Tang 02 alcohols, aldehydes, ketones 2
•
4 likes•1,492 views
This document discusses the nomenclature and properties of alcohols, aldehydes, and ketones. It explains that alcohols contain an -OH functional group and are named by replacing the -e with -ol. Aldehydes have a C=O group at the end of the carbon chain and are named by replacing -e with -al, while ketones have a C=O group within the chain and use -one. Alcohols have higher boiling points than hydrocarbons due to hydrogen bonding, while aldehydes and ketones have lower boiling points without hydroxyl groups.
Report
Share
Report
Share
Recommended
Tang 06 alcohols, aldehydes, ketones 2015

Alcohols, aldehydes, and ketones are organic compounds that contain hydroxyl (-OH), aldehyde (-CHO), or ketone (>C=O) functional groups. The IUPAC naming system is used to systematically name these compounds based on their carbon chain length and functional group position. Alcohols can be primary, secondary, or tertiary depending on whether the hydroxyl group is bonded to a primary, secondary, or tertiary carbon. Aldehydes and ketones differ in that aldehydes have the carbonyl group at the end of the carbon chain while ketones have it within the chain. Alcohols have higher boiling points than hydrocarbons due to hydrogen bonding between hydroxy
Tang 02 alcohols, aldehydes, ketones 2

- Alcohols contain an -OH functional group and are named with the suffix -ol. Shorter chain alcohols are more soluble in water due to hydrogen bonding between OH groups.
- Aldehydes contain a C=O group at the end of the carbon chain and are named with the suffix -al. Ketones contain a C=O group within the chain and are named with the suffix -one.
- Aldehydes and ketones have lower boiling points than alcohols due to an inability to form strong hydrogen bonds like alcohols. They are also less soluble in water for the same reason.
Tang 01 organic chemistry and alkanes

This document discusses organic chemistry, specifically alkanes. It begins by defining organic chemistry and noting that organic molecules contain carbon along with hydrogen, oxygen, nitrogen, and sometimes sulfur and phosphorus. It then discusses what allows for diversity in organic compounds, namely carbon's ability to form four bonds in different combinations. The document goes on to define hydrocarbons as organic molecules composed only of carbon and hydrogen, categorizing them into alkanes, alkenes, and alkynes based on single, double, or triple carbon bonds. Most of the document then focuses on alkanes, discussing their properties, IUPAC naming conventions including naming straight-chain and branched alkanes, drawing their line structures, and noting
Functional group of organic compounds

This document discusses the different functional groups of organic compounds including hydrocarbons, alcohols, aldehydes, ketones, ethers, esters, amines, amides, and aromatic hydrocarbons. It defines each group and provides examples to illustrate their structures and naming conventions. The key types of isomerism - constitutional and stereoisomerism - are also introduced.
Organic Chemistry: Classification of Organic Compounds: Seminar

This document discusses various organic functional groups including alcohols, aldehydes, ketones, carboxylic acids, esters, ethers, amines, amides, halogenoalkanes, nitriles, nitro compounds, and thiols. It provides examples of each functional group and discusses their classifications. Primary, secondary, and tertiary alcohols and amines are defined. Common illegal and recreational drugs like amphetamines, aspirin, cannabis, LSD, cocaine, morphine, and codeine are analyzed in terms of their functional group components.
Tang 08 reactions intro (ethers, esters, amides) 2015

This document discusses 5 types of organic reactions: condensation, addition, substitution, elimination, and oxidation reactions. It focuses on condensation reactions, which form new functional groups by releasing a water molecule. The 3 types of condensation reactions covered are ether formation, ester formation, and amide formation. Ethers are formed from alcohol condensation, esters from acid-alcohol condensation, and amides from acid-amine condensation. Their IUPAC naming conventions and characteristic properties are also described.
Organic Chemistry: Classification of Organic Compounds

Lecture materials for the Introductory Chemistry course for Forensic Scientists, University of Lincoln, UK. See http://forensicchemistry.lincoln.ac.uk/ for more details.
06 nomenclature _hydrocarbons__deriv._i

1. The document discusses the nomenclature and classification of organic compounds including hydrocarbons, alcohols, phenols, ethers, thiols, carbonyl compounds, and carboxylic acids.
2. It describes the IUPAC principles for systematic naming including functional, substitutive, conjunctive, additive, subtractive, and replacement names.
3. The main reactions discussed are substitutions and additions for alkenes, aromaticity and electrophilic substitutions for arenes.
Recommended
Tang 06 alcohols, aldehydes, ketones 2015

Alcohols, aldehydes, and ketones are organic compounds that contain hydroxyl (-OH), aldehyde (-CHO), or ketone (>C=O) functional groups. The IUPAC naming system is used to systematically name these compounds based on their carbon chain length and functional group position. Alcohols can be primary, secondary, or tertiary depending on whether the hydroxyl group is bonded to a primary, secondary, or tertiary carbon. Aldehydes and ketones differ in that aldehydes have the carbonyl group at the end of the carbon chain while ketones have it within the chain. Alcohols have higher boiling points than hydrocarbons due to hydrogen bonding between hydroxy
Tang 02 alcohols, aldehydes, ketones 2

- Alcohols contain an -OH functional group and are named with the suffix -ol. Shorter chain alcohols are more soluble in water due to hydrogen bonding between OH groups.
- Aldehydes contain a C=O group at the end of the carbon chain and are named with the suffix -al. Ketones contain a C=O group within the chain and are named with the suffix -one.
- Aldehydes and ketones have lower boiling points than alcohols due to an inability to form strong hydrogen bonds like alcohols. They are also less soluble in water for the same reason.
Tang 01 organic chemistry and alkanes

