron. Material which composed of 2 or more distinct phases, one of them is a matrix surrounding fibers or particles of the other….-Prop.:- 1-Some form of bonding at the interface bet. Them. 2-designed to provide a combination of desired prop. That isn’t exhibited by either material on its own. 3-one of the materials has distinct advantage examples:- concrete, car tires They are metals which doesn’t contain iron. Porp.:- 1- used as structural materials. 2-have poor mechanical properties So,they are used in the form of alloys except copper, aluminum, nickel can be used pure or alloyed. 3-weaker &softer than ferrous metal 4-resist corrosion & have attractive colors
- 2. Ferrous Materials SteelWrought ironCast ironPig IronP.O.C Fe+ up to 2%Carbon & several elements _Types of steel are classified according to their carbon content pig iron refined in the puddling furnace, Ferrous metal has the same constituents Of pig iron &produced by the further refinement of pig iron in cupola furnace. Ferrous metal obtained by smelting iron ore in the blast furnace with the aid of coal and fluxes. Definition _As the carbon content increase the strength &hardness increase but ductility decrease. have ferrite content about 99.9%contains 2-5% carbon 92-97% Fe +high percentage of carbon in addition to small amounts of Si, Mn, P,S percentage 1-Plain carbon steel:- a)mild steel:0.06- 0.25% Carbon _weak, soft, &ductile& has considerable strength &ductility,so, it can be easily cold or hot worked &machined b)Medium carbon steel: 0.35- 0.65% Carbon c)high carbon steel: 0.65- 1.5% carbon _low melting pt.& fluidity inc. in the liq. State so, it can be casted _Hard, strong, brittle &little ductility &malleability. _used in: cutting tools subjected to impact such as chisels, dies& wood working tools…also the tools subjected to high abrasive action like scribers, punches, files, vice jaws, cold chisels…… _used in: cheap rivets, pipes, wires, angels, car bodies, bolts, screws, nuts _used in: engineering components requiring high strength& moderate hardness like tool holders, axles, gears, &railways rails 2-Alloy steels:- plain carbon steels their mech. Prop. Improved by adding alloying elements..most of them are Cr 4-22%, Cu,Ni 0- 26%,Mn, molybdenum, silicon _HSS(high speed steel) has the ability to keep its hardness up to 500-600°C so they used in producing cutting tools working at moderate speeds by adding 18%tungsten, 4%Cr, 1% V to high carbon steels (0.7%) 1-soft& strong in tension 2-tough &resistant to atmospheric corrosion bec. It is covered by with a protective oxide layer. 3-can't be heated to a liquid State bec. It has v. low carbon content but it becomes pasty. 4-it isn't casted but at red heat it is very soft & welds readily by hammering making it an ideal metal for forge work. 5-easily cold worked by rolling, twisted &bending 1-presented as free graphite when cools slowly & has a grey appearance when it fractures. 2-acts as a lubricant during machining operations. 3-brittle, can't be forged 4-weak in tension but strong under compression 5-easy to machine 6-the amount of carbon gives great fluidity to the molten cast iron. 7-the resulting casting is extremely hard &brittle bec. the carbon no longer presents as free graphite but combines with the metal forming Fe carbide 8-white cast Fe: v. difficult to be machined it has white appearance when it fractures…it is used in rolls, balls in mills 1-brittle and week bec.it includes large amount of carbon &impurities. 2-never used as structural material but used only to produce other ferrous metals -Properties(Kinds) used for: making chains & crane hooks intricate shapes like tool machines, beds & engine bodies _it is the base of production of all ferrous metals. uses
