Anti-eIF4A1 Antibody Validation
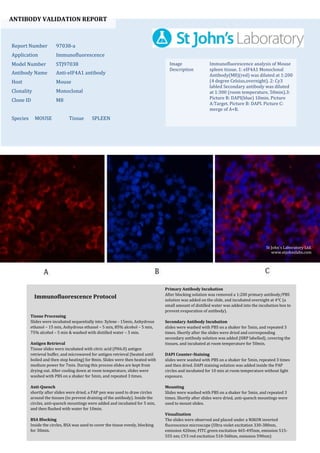
- 1. ANTIBODY VALIDATION REPORT Report Number 97038-a Application Immunofluorescence Model Number STJ97038 Antibody Name Anti-eIF4A1 antibody Host Mouse Clonality Monoclonal Clone ID M8 Species MOUSE Tissue SPLEEN Image Description Immunofluorescence analysis of Mouse spleen tissue. 1: eIF4A1 Monoclonal Antibody(M8)(red) was diluted at 1:200 (4 degree Celsius,overnight). 2: Cy3 labled Secondary antibody was diluted at 1:300 (room temperature, 50min).3: Picture B: DAPI(blue) 10min. Picture A:Target. Picture B: DAPI. Picture C: merge of A+B. Primary Antibody Incubation After blocking solution was removed a 1:200 primary antibody/PBS solution was added on the slide, and incubated overnight at 4°C (a small amount of distilled water was added into the incubation box to prevent evaporation of antibody). Secondary Antibody Incubation slides were washed with PBS on a shaker for 5min, and repeated 3 times. Shortly after the slides were dried and corresponding secondary antibody solution was added (HRP labelled), covering the tissues, and incubated at room temperature for 50min. DAPI Counter-Staining slides were washed with PBS on a shaker for 5min, repeated 3 times and then dried. DAPI staining solution was added inside the PAP circles and incubated for 10 min at room temperature without light exposure. Mounting Slides were washed with PBS on a shaker for 5min, and repeated 3 times. Shortly after slides were dried, anti-quench mountings were used to mount slides. Visualization The slides were observed and placed under a NIKON inverted fluorescence microscope (Ultra violet excitation 330-380nm, emission 420nm; FITC green excitation 465-495nm, emission 515- 555 nm; CY3 red excitation 510-560nm, emission 590nm) Immunofluorescence Protocol Tissue Processing Slides were incubated sequentially into: Xylene - 15min, Anhydrous ethanol – 15 min, Anhydrous ethanol – 5 min, 85% alcohol – 5 min, 75% alcohol – 5 min & washed with distilled water – 5 min. Antigen Retrieval Tissue slides were incubated with citric acid (PH6.0) antigen retrieval buffer, and microwaved for antigen retrieval (heated until boiled and then stop heating) for 8min. Slides were then heated with medium power for 7min. During this process slides are kept from drying out. After cooling down at room temperature, slides were washed with PBS on a shaker for 5min, and repeated 3 times. Anti-Quench shortly after slides were dried, a PAP pen was used to draw circles around the tissues (to prevent draining of the antibody). Inside the circles, anti-quench mountings were added and incubated for 5 min, and then flushed with water for 10min. BSA Blocking Inside the circles, BSA was used to cover the tissue evenly, blocking for 30min. St John's Laboratory Ltd. www.stjohnslabs.com
- 2. ANTIBODY VALIDATION REPORT Report Number 97038-b Application Immunofluorescence Model Number STJ97038 Antibody Name Anti-eIF4A1 antibody Host Mouse Clonality Monoclonal Clone ID M8 Species MOUSE Tissue TESTIS Image Description Immunofluorescence analysis of Mouse testis tissue. 1: eIF4A1 Monoclonal Antibody(M8)(red) was diluted at 1:200 (4 degree Celsius,overnight). 