This document discusses organic chemistry, specifically alkanes. It begins by defining organic chemistry and noting that organic molecules contain carbon along with hydrogen, oxygen, nitrogen, and sometimes sulfur and phosphorus. It then discusses what allows for diversity in organic compounds, namely carbon's ability to form four bonds in different combinations. The document goes on to define hydrocarbons as organic molecules composed only of carbon and hydrogen, categorizing them into alkanes, alkenes, and alkynes based on single, double, or triple carbon bonds. Most of the document then focuses on alkanes, discussing their properties, IUPAC naming conventions including naming straight-chain and branched alkanes, drawing their line structures, and noting
Functional group of organic compounds

This document discusses the different functional groups of organic compounds including hydrocarbons, alcohols, aldehydes, ketones, ethers, esters, amines, amides, and aromatic hydrocarbons. It defines each group and provides examples to illustrate their structures and naming conventions. The key types of isomerism - constitutional and stereoisomerism - are also introduced.
Organic Chemistry: Classification of Organic Compounds: Seminar

This document discusses various organic functional groups including alcohols, aldehydes, ketones, carboxylic acids, esters, ethers, amines, amides, halogenoalkanes, nitriles, nitro compounds, and thiols. It provides examples of each functional group and discusses their classifications. Primary, secondary, and tertiary alcohols and amines are defined. Common illegal and recreational drugs like amphetamines, aspirin, cannabis, LSD, cocaine, morphine, and codeine are analyzed in terms of their functional group components.
Tang 08 reactions intro (ethers, esters, amides) 2015

This document discusses 5 types of organic reactions: condensation, addition, substitution, elimination, and oxidation reactions. It focuses on condensation reactions, which form new functional groups by releasing a water molecule. The 3 types of condensation reactions covered are ether formation, ester formation, and amide formation. Ethers are formed from alcohol condensation, esters from acid-alcohol condensation, and amides from acid-amine condensation. Their IUPAC naming conventions and characteristic properties are also described.
Organic Chemistry: Classification of Organic Compounds

Lecture materials for the Introductory Chemistry course for Forensic Scientists, University of Lincoln, UK. See http://forensicchemistry.lincoln.ac.uk/ for more details.
06 nomenclature _hydrocarbons__deriv._i

1. The document discusses the nomenclature and classification of organic compounds including hydrocarbons, alcohols, phenols, ethers, thiols, carbonyl compounds, and carboxylic acids.
2. It describes the IUPAC principles for systematic naming including functional, substitutive, conjunctive, additive, subtractive, and replacement names.
3. The main reactions discussed are substitutions and additions for alkenes, aromaticity and electrophilic substitutions for arenes.
Chapter 21.1 : Functional Groups and Classes of Organic Compounds

This document defines functional groups and discusses various classes of organic compounds organized by their functional groups, including alcohols, alkyl halides, ethers, aldehydes, ketones, carboxylic acids, esters, and amines. It provides the general formulas and properties of each class and describes their naming conventions based on the functional group present. Key points covered include that functional groups determine properties, common uses of each class, and relationships between structure and reactivity.
Oc 02 Functional Groups Handout

This document discusses common functional groups found in organic compounds. It defines functional groups as atoms or groups of atoms that confer similar chemical properties and reactivity. The document then lists and provides examples of common functional groups including alkanes, alkenes, alkynes, aromatics, haloalkanes, alcohols, ethers, aldehydes, ketones, carboxylic acids, esters, amines, and amides. It emphasizes that functional groups are important for classifying organic compounds, identifying sites of chemical reactions, and naming organic compounds.
Tang 07 carboxylic acids, amines, & thiols 2015

This document discusses carboxylic acids, amines, and thiols. It provides information on their IUPAC naming systems, properties, and examples of naming structures for each functional group. For carboxylic acids, it describes how the "-oic acid" suffix is used in IUPAC naming and how they have higher boiling points than hydrocarbons. For amines, it explains how the "amine" suffix is used and that tertiary amines have lower melting/boiling points than primary or secondary amines. Thiols contain a sulfhydryl group and have strong odors, with the "thiol" suffix added in IUPAC naming.
Functional groups

This document provides information on common functional groups found in organic chemistry. It defines functional groups as specific atomic or bond arrangements within molecules that determine characteristic chemical reactions. The document then lists numerous common functional groups organized by element, including their structural formulas, prefixes, and suffixes. It also defines names used for radical species and moieties.
Esters (Functional Group)

Ester is one of the functional groups in organic chemistry. It is formed by combining alcohols and carboxylic acids in a process called esterification.
Tang 01 organic chemistry and alkanes

Here are the 9 structural isomers of heptane drawn using line structures:
[Draws the 9 line structures of the heptane isomers]
Grade 10 Organic Chemistry

1) Crude oil is a mixture of hydrocarbons formed from the remains of ancient plants and animals over millions of years under high pressure and temperature conditions underground.
2) Fractional distillation is used to separate crude oil into different hydrocarbon fractions like gasoline, kerosene and diesel in a fractionating column based on their boiling points.
3) Short hydrocarbon fractions are collected at the top of the fractionating column while longer fractions are collected at the bottom during the fractional distillation process.
Organic chemistry

This document provides an overview of organic chemistry concepts, including hydrocarbons, organic compounds, and their derivatives. It discusses the structure and properties of carbon-based molecules like alkanes, alkenes, alkynes, aromatics, and polymers. It also covers the four major macromolecules that make up living organisms: proteins, carbohydrates, lipids, and nucleic acids. Finally, it briefly mentions how petroleum and coal can be used to produce synthetic polymers.
Organic Chemistry: Structure and Nomenclature

