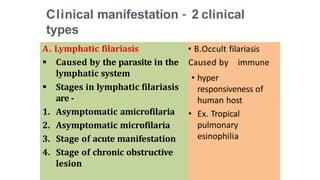
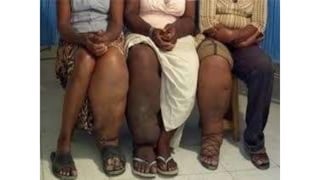
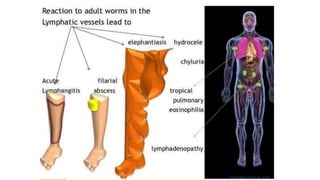
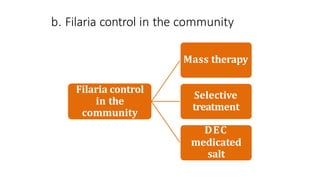
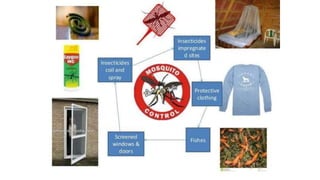
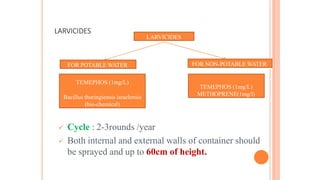
- The document discusses vector borne diseases like malaria and dengue.
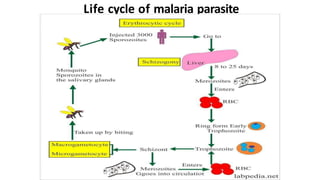
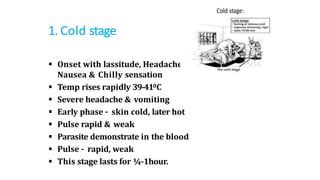
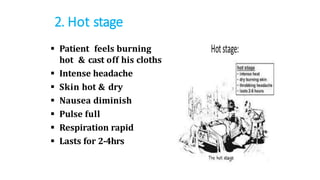
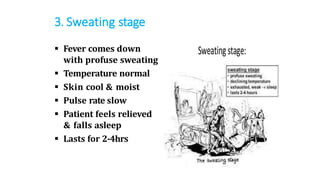
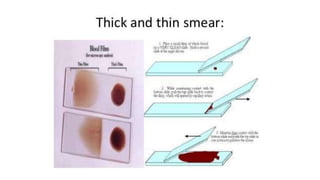
- Malaria is caused by a protozoan parasite Plasmodium and transmitted via the bites of infected Anopheles mosquitoes. It has an incubation period of 12-14 days and symptoms include cold, hot and sweating stages with fever.
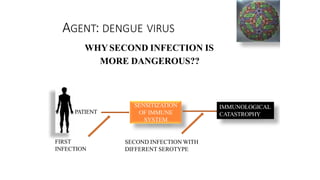
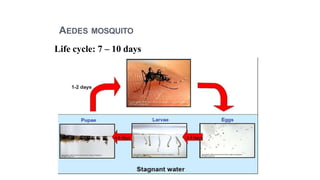
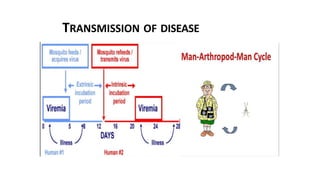
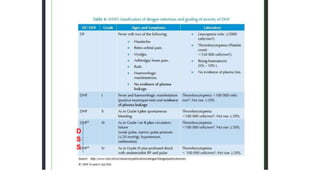
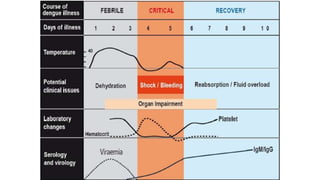
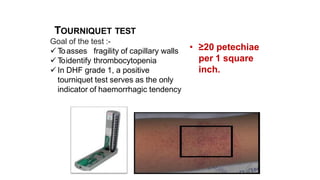
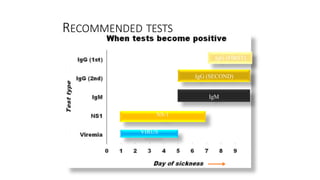
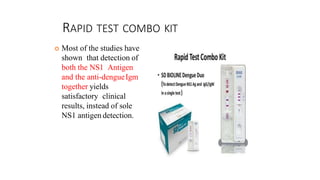
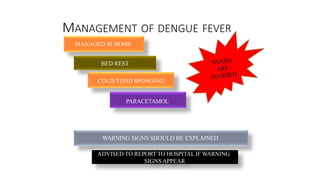
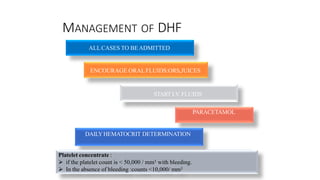
- Dengue is a viral illness transmitted by Aedes aegypti mosquitoes. It has an incubation period of 3-10 days and symptoms range from flu-like illness to potentially lethal dengue hemorrhagic fever.