- 3. Non-ferrous materials P.O.C Aluminum & its alloys Copper-brass alloys lead nickel titanium properties *according to the volume of production aluminum keeps the 1st place among the non- ferrous metals. -ProP.:-1- light (its weight is 1/3 the weight of steel) 2-soft & ductile Copper is next to Al in its use -Prop.:- 1- pure copper (99%) has high electrical & thermal conductivity. 1-It is the heaviest common metal 2-very soft 3-weak in tension 4-highly malleable Contains 97.5-99.5% Ni, 0.6-1% Co 1-high corrosion resistance. 2-ferromagnetic 1-High melting point(1725°C) 2-high strength 3-low specific weight 4-very good corrosion resistance. color -white colorBluish -Blue colorgray Produced in the form of thin sheets, wires, tubes or solid selections. Can be rolled into sheets or tubes but can't be drawn into wires Uses Its alloys used in: 1-means of transport 2-foodstuff industry (foils, kitchen utensils, food cotainers) 3-building engineering due to it corrosion resistance. 1-electrical conductors & cables. Constituent in making bronzes fusible alloys & soft solders &radiation shielding 1-surface protection of metals. 2-chemical & food stuff industry 3-steam seals, coins, resistance thermometers High speed aircraft components alloys Duralumin: one of the most important Al alloys..consists of:95% Al, 4% Cu, 0.5%Mg, 0.5% Mn.. Also it has the advantage of Al& steel strength so it is used in aircraft industry Brass: it consists of Cu & 39-45%Zn..used in cartridge cases, radiators& sealing rings. If copper is alloyed with other elements its often called bronze..some alloys are specified like tin bronze, phosphor bronze Monel alloy contains 30% Cu, has heat& corrosion resistance metals & good mechanical properties…used in making chemical apparatuses, medical instruments Can be alloyed with Cr, Al, Fe, Ni& other certain elements. These alloys applied as heat resistance materials in place of Al alloys& steels to reduce the weight of the construction as in rockets.
- 4. Organic non-metallic materials(polymers)(plastics) It is the material which composed mainly or wholly of carbon compounds P.O.C Thermoplastics (Thermo- softening) Thermosets (thermos- hardening) Inorganic non-metals (ceramics) definition Materials can be shaped & reshaped by the application of heat &pressure as long as the heat doesn't cause damage. Materials that undergo chemical change by subjecting to heat &pressure after which they can't be changed by further heat or pressure (can't be reshaped) Ceramics: Products made from inorganic materials which contain metallic or non-metallic elements properties 1-don’t melt but flow at a suitable temperature & pressure 2-suitable for injection moulding &extrusion 3-behave like glass when blown 4-can be shaped by pressure an vacuum technique 1-they gain rigidity before the applied pressure & heat is removed 1-hard, brittle 2-tend to be more resistant than other metals 3-plastics to high temperatures &to serve enviroments examples bottles Melamine, formaldehyde, bakelite MgO magnesia (ionically bonded), silicon carbide SiC (covalently bonded), floor tiles, bricks, electrical insulators, spark plugs, concrete, glass, clay, stone Mention the factors of selecting materials. 1-the ability to stand up to service conditions 2-the suitability for the process used to manufacture it 3-the material cost & availability
- 5. Classifications of properties (physical, chemical, thermal, electrical& magnetic, optical, Mechanical ) Mechanical property definition Elasticity The ability of material to restore its original shape or volume at once when the external force is removed Ductility The ability of the material to be drawn into wires by a tensile force… the ductile material must be strong & plastic Ex. Copper aluminum, mild steel, nickel, zinc, tin &lead Hardness It has a wide variety of meaning from them. The resistance to wear, penetration, scratching& machining, Also; it is the ability of the metal to cut another metals. It is very important in making cutting tools Strength The ability of the material to resist fracture or yielding when subjecting to external load Brittlness The opposite of ductility or it means that property of breaking the material with little permanent elongation or distortion ..it is also associated with hardness So, the brittle materials can't be forged Ex. Cast iron Toughness It is the opposite of brittles OR the ability of the material to resist fracture due to high impact loads like sudden shocks The toughness decreases by heating Ex. Hammer head Malleability Special case of ductility that permits material to be rolled or hammered into thin sheets or different shape without fracturing.. Heat may be used to make material more malleable There is little difference bet. Malleability & ductility for example Lead is malleable bec. It can be shaped by hammering but not ductile bec. It isn’t strong enough to be drawn into wires Fusibility The ability of a material to become easily liquid when heated Conductivity The ability of material to conduct heat or electricity