This document provides an overview of organic chemistry structure and nomenclature. It discusses various ways of representing organic molecules, including 3D formulas, skeletal formulas, and structural formulas. Skeletal formulas are emphasized as they make organic structures easier to understand. The document also covers IUPAC nomenclature rules for naming organic compounds based on functional groups and carbon chain length and structure. Finally, it discusses different types of isomerism that can occur in organic molecules, including structural isomers, stereoisomers (cis-trans and optical), and functional group isomers.
General view of organic chemistry and iupac

Organic chemistry is the study of the structure, properties, composition, reactions, and preparation of carbon-containing compounds, which include not only hydrocarbons but also compounds with any number of other elements, including hydrogen (most compounds contain at least one carbon–hydrogen bond), nitrogen, oxygen,
Nomenclature

The lecture is devoted to newbies in chemistry eager to learn the basics of nomenclature. The tips given re easy and can be practiced to get perfection.
Introduction to organic chemistry Foundation In science

Organic chemistry is the study of carbon compounds. The four main types of hydrocarbons are saturated, unsaturated, aliphatic, and aromatic. Saturated hydrocarbons contain only single bonds and their general formula is CnH2n+2. Unsaturated hydrocarbons contain double or triple bonds and include alkenes and alkynes. Aromatic hydrocarbons contain benzene or benzene-like rings. Functional groups are atoms or groups of atoms that are largely responsible for the chemical behavior of organic compounds. Common functional groups include alcohols, aldehydes, ketones, carboxylic acids, esters, and amines. Organic compounds can exhibit isomerism when they
alcohol

In chemistry, alcohols are organic compounds that carry at least one hydroxyl functional group bound to their aliphatic substructure. The term alcohol originally referred to the primary alcohol ethanol, which is used as a drug and is the main alcohol present in alcoholic beverages.
Organic chemistry nomenclature

The document provides information about organic chemistry nomenclature. It discusses the need for systematic nomenclature given the large number of known organic compounds. It introduces IUPAC nomenclature rules which allow any complex organic compound to be systematically named. The key aspects covered are:
- Root words, primary suffixes to indicate saturation/unsaturation, and secondary suffixes to indicate functional groups are used to systematically name compounds.
- Prefixes are used to indicate substituents.
- Examples demonstrate how to apply nomenclature rules to arrive at IUPAC systematic names for various organic structures.
Organic Chemistry: Introductory Topics

[ Visit http://www.wewwchemistry.com ] This is a summary presentation of the introductory topics in Organic Chemistry, prepared according to the Singapore-Cambridge GCE A Level 9647 H2 Chemistry syllabus.
Unit 6 Organic Chemistry

Organic chemistry revision notes cover the formation of fossil fuels like oil from dead marine organisms under heat and pressure. Crude oil is separated into fractions like gasoline and kerosene through fractional distillation, and combustion produces pollution like carbon monoxide and nitrogen oxides. The energy released during combustion can be measured using a calorimeter. Homologous series are families of compounds with the same functional group and general formula that differ by CH2. Main series include alkanes, alkenes, and alcohols, which are named based on their carbon chain and functional group.
Chapter 2 nomenclature of organic compounds

1. Alkanes, alkenes, alkynes, and aromatics are classes of organic compounds. Nomenclature systems have evolved over time to systematically name organic structures.
2. The IUPAC system involves identifying the parent chain, numbering it to give substituents the lowest numbers, and naming substituents as prefixes to the parent name.
3. Functional groups like alcohols and carboxylic acids are named by identifying the parent chain, replacing the ending with the functional group suffix, and numbering/naming substituents.
Tang 02 alkenes

Alkenes are unsaturated hydrocarbons containing one or more carbon-carbon double bonds. They are named using the IUPAC system which indicates the number of carbons in the longest chain, the location of the double bond, and any substituents. Alkenes can exist as geometric isomers (cis or trans) depending on the orientation of groups around the double bond. They are less volatile than similar alkanes due to stronger intermolecular forces between molecules.
Alcohol, phenol ,and ethers

Alcohols, phenols, and ethers are organic compounds with different structures and physical properties. Alcohols contain a hydroxyl group attached to an alkyl group and have general formula R-OH. Phenols have a hydroxyl group attached to a benzene ring. Ethers have an oxygen atom connected to two hydrocarbon groups. These compounds differ in their solubility, boiling points, and medical and industrial applications due to their varying abilities to form intermolecular bonds like hydrogen bonds. Their structures also determine IUPAC nomenclature rules for systematic naming.
Tang 06 condensations-ethers-esters-amides 2

This document discusses three types of condensation reactions:
1) Ethers are formed from two alcohol groups, releasing a water molecule. Common names use "ether" and IUPAC names specify alkyl groups.
2) Esters are formed from an alcohol and carboxylic acid, known as esterification. IUPAC names specify the alcohol as an alkyl group and change "-oic acid" to "-oate".
3) Amides are formed from a carboxylic acid and amine/ammonia. IUPAC names change "-oic acid" to "-amide" and specify the amine as an alkyl group. These reactions release water.
Tang 06 condensations-ethers-esters-amides 2

This document discusses condensation reactions that form ethers, esters, and amides, where new functional groups are produced through the elimination of a water molecule. Ethers are formed through the condensation of two alcohol groups, esters through an alcohol and a carboxylic acid, and amides through a carboxylic acid and an amine. Examples are provided of naming and drawing the structures of ethers, esters, and amides formed through various condensation reactions.
materi kimia alcohol and ether kelas XII

materi sma alkohol dan eter, pengertian, rumus, dan contoh soal
More Related Content
What's hot
Chapter 21.1 : Functional Groups and Classes of Organic Compounds

This document defines functional groups and discusses various classes of organic compounds organized by their functional groups, including alcohols, alkyl halides, ethers, aldehydes, ketones, carboxylic acids, esters, and amines. It provides the general formulas and properties of each class and describes their naming conventions based on the functional group present. Key points covered include that functional groups determine properties, common uses of each class, and relationships between structure and reactivity.
Oc 02 Functional Groups Handout

