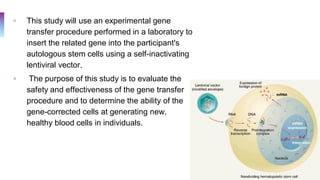
A 25-year-old Canadian woman of Vietnamese origin presented with mild microcytic anemia during routine prenatal care. Testing revealed she had beta-thalassemia trait due to a nonsense mutation in her beta-globin gene. Her Greek husband also carried the same mutation, giving them a 25% risk of having a child with beta-thalassemia major. Beta-thalassemia is caused by a deficiency in alpha or beta globin production and results in anemia and other symptoms. Current treatments include blood transfusions, chelation therapy, and potentially gene therapy through stem cell gene correction.
![The Case
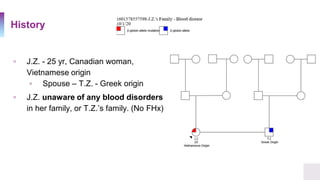
▫ A 25 year old Healthy Canadian woman, presented to her
obstetrician for routine prenatal care.
▫ Results of her complete blood count showed signs of a mild
microcytic anemia
▫ Hemoglobin 98 g/L [ref. 121-151 g/L]
▫ Mean Corpuscular Volume 75 µm³ [ref. 87 ± 5]](https://image.slidesharecdn.com/thalassemia-dilinaaarewattecase44-201008082325/85/Thalassemia-Case-Presentation-2-320.jpg)




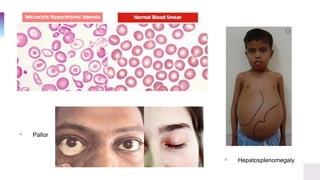
![Clinical Symptoms & Diagnosis
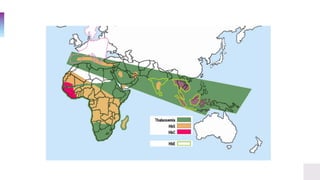
▫ In a population, the α-globin mutations are reflected by the
phenotypes observed in the population.
▫ Clinical symptoms
▫ Anemia [hypochromic
microcytic]
▫ Pallor
▫ Fatigue
▫ Hepatosplenomegaly](https://image.slidesharecdn.com/thalassemia-dilinaaarewattecase44-201008082325/85/Thalassemia-Case-Presentation-9-320.jpg)