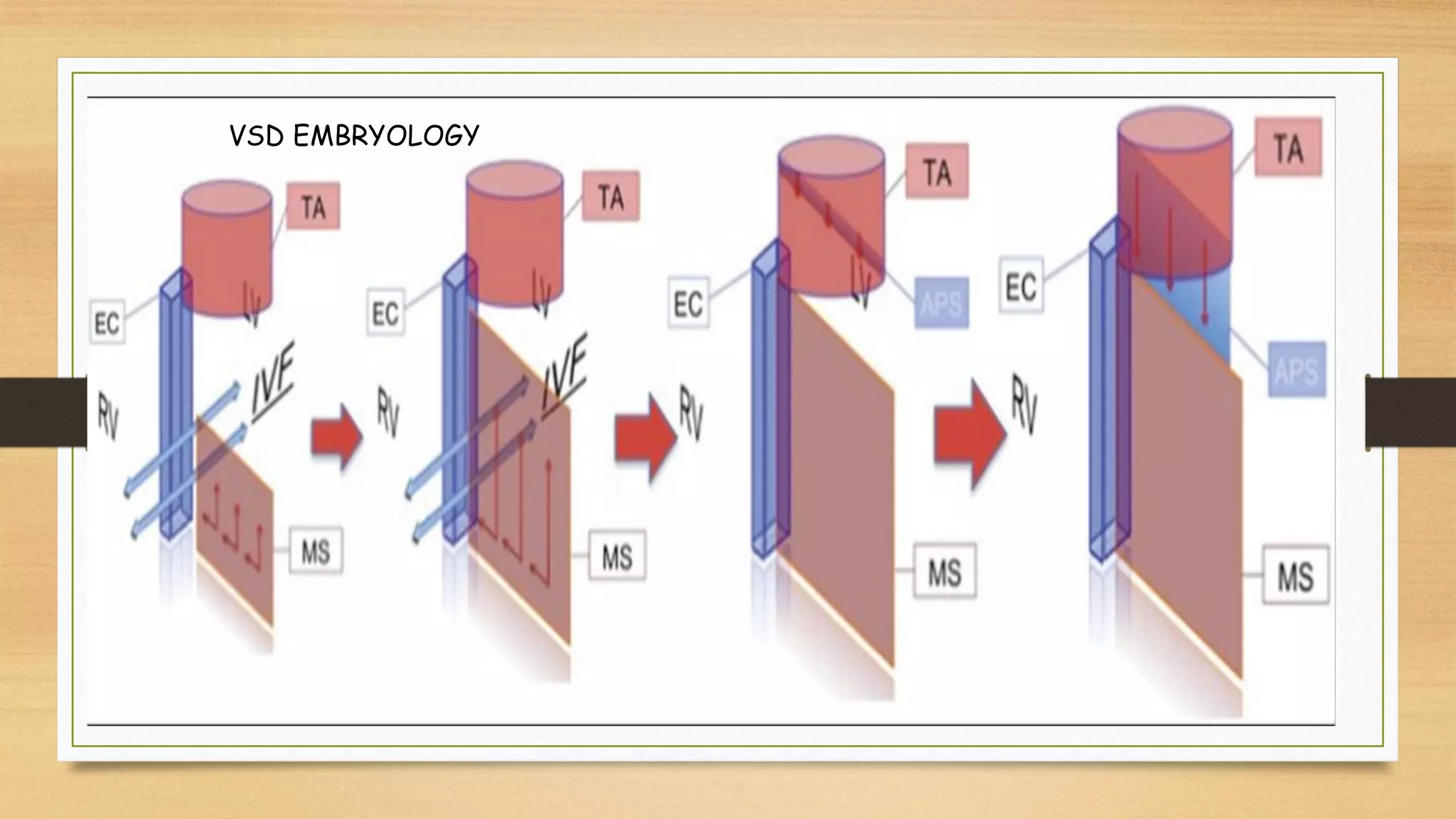
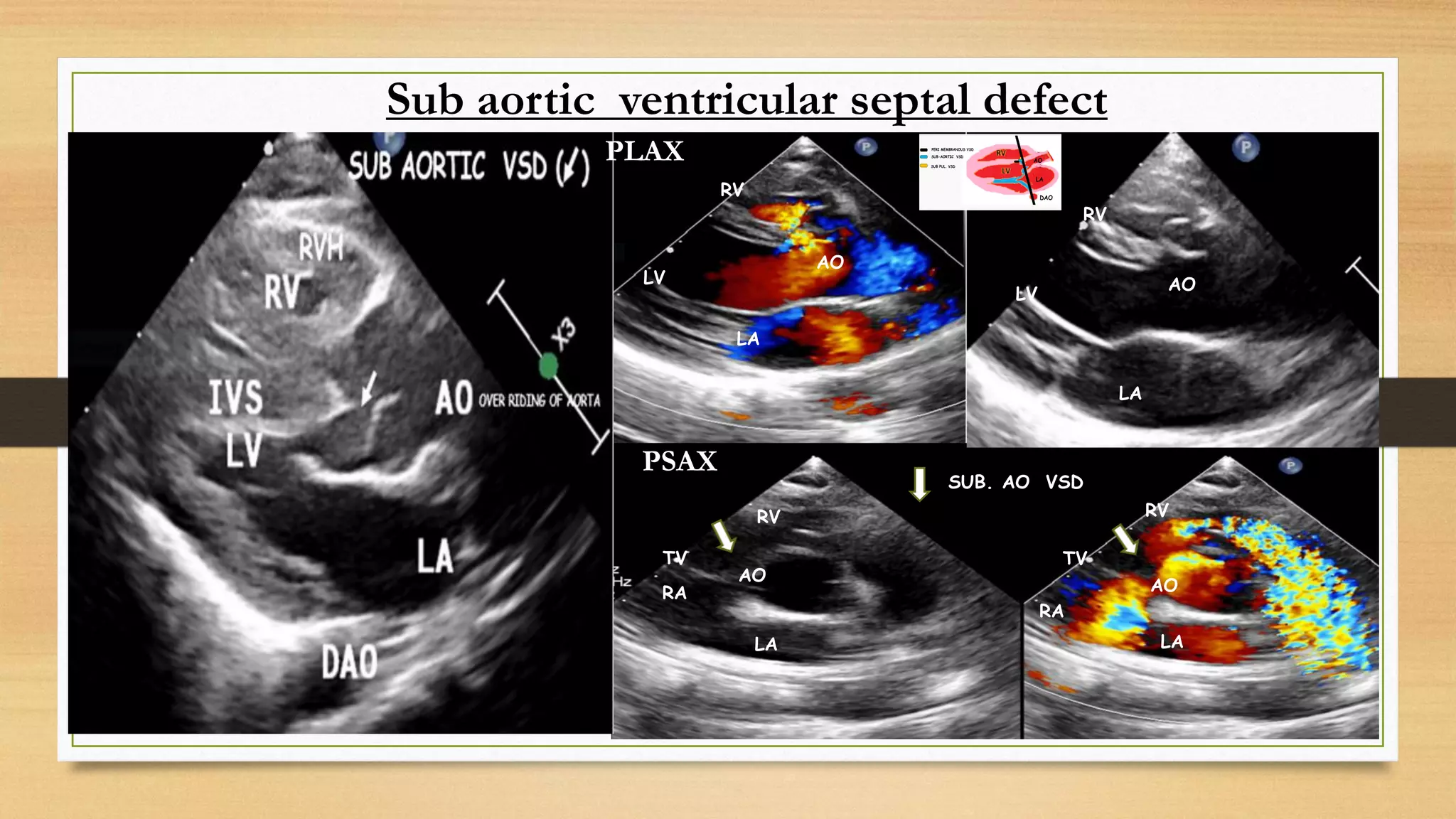
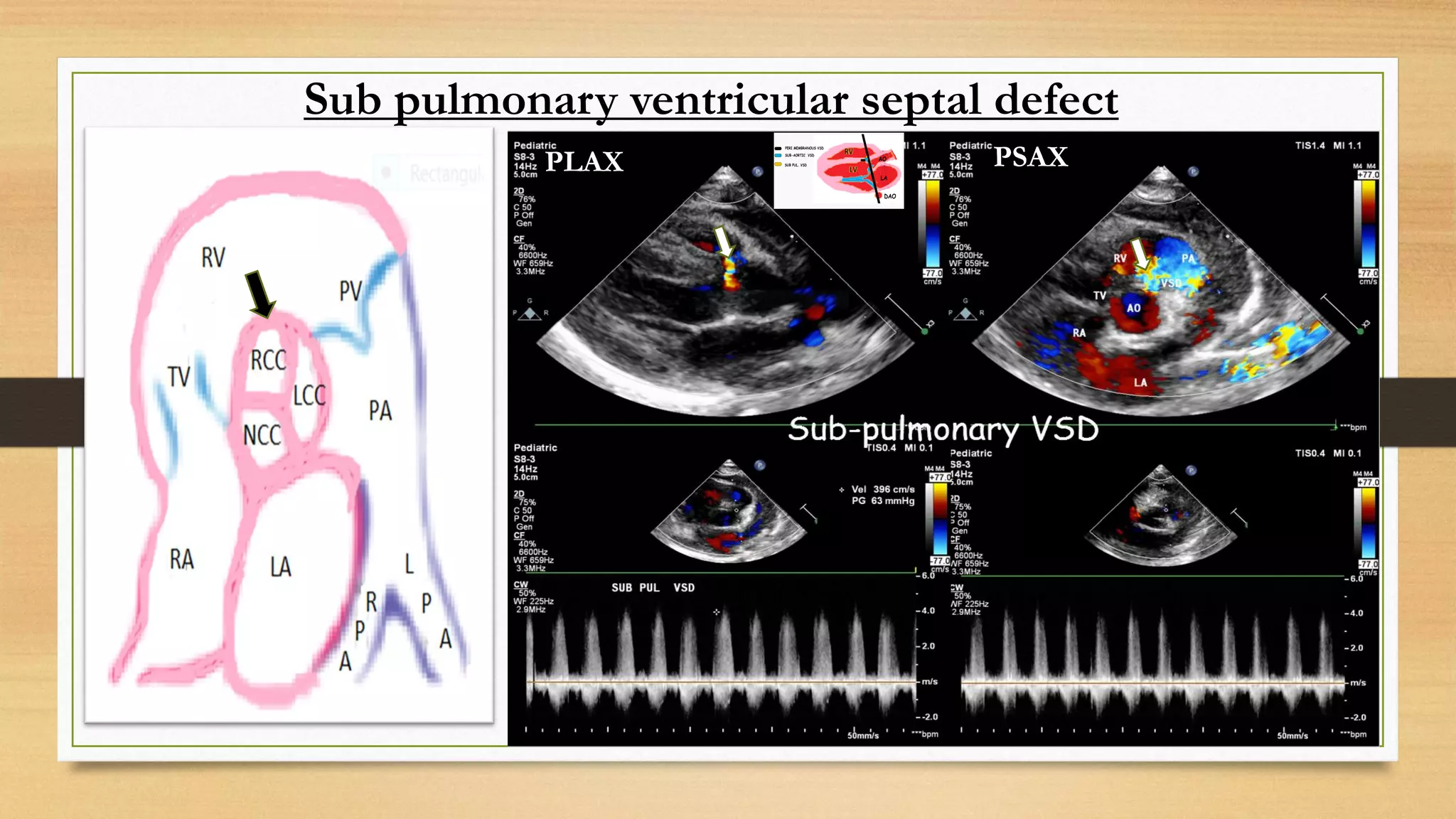
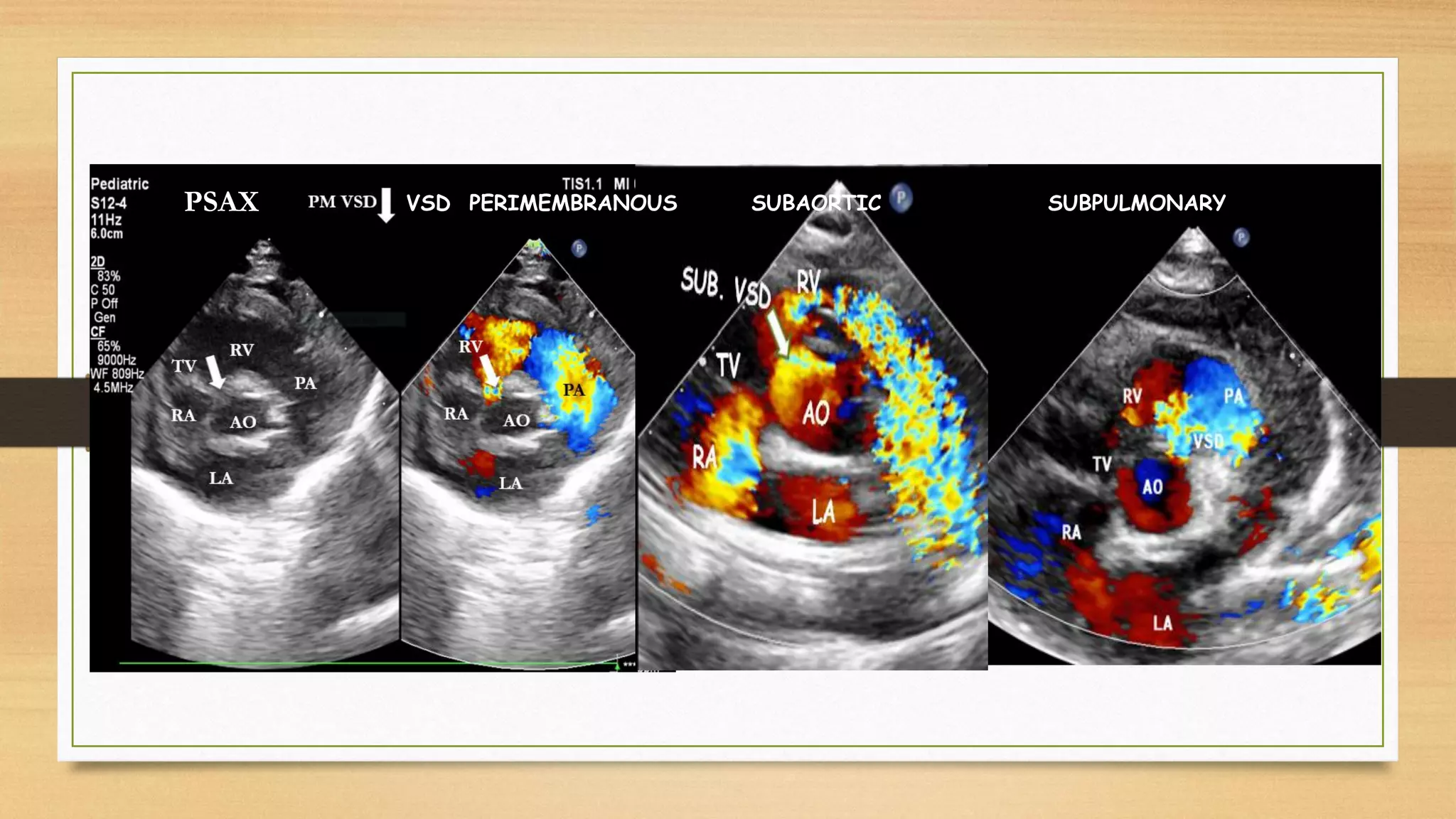
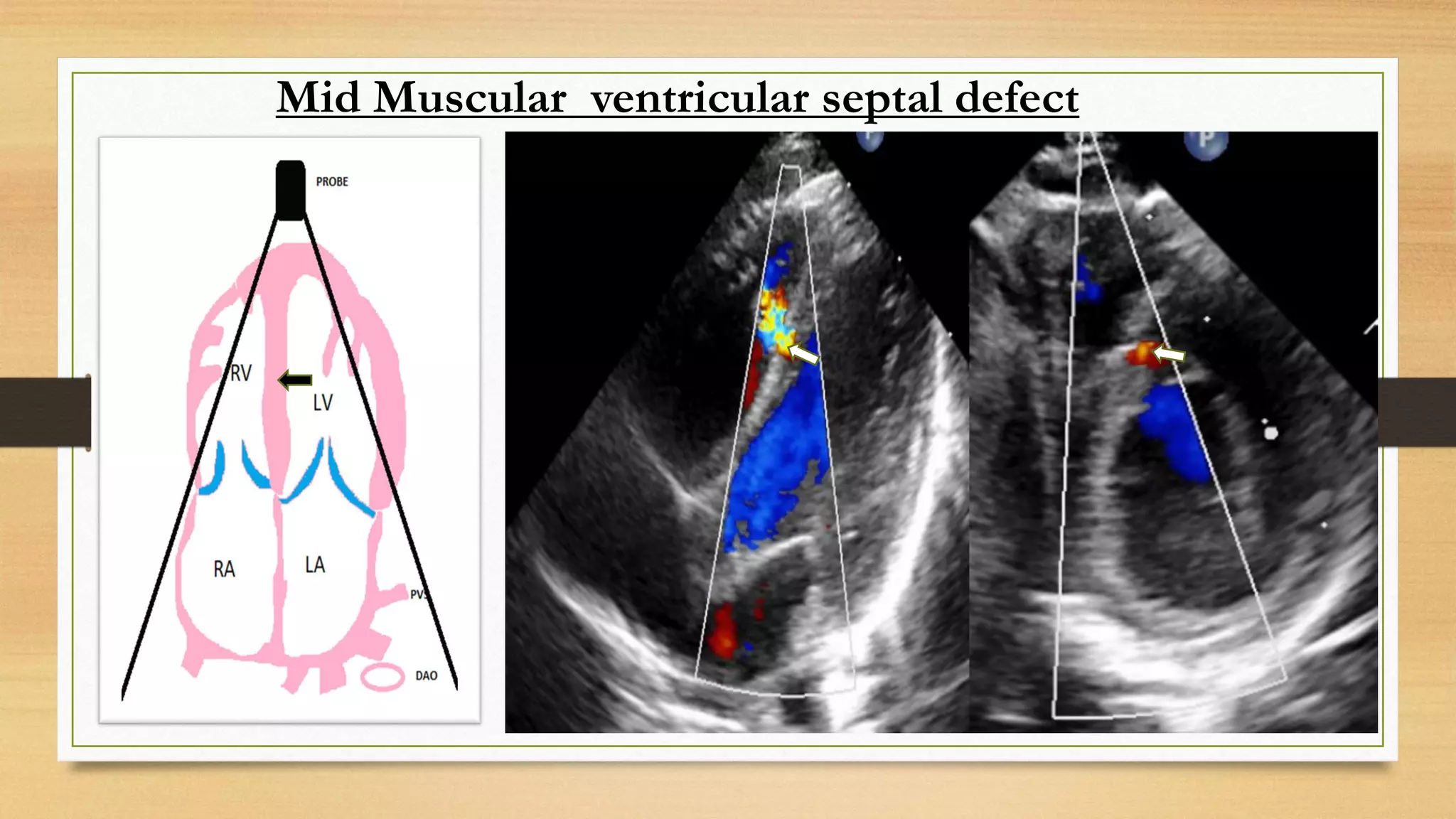
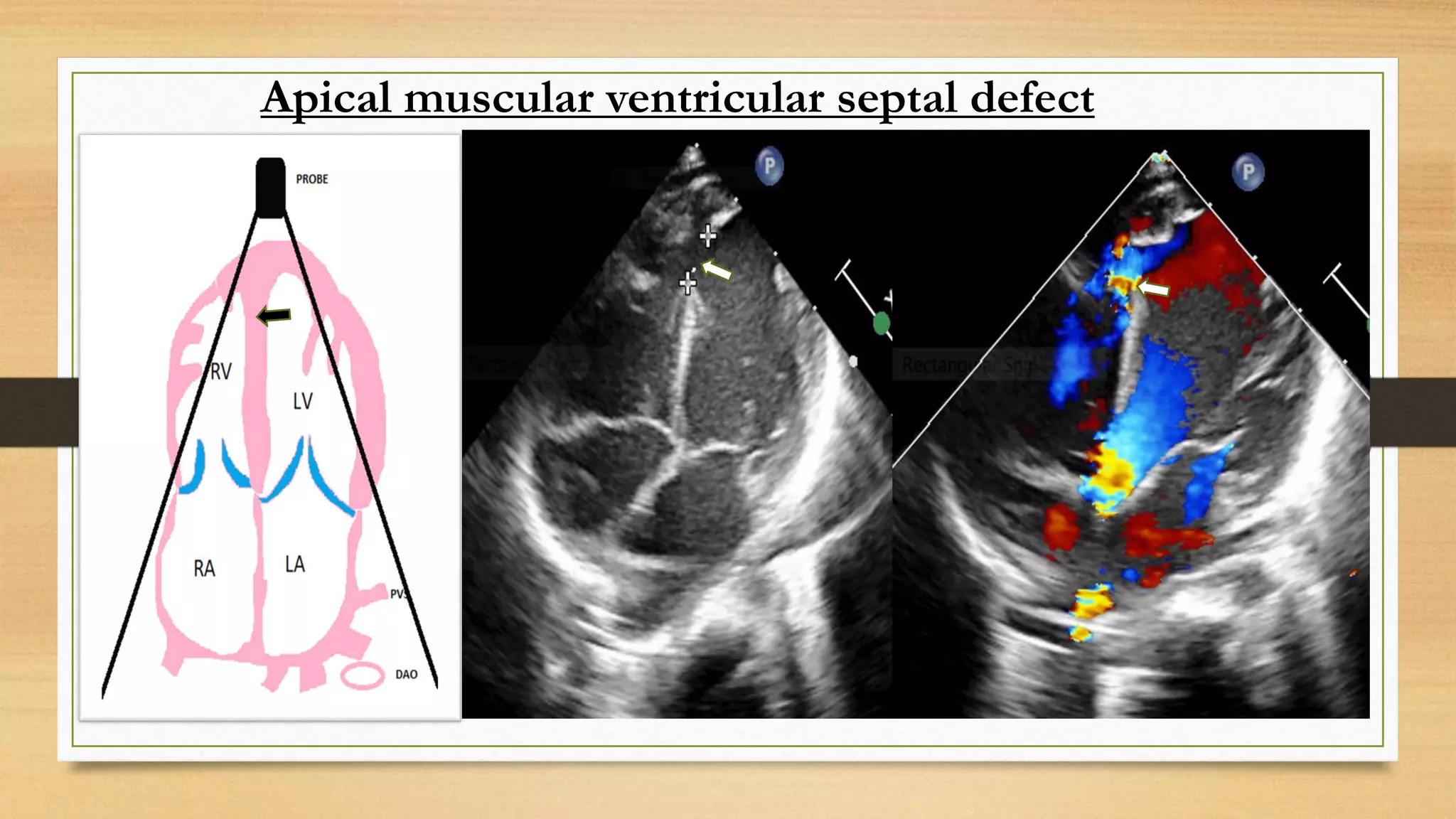
