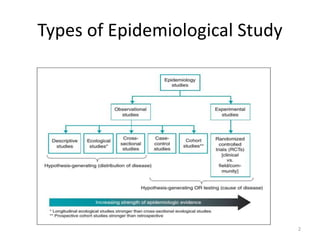
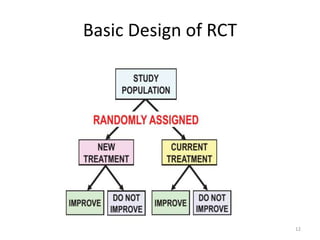
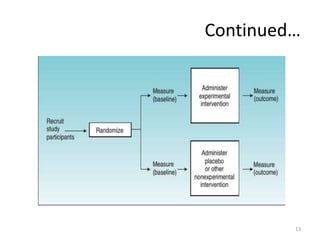
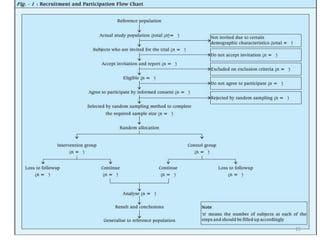
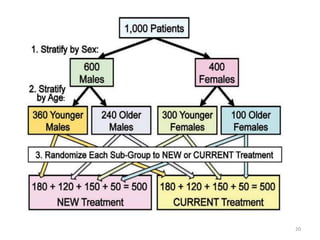
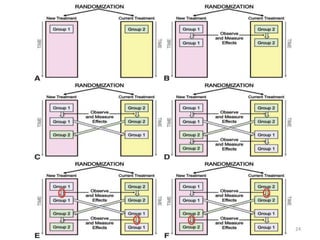
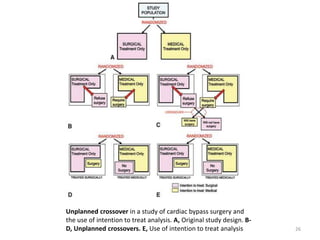
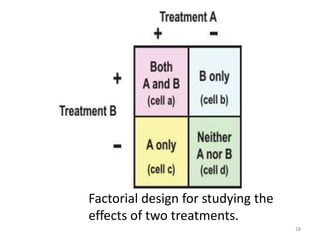
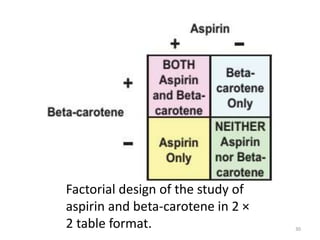
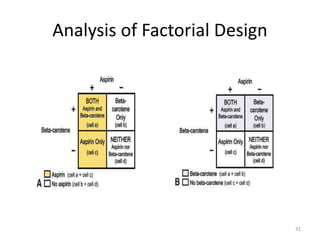
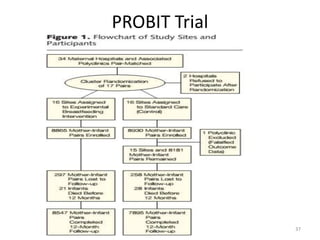
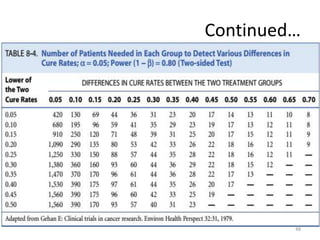
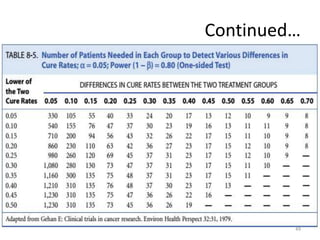
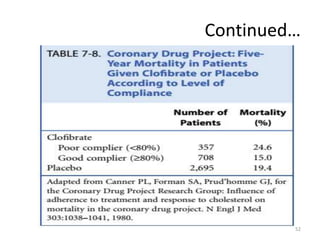
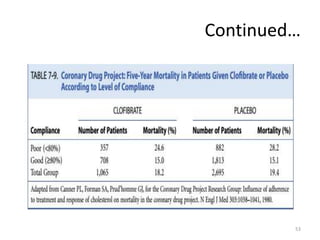
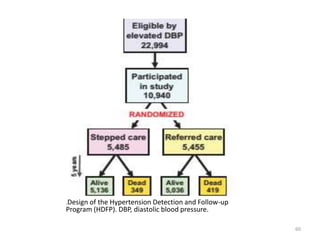
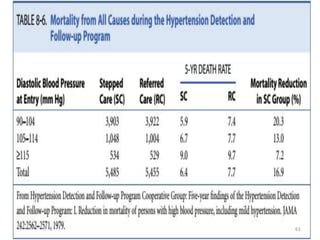
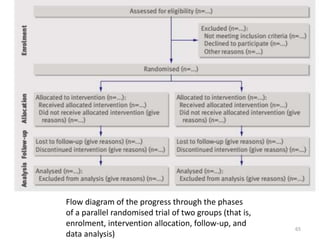
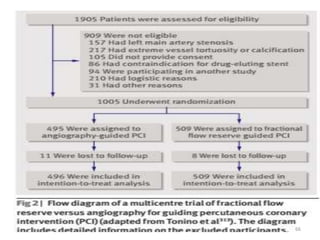
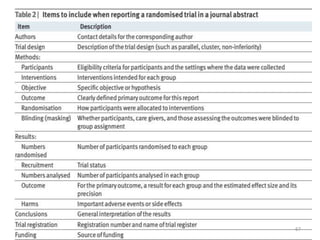
Randomized controlled trials (RCTs) are considered the gold standard for evaluating the efficacy of therapeutic, preventive, and other measures. There are different types of RCT designs, including stratified, crossover, factorial, and cluster RCTs. Key steps in conducting an RCT include developing a protocol, selecting and randomizing a study population, implementing the intervention, following up participants, and assessing outcomes. RCTs aim to reduce biases by creating comparable intervention and control groups through randomization. While powerful, RCTs also have limitations such as cost, time requirements, and lack of applicability to entire populations. Reporting guidelines like CONSORT provide guidance on transparently reporting RCT methods and results.