This document discusses common functional groups found in organic compounds. It defines functional groups as atoms or groups of atoms that confer similar chemical properties and reactivity. The document then lists and provides examples of common functional groups including alkanes, alkenes, alkynes, aromatics, haloalkanes, alcohols, ethers, aldehydes, ketones, carboxylic acids, esters, amines, and amides. It emphasizes that functional groups are important for classifying organic compounds, identifying sites of chemical reactions, and naming organic compounds.
Tang 07 carboxylic acids, amines, & thiols 2015

This document discusses carboxylic acids, amines, and thiols. It provides information on their IUPAC naming systems, properties, and examples of naming structures for each functional group. For carboxylic acids, it describes how the "-oic acid" suffix is used in IUPAC naming and how they have higher boiling points than hydrocarbons. For amines, it explains how the "amine" suffix is used and that tertiary amines have lower melting/boiling points than primary or secondary amines. Thiols contain a sulfhydryl group and have strong odors, with the "thiol" suffix added in IUPAC naming.
Functional groups

This document provides information on common functional groups found in organic chemistry. It defines functional groups as specific atomic or bond arrangements within molecules that determine characteristic chemical reactions. The document then lists numerous common functional groups organized by element, including their structural formulas, prefixes, and suffixes. It also defines names used for radical species and moieties.
Esters (Functional Group)

Ester is one of the functional groups in organic chemistry. It is formed by combining alcohols and carboxylic acids in a process called esterification.
Tang 01 organic chemistry and alkanes

Here are the 9 structural isomers of heptane drawn using line structures:
[Draws the 9 line structures of the heptane isomers]
Grade 10 Organic Chemistry

1) Crude oil is a mixture of hydrocarbons formed from the remains of ancient plants and animals over millions of years under high pressure and temperature conditions underground.
2) Fractional distillation is used to separate crude oil into different hydrocarbon fractions like gasoline, kerosene and diesel in a fractionating column based on their boiling points.
3) Short hydrocarbon fractions are collected at the top of the fractionating column while longer fractions are collected at the bottom during the fractional distillation process.
Organic chemistry

This document provides an overview of organic chemistry concepts, including hydrocarbons, organic compounds, and their derivatives. It discusses the structure and properties of carbon-based molecules like alkanes, alkenes, alkynes, aromatics, and polymers. It also covers the four major macromolecules that make up living organisms: proteins, carbohydrates, lipids, and nucleic acids. Finally, it briefly mentions how petroleum and coal can be used to produce synthetic polymers.
Organic Chemistry: Structure and Nomenclature

This document provides an overview of organic chemistry structure and nomenclature. It discusses various ways of representing organic molecules, including 3D formulas, skeletal formulas, and structural formulas. Skeletal formulas are emphasized as they make organic structures easier to understand. The document also covers IUPAC nomenclature rules for naming organic compounds based on functional groups and carbon chain length and structure. Finally, it discusses different types of isomerism that can occur in organic molecules, including structural isomers, stereoisomers (cis-trans and optical), and functional group isomers.
General view of organic chemistry and iupac

Organic chemistry is the study of the structure, properties, composition, reactions, and preparation of carbon-containing compounds, which include not only hydrocarbons but also compounds with any number of other elements, including hydrogen (most compounds contain at least one carbon–hydrogen bond), nitrogen, oxygen,
Nomenclature

The lecture is devoted to newbies in chemistry eager to learn the basics of nomenclature. The tips given re easy and can be practiced to get perfection.
Introduction to organic chemistry Foundation In science

Organic chemistry is the study of carbon compounds. The four main types of hydrocarbons are saturated, unsaturated, aliphatic, and aromatic. Saturated hydrocarbons contain only single bonds and their general formula is CnH2n+2. Unsaturated hydrocarbons contain double or triple bonds and include alkenes and alkynes. Aromatic hydrocarbons contain benzene or benzene-like rings. Functional groups are atoms or groups of atoms that are largely responsible for the chemical behavior of organic compounds. Common functional groups include alcohols, aldehydes, ketones, carboxylic acids, esters, and amines. Organic compounds can exhibit isomerism when they
alcohol

In chemistry, alcohols are organic compounds that carry at least one hydroxyl functional group bound to their aliphatic substructure. The term alcohol originally referred to the primary alcohol ethanol, which is used as a drug and is the main alcohol present in alcoholic beverages.
Organic chemistry nomenclature

The document provides information about organic chemistry nomenclature. It discusses the need for systematic nomenclature given the large number of known organic compounds. It introduces IUPAC nomenclature rules which allow any complex organic compound to be systematically named. The key aspects covered are:
- Root words, primary suffixes to indicate saturation/unsaturation, and secondary suffixes to indicate functional groups are used to systematically name compounds.
- Prefixes are used to indicate substituents.
- Examples demonstrate how to apply nomenclature rules to arrive at IUPAC systematic names for various organic structures.
Organic Chemistry: Introductory Topics

[ Visit http://www.wewwchemistry.com ] This is a summary presentation of the introductory topics in Organic Chemistry, prepared according to the Singapore-Cambridge GCE A Level 9647 H2 Chemistry syllabus.
Unit 6 Organic Chemistry

Organic chemistry revision notes cover the formation of fossil fuels like oil from dead marine organisms under heat and pressure. Crude oil is separated into fractions like gasoline and kerosene through fractional distillation, and combustion produces pollution like carbon monoxide and nitrogen oxides. The energy released during combustion can be measured using a calorimeter. Homologous series are families of compounds with the same functional group and general formula that differ by CH2. Main series include alkanes, alkenes, and alcohols, which are named based on their carbon chain and functional group.
Chapter 2 nomenclature of organic compounds