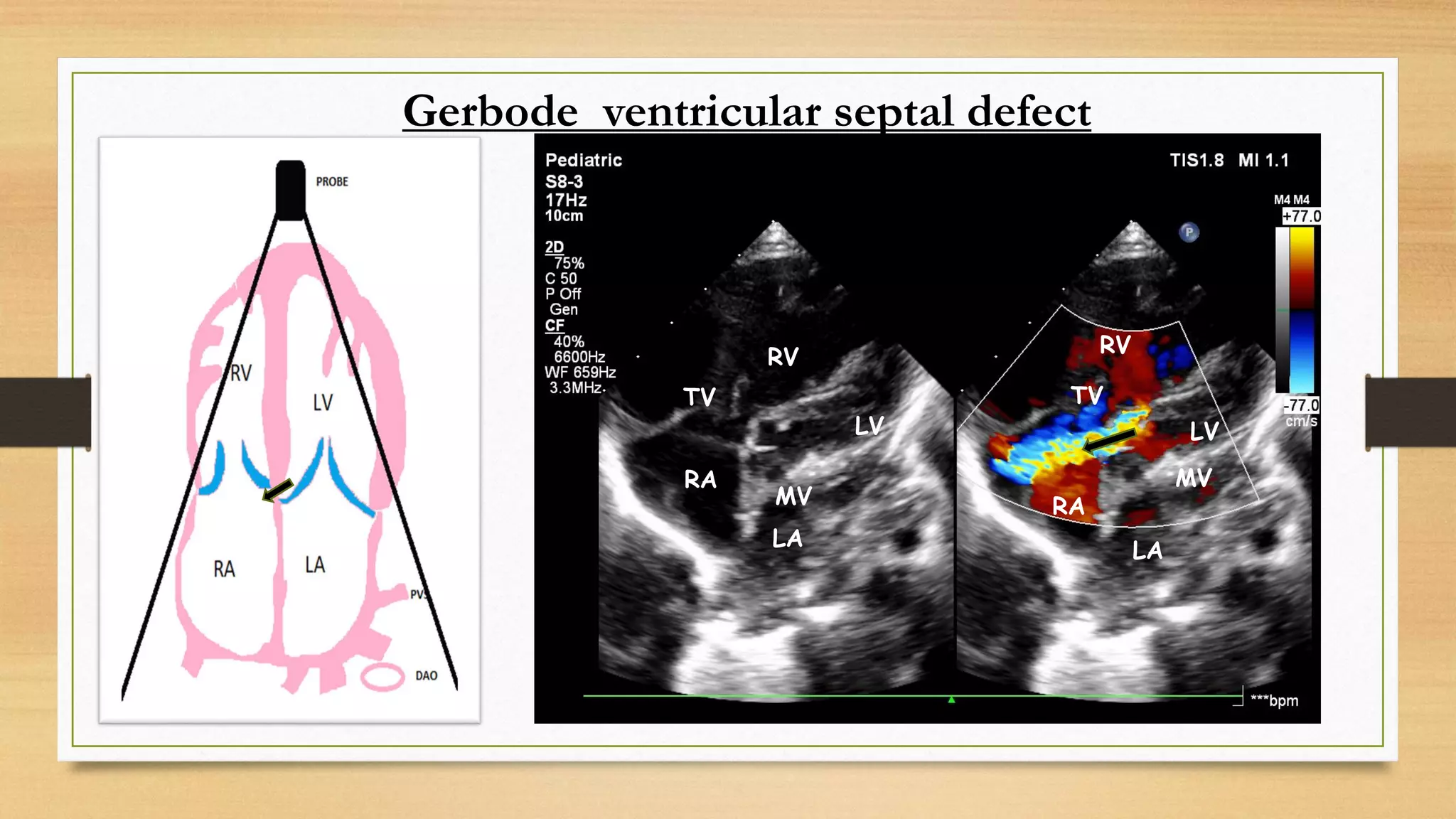
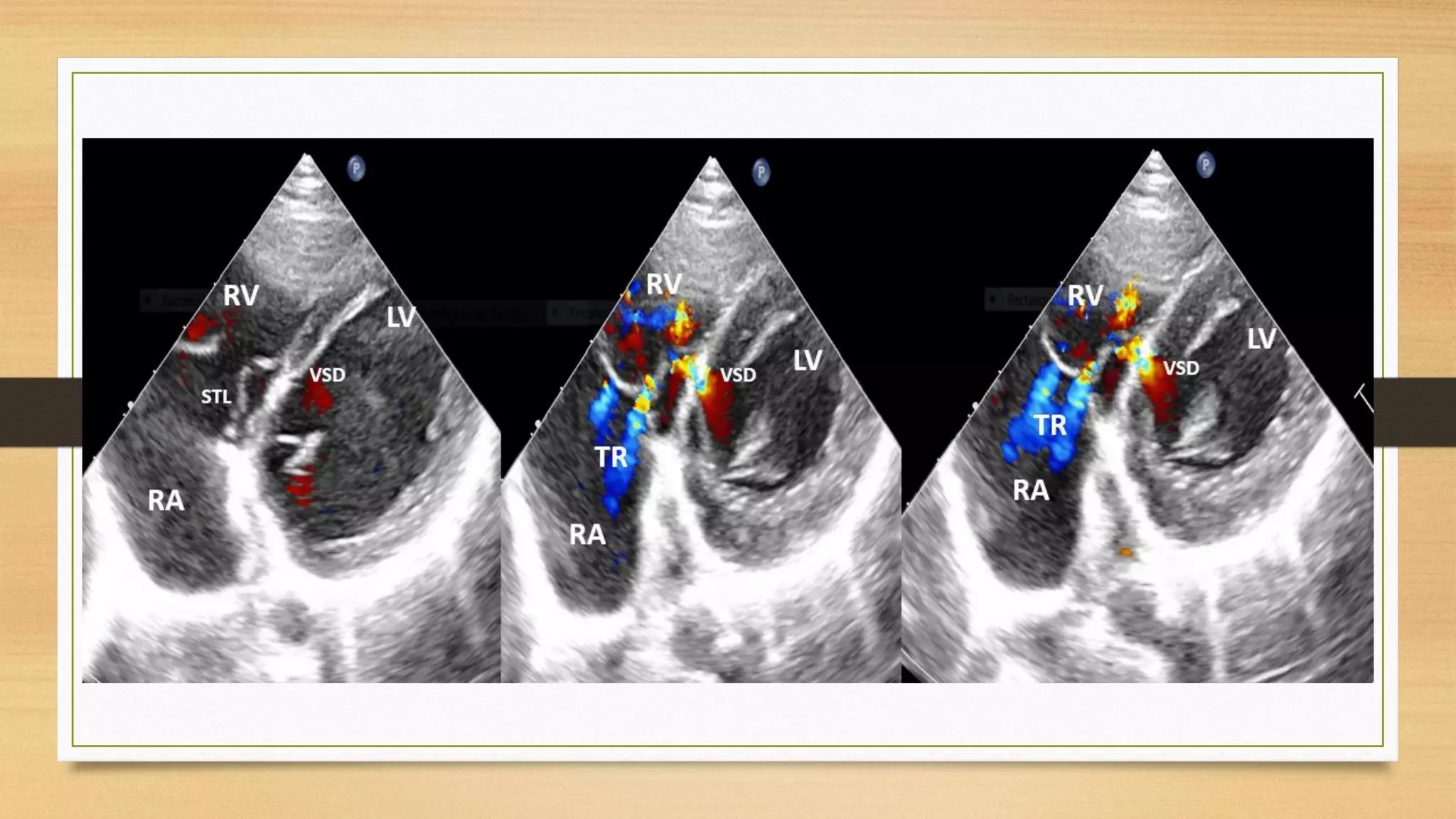
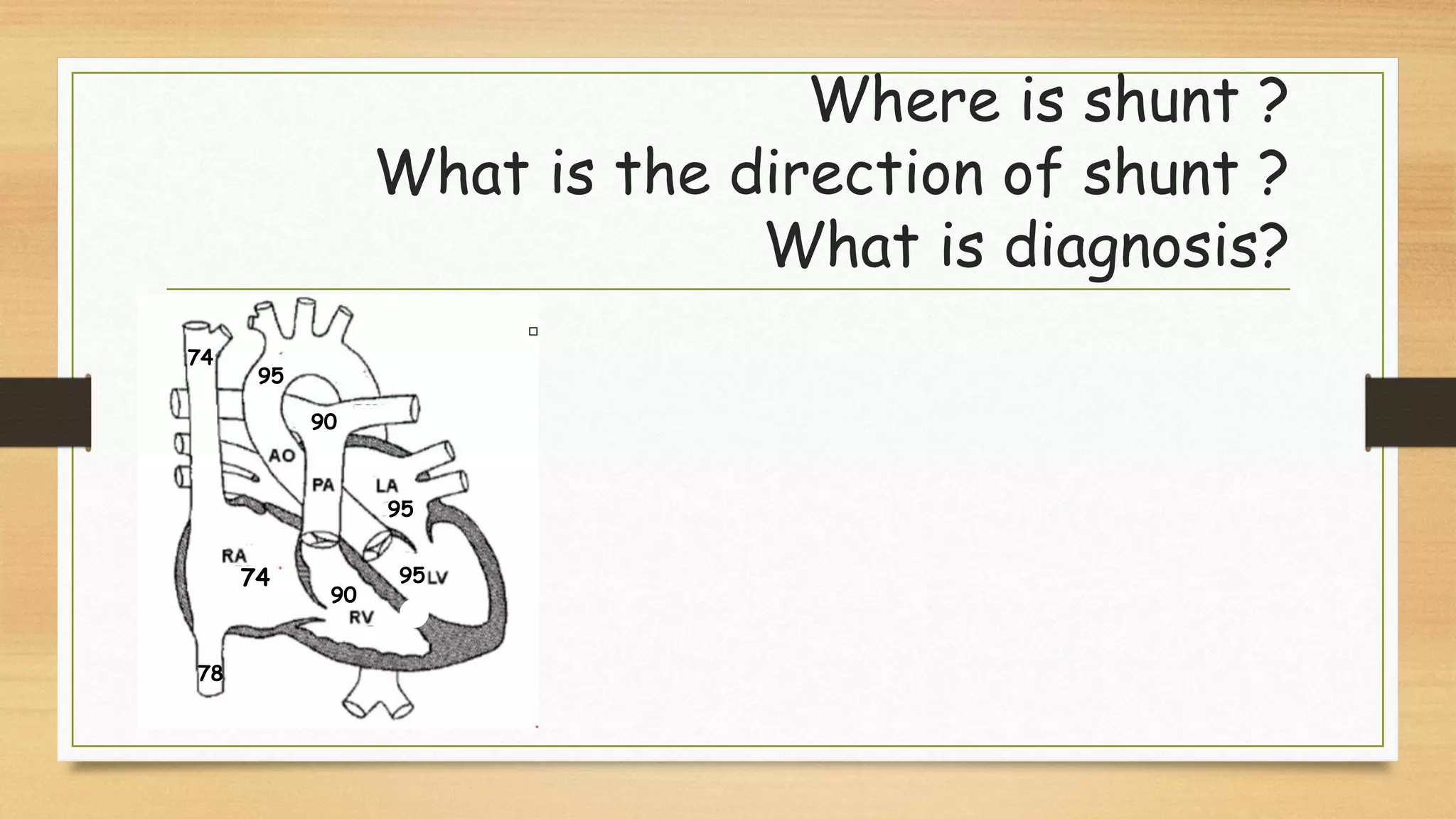
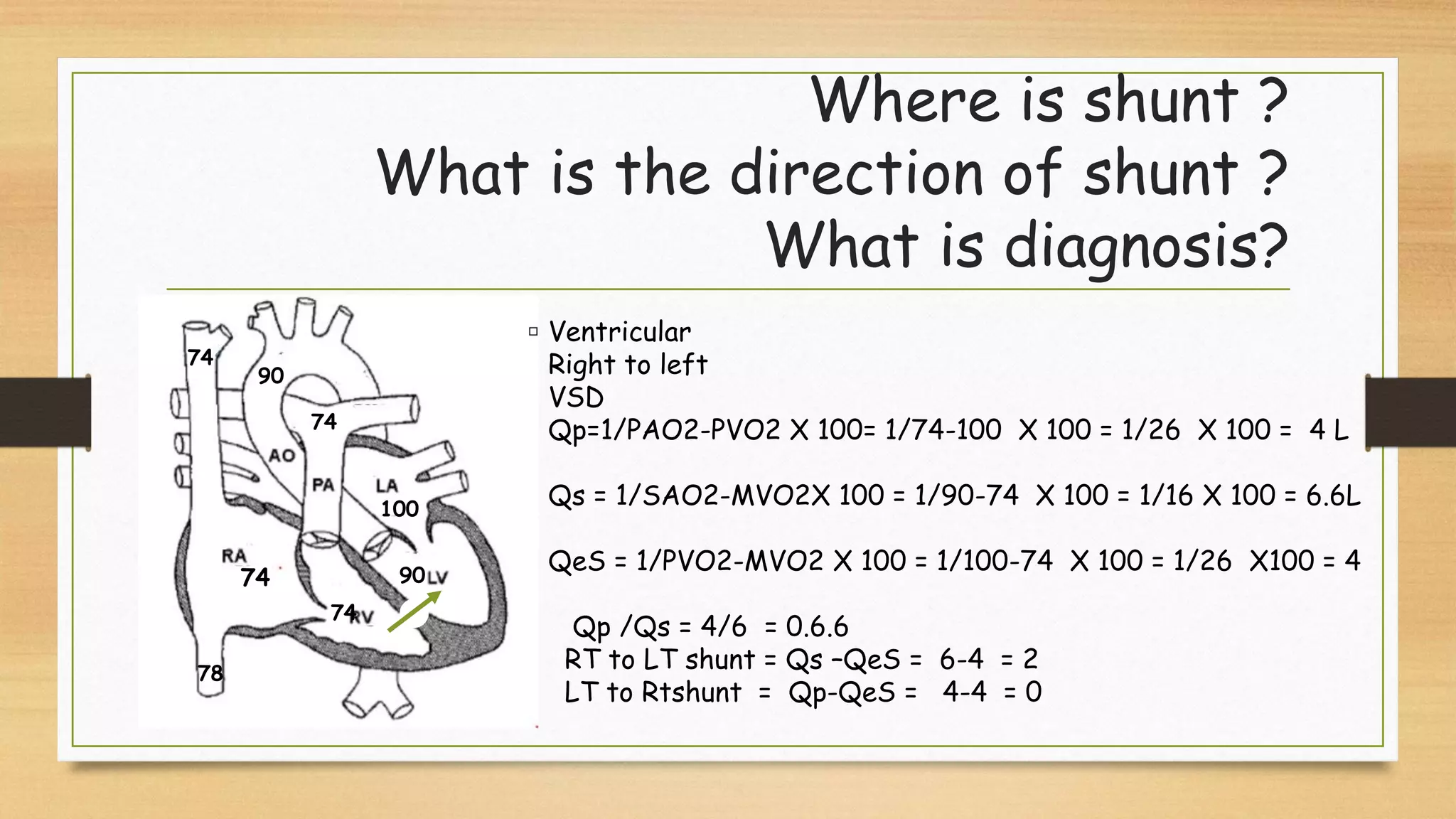
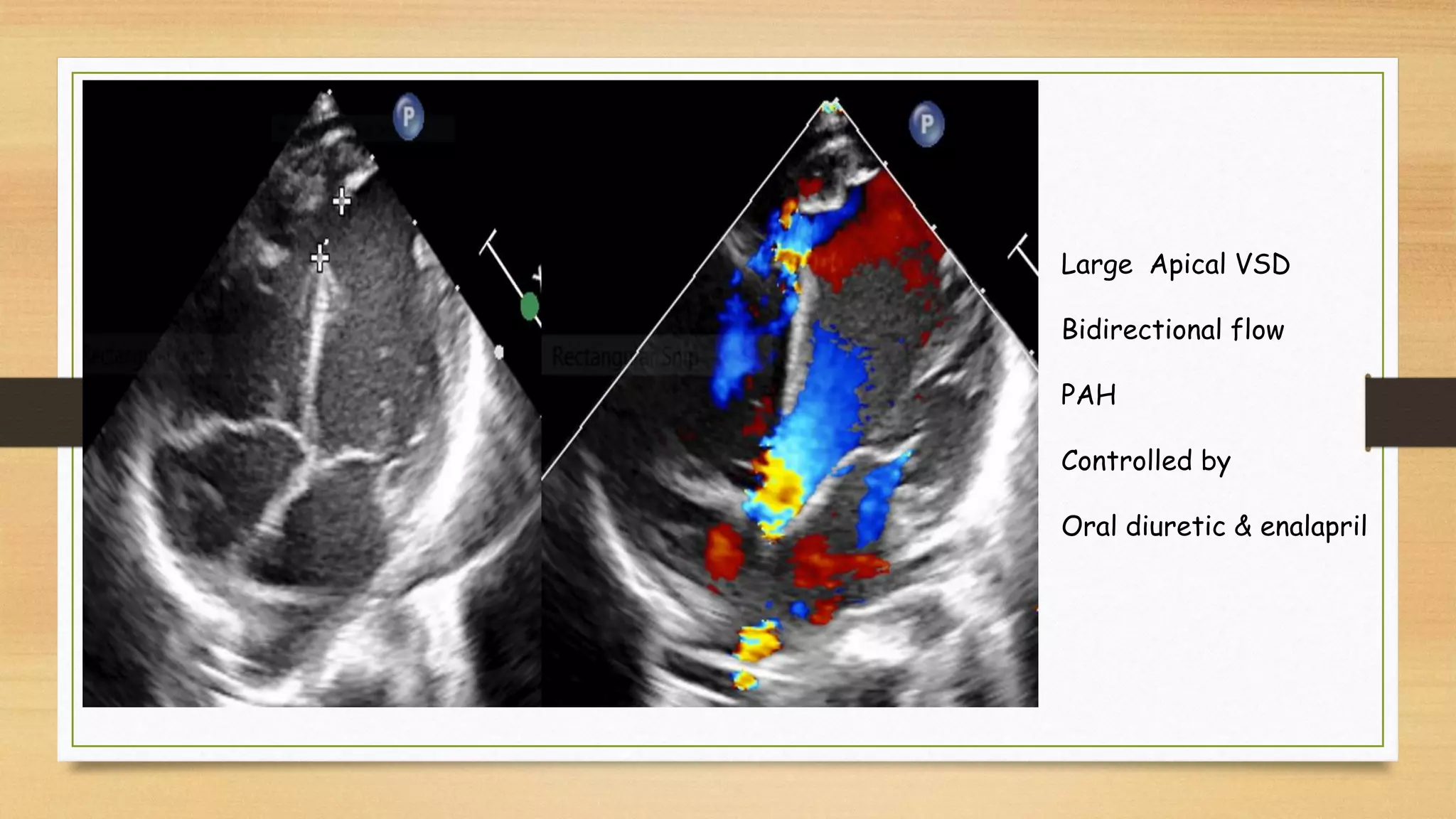
The document discusses ventricular septal defects (VSD), the most common congenital heart defect, detailing their classifications, embryology, clinical manifestations, and management strategies. It highlights the prevalence of VSDs at 3 to 5 per 1000 live births, with increased detection due to echocardiography, and outlines the varying prognoses based on defect size and location. Additionally, it addresses associated risks, complications, and necessary investigations for diagnosis, as well as genetic implications for affected fetuses.


![Ventricular septal defect (VSD) is the most common congenital heart defect (excluding
bicuspid aortic valve [BAV]) and constitutes 20%–30% of all congenital heart defects.
The prevalence varies from 3 to 5/1000 live births. However, a much higher prevalence
(50/1000 live births) is reported due to ease of detection of small muscular VSDs by
echocardiography.
Clinical manifestations depend on the size of the defect and the pulmonary and systemic
vascular resistances. About 10% of patients with large VSDs die in 1st year, primarily due to
congestive heart failure.
Rate of spontaneous closure depends on the size and location of the defect. Spontaneous
closure is uncommon in large VSDs. Inlet and malaligned VSDs almost never close spontaneously.](https://image.slidesharecdn.com/vsdmay2021-210513150154/75/V-s-d-may-2021-4-2048.jpg)