Carboxylic acid
•
21 likes•11,514 views
Dr James Chemistry SPM Form 5 Chapter 2 Carbon Compounds
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (20)
Viewers also liked
Viewers also liked (15)
Natural Rubber - Sources, Coagulation & Processing of Coagulate, Structure & ...

Natural Rubber - Sources, Coagulation & Processing of Coagulate, Structure & ...
Natural rubber, synthetic rubber and neoprene rubber

Natural rubber, synthetic rubber and neoprene rubber
Similar to Carboxylic acid
Similar to Carboxylic acid (20)
More from Wong Hsiung
More from Wong Hsiung (20)
Chapter 04 stereochemistry of alkanes and cycloalkanes

Chapter 04 stereochemistry of alkanes and cycloalkanes
Chapter 03 organic compounds alkanes and cycloalkanes

Chapter 03 organic compounds alkanes and cycloalkanes
Recently uploaded
Recently uploaded (20)
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
social pharmacy d-pharm 1st year by Pragati K. Mahajan

social pharmacy d-pharm 1st year by Pragati K. Mahajan
Interactive Powerpoint_How to Master effective communication

Interactive Powerpoint_How to Master effective communication
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...

Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...
Beyond the EU: DORA and NIS 2 Directive's Global Impact

Beyond the EU: DORA and NIS 2 Directive's Global Impact
Web & Social Media Analytics Previous Year Question Paper.pdf

Web & Social Media Analytics Previous Year Question Paper.pdf
Russian Escort Service in Delhi 11k Hotel Foreigner Russian Call Girls in Delhi

Russian Escort Service in Delhi 11k Hotel Foreigner Russian Call Girls in Delhi
Disha NEET Physics Guide for classes 11 and 12.pdf

Disha NEET Physics Guide for classes 11 and 12.pdf
Carboxylic acid
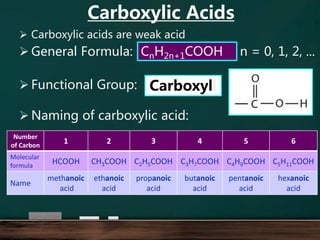
- 1. Carboxylic acids are weak acid General Formula: Functional Group: Naming of carboxylic acid: CnH2n+1COOH Carboxyll Number of Carbon 1 2 3 4 5 6 Molecular formula HCOOH CH3COOH C2H5COOH C3H7COOH C4H9COOH C5H11COOH Name methanoic acid ethanoic acid propanoic acid butanoic acid pentanoic acid hexanoic acid n = 0, 1, 2, ... Carboxylic Acids
- 2. Physical Properties of Carboxylic Acids Colour - Colourless liquid State - Larger molecules (C10 above) are wax-like solids Odour - sharp/ unpleasant smell Boiling point - High boiling point Solubility - Simple molecules are very soluble in water - Due to water molecule being strongly attracted to the – COOH group - Solubility ↓ when number of C ↑
- 3. Alcohols can be oxidised to carboxylic acids using potassium dichromate(VI) solution in the presence of dilute sulphuric acid. Chemical Equation below represent the reaction. C2H5OH+ 2[O] → CH3COOH + H2O Preparing Carboxylic Acids
- 4. Alcohols can be oxidised to produce carboxylic acid when react with oxidising agents
- 5. a. Acid properties CH3COOH is a weak monoprotic acid Only one hydrogen atom can ionize in water to produce H+ ion Partially dissociate in water Turn moist blue litmus → red React slowly with metals, bases and carbonates CH3COOH ↔ CH3COO- + H+ Ethanoic acid Ethanoate ion Chemical Properties Of Carboxylic Acids
- 6. b. Reaction with metals Carboxylic acid + metal → salt + H2 c. Reaction with carbonates Carboxylic acid + carbonates → salt + CO2 + H2O 2CH3COOH + Zn → Zn(CH3COO)2 + H2 Ethanoic acid Zinc ethanoate 2CH3COOH + CaCO3 → Ca(CH3COO)2 + CO2 + H2O Ethanoic acid Calcium ethanoate
- 7. d. Reaction with bases Carboxylic acid + bases → salt + H2O CH3COOH + NaOH → CH3COONa + H2O Ethanoic acid Sodium ethanoate 2CH3COOH + CuO → Cu(CH3COO)2 + H2O Ethanoic acid Copper (II) ethanoate
- 8. e. Reaction with alcohols (esterification) Carboxylic acid + alcohol → ester + H2O Catalyst: Concentrated H2SO4 CH3COOH + C4H9OH → CH3COOC4H9 + H2O Ethanoic acid Butan-1-ol Buthyl ethanoate
- 9. Ethanoic acid (acetic acid) • As a flavouring • As a preservative • Used with other chemicals to make drugs, dyes, paints, insecticides and plastics Methanoic acid (formic acid) • Used to coagulate latex Fatty acids (long-chain carboxylic acids) • Used in making soaps Benzoic acid • Preservative in food Uses of Carboxylic Acids