1. Alkanes, alkenes, alkynes, and aromatics are classes of organic compounds. Nomenclature systems have evolved over time to systematically name organic structures.
2. The IUPAC system involves identifying the parent chain, numbering it to give substituents the lowest numbers, and naming substituents as prefixes to the parent name.
3. Functional groups like alcohols and carboxylic acids are named by identifying the parent chain, replacing the ending with the functional group suffix, and numbering/naming substituents.
Tang 02 alkenes

Alkenes are unsaturated hydrocarbons containing one or more carbon-carbon double bonds. They are named using the IUPAC system which indicates the number of carbons in the longest chain, the location of the double bond, and any substituents. Alkenes can exist as geometric isomers (cis or trans) depending on the orientation of groups around the double bond. They are less volatile than similar alkanes due to stronger intermolecular forces between molecules.
What's hot (18)
Chapter 21.1 : Functional Groups and Classes of Organic Compounds

Chapter 21.1 : Functional Groups and Classes of Organic Compounds
Tang 07 carboxylic acids, amines, & thiols 2015

Tang 07 carboxylic acids, amines, & thiols 2015
Introduction to organic chemistry Foundation In science

Introduction to organic chemistry Foundation In science
Similar to Tang 02 alcohols, aldehydes, ketones 2
Alcohol, phenol ,and ethers

Alcohols, phenols, and ethers are organic compounds with different structures and physical properties. Alcohols contain a hydroxyl group attached to an alkyl group and have general formula R-OH. Phenols have a hydroxyl group attached to a benzene ring. Ethers have an oxygen atom connected to two hydrocarbon groups. These compounds differ in their solubility, boiling points, and medical and industrial applications due to their varying abilities to form intermolecular bonds like hydrogen bonds. Their structures also determine IUPAC nomenclature rules for systematic naming.
Tang 06 condensations-ethers-esters-amides 2

This document discusses three types of condensation reactions:
1) Ethers are formed from two alcohol groups, releasing a water molecule. Common names use "ether" and IUPAC names specify alkyl groups.
2) Esters are formed from an alcohol and carboxylic acid, known as esterification. IUPAC names specify the alcohol as an alkyl group and change "-oic acid" to "-oate".
3) Amides are formed from a carboxylic acid and amine/ammonia. IUPAC names change "-oic acid" to "-amide" and specify the amine as an alkyl group. These reactions release water.
Tang 06 condensations-ethers-esters-amides 2

This document discusses condensation reactions that form ethers, esters, and amides, where new functional groups are produced through the elimination of a water molecule. Ethers are formed through the condensation of two alcohol groups, esters through an alcohol and a carboxylic acid, and amides through a carboxylic acid and an amine. Examples are provided of naming and drawing the structures of ethers, esters, and amides formed through various condensation reactions.
materi kimia alcohol and ether kelas XII

materi sma alkohol dan eter, pengertian, rumus, dan contoh soal
Chapter 6 hydroxyl compounds

This document provides an overview of Chapter 6 in Organic Chemistry, which covers hydroxyl compounds (alcohols and phenols). It discusses the nomenclature, classification, physical properties, acidity, and common reactions of alcohols and phenols. Specifically, it describes how to name alcohols according to IUPAC rules and classify them based on the type of carbon to which the hydroxyl group is bonded. It also explains that alcohols have higher boiling points than similar hydrocarbons due to hydrogen bonding, and how their solubility in water depends on factors like chain length. The chapter then covers the acidity of alcohols and phenols, and some major reaction types
00000000lech202.pdf

This document discusses the classification and nomenclature of alcohols, phenols, and ethers according to IUPAC rules. It states that alcohols and phenols can be classified as mono-, di-, tri-, or polyhydric depending on the number of hydroxyl groups. Ethers are classified as simple/symmetrical if the alkyl groups are the same, or mixed/unsymmetrical if they are different. The document then provides examples of common and IUPAC names for some representative alcohols, phenols, and ethers.
Selected topics in Chemistry

Here are the names for the alkynes in the sample exercises:
1. 5-ethyl-3-octyne
2. 2,2-dimethyl-3-pentyne
3. 1-butyne
Alcohols,_Phenols,_Thiols,_and_Ethers_Simple.ppt

This document provides an introduction to alcohols, phenols, thiols, and ethers. It defines each compound group and provides examples. Alcohols contain a hydroxyl group attached to an alkyl group. Phenols have a hydroxyl group attached to an aryl group. Ethers have two alkyl or aryl groups attached to an oxygen atom. Thiols are similar to alcohols but with a sulfur atom replacing oxygen. The document discusses hydrogen bonding in alcohols, solubility, naming conventions, and reactions including oxidation, dehydration, and hydration. It also introduces common phenols, properties and uses of ethers, and applications of thiols.
Introduction To Carbon Compound

This document provides a summary of Lesson 1 from an organic chemistry textbook chapter on carbon compounds. It covers 10 learning objectives related to the unique properties of carbon, isomers, functional groups, saturated vs unsaturated compounds, and IUPAC naming conventions. Key topics include how carbon can form multiple bonds and chains/rings, the importance of functional groups for classifying compounds, and systematic naming of organic molecules.
Pp organic 2

The document provides an overview of organic chemistry concepts including:
1. The main classes of organic compounds are hydrocarbons, alcohols, ethers, aldehydes, ketones, carboxylic acids, esters, amines, and aromatic compounds.
2. Hydrocarbons are divided into alkanes, alkenes and alkynes based on the presence of single, double or triple carbon bonds. Nomenclature provides the naming structure for different compounds.
3. Functional groups determine the properties and reactivity of organic molecules. Common reactions include combustion, substitution, and addition. Isomers have the same molecular formula but different structural formulas.
Or Ganic Intro