2: Cy3 labled Secondary antibody was diluted at 1:300 (room temperature, 50min).3: Picture B: DAPI(blue) 10min. Picture A:Target. Picture B: DAPI. Picture C: merge of A+B. Primary Antibody Incubation After blocking solution was removed a 1:200 primary antibody/PBS solution was added on the slide, and incubated overnight at 4°C (a small amount of distilled water was added into the incubation box to prevent evaporation of antibody). Secondary Antibody Incubation slides were washed with PBS on a shaker for 5min, and repeated 3 times. Shortly after the slides were dried and corresponding secondary antibody solution was added (HRP labelled), covering the tissues, and incubated at room temperature for 50min. DAPI Counter-Staining slides were washed with PBS on a shaker for 5min, repeated 3 times and then dried. DAPI staining solution was added inside the PAP circles and incubated for 10 min at room temperature without light exposure. Mounting Slides were washed with PBS on a shaker for 5min, and repeated 3 times. Shortly after slides were dried, anti-quench mountings were used to mount slides. Visualization The slides were observed and placed under a NIKON inverted fluorescence microscope (Ultra violet excitation 330-380nm, emission 420nm; FITC green excitation 465-495nm, emission 515- 555 nm; CY3 red excitation 510-560nm, emission 590nm) Immunofluorescence Protocol Tissue Processing Slides were incubated sequentially into: Xylene - 15min, Anhydrous ethanol – 15 min, Anhydrous ethanol – 5 min, 85% alcohol – 5 min, 75% alcohol – 5 min & washed with distilled water – 5 min. Antigen Retrieval Tissue slides were incubated with citric acid (PH6.0) antigen retrieval buffer, and microwaved for antigen retrieval (heated until boiled and then stop heating) for 8min. Slides were then heated with medium power for 7min. During this process slides are kept from drying out. After cooling down at room temperature, slides were washed with PBS on a shaker for 5min, and repeated 3 times. Anti-Quench shortly after slides were dried, a PAP pen was used to draw circles around the tissues (to prevent draining of the antibody). Inside the circles, anti-quench mountings were added and incubated for 5 min, and then flushed with water for 10min. BSA Blocking Inside the circles, BSA was used to cover the tissue evenly, blocking for 30min. St John's Laboratory Ltd. www.stjohnslabs.com
- 3. ANTIBODY VALIDATION REPORT Report Number 97038-c Application Immunofluorescence Model Number STJ97038 Antibody Name Anti-eIF4A1 antibody Host Mouse Clonality Monoclonal Clone ID M8 Species MOUSE Tissue TESTIS Image Description Immunofluorescence analysis of Mouse testis tissue. 1: eIF4A1 Monoclonal Antibody(M8)(red) was diluted at 1:200 (4 degree Celsius,overnight). 2: Cy3 labled Secondary antibody was diluted at 1:300 (room temperature, 50min).3: Picture B: DAPI(blue) 10min. Picture A:Target. Picture B: DAPI. Picture C: merge of A+B. Primary Antibody Incubation After blocking solution was removed a 1:200 primary antibody/PBS solution was added on the slide, and incubated overnight at 4°C (a small amount of distilled water was added into the incubation box to prevent evaporation of antibody). Secondary Antibody Incubation slides were washed with PBS on a shaker for 5min, and repeated 3 times. Shortly after the slides were dried and corresponding secondary antibody solution was added (HRP labelled), covering the tissues, and incubated at room temperature for 50min. DAPI Counter-Staining slides were washed with PBS on a shaker for 5min, repeated 3 times and then dried. DAPI staining solution was added inside the PAP circles and incubated for 10 min at room temperature without light exposure. Mounting Slides were washed with PBS on a shaker for 5min, and repeated 3 times. Shortly after slides were dried, anti-quench mountings were used to mount slides. Visualization The slides were observed and placed under a NIKON inverted fluorescence microscope (Ultra violet excitation 330-380nm, emission 420nm; FITC green excitation 465-495nm, emission 515- 555 nm; CY3 red excitation 510-560nm, emission 590nm) Immunofluorescence Protocol Tissue Processing Slides were incubated sequentially into: Xylene - 15min, Anhydrous ethanol – 15 min, Anhydrous ethanol – 5 min, 85% alcohol – 5 min, 75% alcohol – 5 min & washed with distilled water – 5 min. Antigen Retrieval Tissue slides were incubated with citric acid (PH6.0) antigen retrieval buffer, and microwaved for antigen retrieval (heated until boiled and then stop heating) for 8min. Slides were then heated with medium power for 7min. During this process slides are kept from drying out. After cooling down at room temperature, slides were washed with PBS on a shaker for 5min, and repeated 3 times. Anti-Quench shortly after slides were dried, a PAP pen was used to draw circles around the tissues (to prevent draining of the antibody). Inside the circles, anti-quench mountings were added and incubated for 5 min, and then flushed with water for 10min. BSA Blocking Inside the circles, BSA was used to cover the tissue evenly, blocking for 30min. St John's Laboratory Ltd. www.stjohnslabs.com
- 4. ANTIBODY VALIDATION REPORT Report Number 97038-d Application Immunofluorescence Model Number STJ97038 Antibody Name Anti-eIF4A1 antibody Host Mouse Clonality Monoclonal Clone ID M8 Species MOUSE Tissue TESTIS Image Description Immunofluorescence analysis of Mouse testis tissue. 1: eIF4A1 Monoclonal Antibody(M8)(red) was diluted at 1:200 (4 degree Celsius,overnight). 2: Cy3 labled Secondary antibody was diluted at 1:300 (room temperature, 50min).3: Picture B: DAPI(blue) 10min. Picture A:Target. Picture B: DAPI. Picture C: merge of A+B. Primary Antibody Incubation After blocking solution was removed a 1:200 primary antibody/PBS solution was added on the slide, and incubated overnight at 4°C (a small amount of distilled water was added into the incubation box to prevent evaporation of antibody). Secondary Antibody Incubation slides were washed with PBS on a shaker for 5min, and repeated 3 times. Shortly after the slides were dried and corresponding secondary antibody solution was added (HRP labelled), covering the tissues, and incubated at room temperature for 50min. DAPI Counter-Staining slides were washed with PBS on a shaker for 5min, repeated 3 times and then dried. DAPI staining solution was added inside the PAP circles and incubated for 10 min at room temperature without light exposure. Mounting Slides were washed with PBS on a shaker for 5min, and repeated 3 times. Shortly after slides were dried, anti-quench mountings were used to mount slides. Visualization The slides were observed and placed under a NIKON inverted fluorescence microscope (Ultra violet excitation 330-380nm, emission 420nm; FITC green excitation 465-495nm, emission 515- 555 nm; CY3 red excitation 510-560nm, emission 590nm) Immunofluorescence Protocol Tissue Processing Slides were incubated sequentially into: Xylene - 15min, Anhydrous ethanol – 15 min, Anhydrous ethanol – 5 min, 85% alcohol – 5 min, 75% alcohol – 5 min & washed with distilled water – 5 min. Antigen Retrieval Tissue slides were incubated with citric acid (PH6.0) antigen retrieval buffer, and microwaved for antigen retrieval (heated until boiled and then stop heating) for 8min. Slides were then heated with medium power for 7min. During this process slides are kept from drying out. After cooling down at room temperature, slides were washed with PBS on a shaker for 5min, and repeated 3 times. Anti-Quench shortly after slides were dried, a PAP pen was used to draw circles around the tissues (to prevent draining of the antibody). Inside the circles, anti-quench mountings were added and incubated for 5 min, and then flushed with water for 10min. BSA Blocking Inside the circles, BSA was used to cover the tissue evenly, blocking for 30min. St John's Laboratory Ltd. www.stjohnslabs.com
