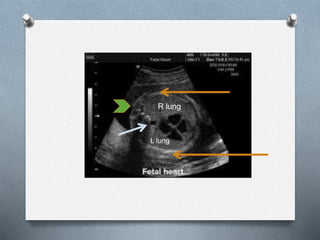
The document discusses various fetal thoracic abnormalities that can be observed on ultrasound, including:
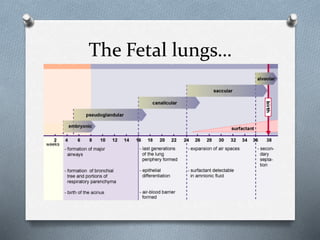
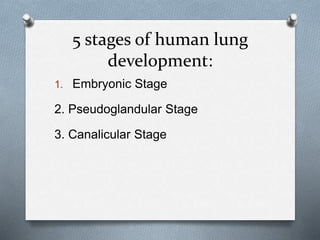
1. The 5 stages of lung development: embryonic, pseudoglandular, canalicular, saccular, and alveolar.
2. Specific lung abnormalities like lung hypoplasia, CCAM (congenital cystic adenomatoid malformation), bronchopulmonary sequestration, CHAOS (congenital high airway obstruction syndrome), and others.
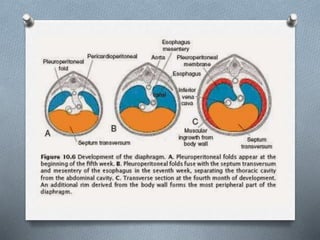
3. Details on diaphragmatic development and abnormalities like CDH (congenital diaphragmatic hernia).
4. Other thoracic issues like congenital hydrothorax. Diagnostic features,