This document provides an introduction to organic chemistry for A-level students. It begins with an overview of organic chemistry and the special properties of carbon that allow for the vast number of carbon compounds. It then discusses specific topics like functional groups, isomers, naming conventions (IUPAC), and more. The document is intended to help students understand key concepts in organic chemistry.
Ibdp organic introduction

This document provides an introduction to organic chemistry, focusing on hydrocarbons. It defines organic chemistry as the study of carbon-based compounds, particularly hydrocarbons rather than metal carbonates or oxides. There are four main classes of hydrocarbons discussed: alkanes, alkenes, alkynes, and aromatics. Alkanes contain only single bonds, while alkenes contain double bonds and alkynes contain triple bonds. Functional groups are discussed, which determine the physical and chemical properties of organic compounds. Homologous series are introduced as families of compounds that differ by CH2 units and have similar properties.
Carbono inicial

The document discusses carbon, an essential component of cells. Carbon permeates all living things, from the structural organization of cells to energy requirements and genetic information storage. Carbon is necessary for the existence of the biosphere. The document then discusses how hydrogen and other elements bond covalently to carbon to form important organic compounds in living things like carbohydrates, lipids, proteins, and nucleic acids. It also examines the electronic configuration and hybridization of carbon atoms that allows them to form chains, branches, and rings with other carbon and non-carbon atoms.
Alcohols. Kamran Mammadli

This document provides information about alcohols, including their structure, nomenclature, and classification. It begins by reviewing hydrocarbons and noting that alcohols differ in that they contain an OH (hydroxyl) functional group. Alcohols are classified as primary, secondary, or tertiary depending on whether the alcohol carbon is bonded to 1, 2, or 3 carbon atoms. Naming involves identifying the parent chain and lowest locant for the OH group. Examples of methanol, butanol, and isopropanol are provided to illustrate the nomenclature rules.
P O S T L A B Biochem D L S L

This document summarizes key differences between organic and inorganic compounds based on a biochemistry lab experiment. Organic compounds contain carbon and are found in living organisms, while inorganic compounds do not contain carbon or result from non-living processes. The document outlines tests to detect carbon, hydrogen, oxygen, nitrogen, and sulfur in organic compounds, such as evolving gases upon heating with bases or forming precipitates with reagents. Common organic compounds and their properties are also discussed.
Alcohol chemistry

The document discusses several topics related to chemistry:
1) The voltage needed to create an electron is about one million volts, the same voltage as lightning. This high voltage accelerates electrons from the sky to the ground.
2) Alcohols are derivatives of hydrocarbons where an –OH group replaces a hydrogen. They can act as both acids and bases.
3) Phenols have a hydroxyl group bonded directly to a benzene ring. They are named based on the carbon the hydroxyl group is bonded to, such as phenol itself or cresols which are methyl phenols.
Nomenclature

APEX INSTITUTE was conceptualized in May 2008, keeping in view the dreams of young students by the vision & toil of Er. Shahid Iqbal. We had a very humble beginning as an institute for IIT-JEE / Medical, with a vision to provide an ideal launch pad for serious JEE students . We actually started to make a difference in the way students think and approach problems. We started to develop ways to enhance students IQ. We started to leave an indelible mark on the students who have undergone APEX training. That is why APEX INSTITUTE is very well known of its quality of education
Organic chemistry

This document covers organic chemistry concepts including alkanes, cycloalkanes, and their nomenclature. Key points include:
- The lesson will introduce alkanes and cycloalkanes, with two quizzes to follow. Naming and drawing structural formulas using IUPAC nomenclature will be covered.
- Standards include describing homologous series, naming organic compounds using IUPAC, and drawing and naming alkanes and cyclic structures up to C10.
- Introduction covers bonding rules for carbon, oxygen, nitrogen, hydrogen and halogens in organic compounds. Bonding and structural representations such as Lewis, condensed and line structures are defined.
22.1-Alcohol, Phenols and Ethers(PU).pptx

1) Alcohols are formed when a hydrogen atom of a hydrocarbon is replaced by an -OH group, and ethers are formed when a hydrogen atom is replaced by an alkoxy or aryloxy group.
2) Alcohols and phenols are classified as mono, di, tri, or polyhydric depending on the number of hydroxyl groups in their structure.
3) Mono hydric alcohols are further classified based on whether the -OH group is attached to an sp3, sp2, or aromatic carbon atom.
Similar to Tang 02 alcohols, aldehydes, ketones 2 (20)
More from mrtangextrahelp
17 stoichiometry

64 batteries will allow you to make 64 iPhones. The batteries are the limiting reagent.
Tang 04 periodic trends

1) Atomic radius generally increases down a group and decreases across a period due to shielding effects of additional electron shells and increasing nuclear charge, respectively.
2) Ionization energy generally increases down a group and across a period as it becomes increasingly difficult to remove an electron due to greater nuclear attraction from less shielding and more protons. Exceptions occur when removing an electron would destabilize a full shell.
3) Electronegativity generally increases across a period as the effective nuclear charge felt by valence electrons increases with fewer shielding shells and more protons.
Tang 02 wave quantum mechanic model

This document provides information on wave quantum mechanics and electron configurations. It discusses:
- Erwin Schrodinger's contributions to developing quantum mechanics and proposing the wave-like nature of electrons.
- How electrons occupy distinct energy levels and orbitals around the nucleus, rather than defined circular orbits. Electrons have wave-like properties.
- The shapes of s, p, d and f orbitals and how electrons fill these orbitals according to various principles like Aufbau and Hund's rule.
- Exceptions to the Aufbau principle seen in some elements.
- How to represent electron configurations using both energy level diagrams and shorthand notation.
23 gases

