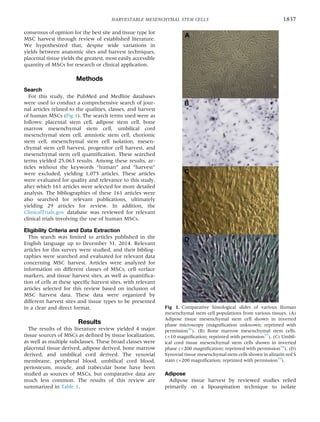
This literature review finds that umbilical cord tissue, specifically Wharton's jelly, offers the greatest number of harvestable mesenchymal stem cells. The review analyzed 161 studies reporting mesenchymal stem cell yields from various tissue sources, including adipose tissue, bone marrow, umbilical cord tissue, and placental tissue. It found that yields from umbilical cord tissue ranged from 10,000 to 4,700,000 cells per milliliter, far exceeding yields from other sources. Adipose tissue provided the next highest yields, ranging from 4,737 to 1,550,000 cells per milliliter. Bone marrow yields ranged more widely from 1 to 317,400 cells per millil