- Gases have negligible volume and molecules are far apart with mostly empty space between them. Gas molecules move randomly in straight lines and collide elastically.
- The kinetic molecular theory describes gas properties including high translational energy of gas molecules that move randomly in all directions.
- Pressure, volume, temperature of gases are directly or inversely related as described by Boyle's, Charles', and Gay-Lussac's laws and the combined gas law.
04 periodic trends v2

- Elements in the same group have similar properties due to having the same number of valence electrons. Atoms get smaller from left to right in a period as protons are added, and larger from top to bottom in a group as energy levels are added.
- Electronegativity, ionization energy, and electron affinity all generally increase from left to right across a period as the effective nuclear charge increases. Electronegativity also increases up a group as the distance from valence electrons to the nucleus decreases.
- Ionization energy increases from top to bottom within a group as it is easier to remove an electron from an atom with fewer energy levels. Electron affinity generally increases from left to right across a period and decreases
22 acids + bases

This document discusses acids and bases. It defines their key properties including reacting with metals, carbonates, conducting electricity, turning litmus paper colors, and neutralizing each other. It explains the theories of Arrhenius, Brønsted-Lowry, and Lewis on acids and bases. It also covers acid-base reactions, indicators, pH, titrations, strong/weak acids and bases, and acid-base stoichiometry.
23 gases

Gases are composed of molecules that are far apart from each other and occupy the entire volume of their container. Gas molecules move randomly in straight lines and collide elastically with each other and the container walls. The kinetic molecular theory describes gases as having negligible intermolecular forces and volume. Pressure, volume, temperature of a gas are related by the combined gas law and ideal gas law. Gas calculations involve using stoichiometry, the ideal gas law, and gas laws like Boyle's, Charles', and Gay-Lussac's to determine volume, pressure, amount, or temperature changes.
22 acids + bases

The document discusses the key properties and reactions of acids and bases. It defines acids as substances that produce hydrogen ions (H+) in water and bases as substances that produce hydroxide ions (OH-). Acids react with metals, carbonates, conduct electricity, turn litmus paper red, and neutralize bases. Bases conduct electricity, turn litmus paper blue, and neutralize acids. Theories of acids and bases including Arrhenius, Brønsted-Lowry, and Lewis are explained. Strong and weak acids/bases, monoprotic/diprotic/triprotic acids, pH, titrations, and acid-base indicators are also covered.
22 solution stoichiometry new

This document provides an overview of solution stoichiometry and includes 3 sample problems demonstrating how to use a balanced chemical equation to solve for unknown quantities. The general steps are: 1) Write the balanced equation, 2) Convert given amounts to moles, 3) Use the mole ratio to calculate moles of the unknown, 4) Convert moles to the required unit. Sample Problem 1 calculates molar concentration of sulfuric acid. Sample Problem 2 determines the minimum volume of sodium carbonate needed. Sample Problem 3 calculates the mass of silver chromate precipitate formed.
21 water treatment

This document discusses water treatment processes including water softening. Water softening is a treatment process that uses chemicals to remove minerals like calcium and magnesium from hard water to make it softer. Acceptable concentrations of chemicals used in water treatment processes are also mentioned.
20 concentration of solutions

- Concentration can be expressed in several ways including mass/volume percent, mass/mass percent, volume/volume percent, parts per million (ppm), and molar concentration (mol/L).
- Mass/volume percent is the mass of solute divided by the volume of solution. Volume/volume percent is the volume of solute divided by the volume of solution.
- Mass/mass percent is the mass of solute divided by the mass of solution. Parts per million (ppm) and parts per billion (ppb) express very small concentrations.
- Molar concentration expresses the number of moles of solute per liter of solution and is the standard unit for expressing concentration in chemistry
22 acids + bases

This document discusses acids and bases according to several theories. It begins by describing the properties of acids, including reacting with metals and carbonates, conducting electricity, turning litmus paper colors, and neutralizing bases. It then discusses the properties of bases. The Arrhenius theory defines acids as substances that produce H+ ions in water and bases as those that produce OH- ions. However, this theory has limitations and does not account for all acids and bases. The Brønsted-Lowry theory broadens the definition to any substance that can donate or accept protons. Strong acids fully dissociate in water while weak acids only partially dissociate. The pH scale measures the concentration of H+ ions on a
19 solutions and solubility

The document discusses key concepts about solutions including:
- The components of a solution are a solvent and one or more solutes. Common solvents include water and gases.
- Factors that affect solubility and the rate of dissolving include temperature, agitation, and surface area of the solute. Increasing these factors generally increases solubility.
- Solutions can be classified as saturated, unsaturated, or supersaturated depending on the amount of solute dissolved compared to the maximum amount possible at a given temperature.
- Solubility is also affected by properties of the solute molecules like polarity, charge, and size - more polar or charged molecules or smaller ions tend to be more soluble.
- Solub
18 percentage yield

The document discusses percentage yield and percentage purity calculations for chemical reactions. It provides an example calculation for percentage yield where 1.72g of ammonia is obtained from a reaction using 7.5g of nitrogen. The percentage yield is calculated to be 18.9%. An example is also given for calculating percentage purity of an impure iron pyrite sample where the purity is found to be 86.3%. Formulae for percentage yield and percentage purity are defined. Key steps like using stoichiometry and unit conversions are highlighted.
14 the mole!!!

The document discusses the mole concept in chemistry. It defines the mole as 6.022x10^23 particles, which is known as Avogadro's number. The mole can refer to individual atoms, molecules, ions or other particles. The document provides examples of how to calculate the number of particles or moles of a substance using the formula N=n×NA. It also discusses molar mass and how to calculate the mass of a substance using the formula m=n×M.
01 significant digits

This document discusses the rules for determining significant digits in measurements and calculations. It defines four main rules:
1. All non-zero digits are significant, as are zeros between non-zero digits.
2. Leading zeros are not significant.
3. Trailing zeros may or may not be significant, depending on whether a decimal is present.
4. In calculations, the answer should match the least number of significant digits in the input values.
It provides examples like 0.08006 having 4 significant digits, and 1000mL having 1 significant digit. The document also covers rounding rules and calculating volumes and perimeters based on measurements to the correct number of significant digits.
13 nuclear reactions

This document discusses isotopes and nuclear reactions. It defines isotopes as atoms of the same element with different numbers of neutrons. It also describes how atomic mass is calculated based on isotope abundances. The document then discusses four types of nuclear reactions: fusion, fission, alpha decay, and beta decay. It provides examples of writing balanced nuclear equations and calculating half-life. Artificial transmutation and uses of nuclear technology like reactors and weapons are also summarized.
13 isotopes

Isotopes are atoms of the same element that have different numbers of neutrons. Atoms of the same element can have different mass numbers depending on their number of neutrons. The atomic mass listed on the periodic table is an average that takes into account the relative abundances of each isotope of that element. Atomic mass calculations involve multiplying the mass and abundance percentage of each isotope and adding them together.
12 types of chemical reactions

This document defines and provides examples of different types of chemical reactions including synthesis, decomposition, single displacement, double displacement, neutralization, and combustion reactions. Key aspects that determine the reaction type are whether reactants are elements, compounds, or if oxygen is involved. The position of elements in the periodic table also provides clues about reactivity in displacement reactions.
More from mrtangextrahelp (20)
Tang 02 alcohols, aldehydes, ketones 2
- 1. ALCOHOLS, ALDEHYDES, & KETONES
- 2. ALCOHOLS, ALDEHYDES, & KETONES
- 3. ALCOHOLS Alcohols (-OH) IUPAC naming system A C-chain containing an –OH (alcohol / hydroxyl) functional group will have an –ol ending that replaces the “e” of an –ane, – ene or –yne ending. CH3OH methanol CH3CH2OH ethanol CH3CH2CH2OH propan-1-ol
- 4. ALCOHOLS Example #1 a) CH3CHOHCH3 propan-2-ol or isopropanol b) CH2=CHCH2CH2OH but-3-en-1-ol CH3 c) 4-methylcyclohexane-1,2-diol HO OH Note that the e is reinstated when there are multiple alcohol groups
- 5. ALCOHOLS Example #2 Draw the following molecules: a) 3-ethylhexan-1-ol OH b) 2,3,3-trimethylpentan-1-ol OH OH c) 3-ethyl-2,4-dimethyl-5- propyloct-6-yne-1,2,5-triol OH OH
- 6. ALCOHOLS Example #2 d) Hepta-1,5-dien-3-ol OH e) cyclopentane-1,2,3-triol OH OH OH
- 7. ALCOHOLS Alcohol Nomenclature Common naming system Although the IUPAC system is preferred, many compounds are still referred by their “common” names. methanol methyl alcohol ethanol ethyl alcohol isopropanol isopropyl alcohol
- 8. ALCOHOLS Properties of Alcohols Alcohols typically have higher boiling points than their hydrocarbon counterparts. Why? Alcohols have hydroxyl groups (OH) which can form hydrogen bonds with one another. Hydrogen bonds are stronger than the London dispersion forces that form between their hydrocarbon counterparts. Thus more energy is required to break these stronger intermolecular bonds. The shorter the carbon chain of an alcohol, the more water soluble the compound. Why? Water bonds to other water molecules via hydrogen bonds. They would preferentially form hydrogen bonds with each other over weaker Van der Waals forces with long carbon chains.
- 9. ALCOHOLS Primary, secondary, and tertiary alcohols OH OH HO primary alcohol secondary alcohol tertiary alcohol
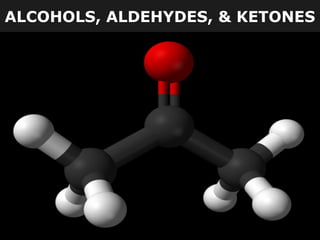
- 11. ALDEHYDES Aldehydes and Ketones Both of these groups contain a C=O (carbonyl group). The location of this group determines whether a compound is an aldehyde or a ketone. aldehyde ketone
- 12. ALDEHYDES Aldehyde Nomenclature The aldehyde group must be included in the main C-chain to be named and labeled as carbon #1. IUPAC naming system The “e” of –ane, –ene, or –yne will be replaced with an “–al” ending.
- 13. ALDEHYDES Other Nomenclature Prefixes # of IUPAC Common carbons System 1 meth- form- 2 eth- acet- 3 prop- proprion- 4 but- butyr-
- 14. ALDEHYDES Aldehyde Nomenclature Common naming system methanal formaldehyde ethanal acetaldehyde propanal proprionaldehyde butanal butyraldehyde
- 15. ALDEHYDES Example #3 Redraw and name the following compound: CH2=CHCH2CHO O H but-3-enal
- 16. ALDEHYDES Example #4 Draw and name all aldehydes with the formula C5H10O. O O H H pentanal 3-methylbutanal O O H H 2-methylbutanal 2,2-dimethylpropanal
- 17. KETONES Ketone Nomenclature A compound is a ketone when the carbonyl group is not found on the end of a carbon chain. When naming, the main chain must contain this group. IUPAC naming system The “e” of the –ane, –ene or –yne ending will be replaced with an “–one” ending.
- 18. KETONES Example #5 Redraw and name the following compound: (CH3)2CHCH2COC(CH3)3 O 2,2,5-trimethylhexan-3-one
- 19. KETONES Example #6 Draw the following compounds: O a) 2-methylcyclohexane-1,4-dione O b) 4-ethyl-2-methylcyclopentanone O
- 20. KETONES Example #6 c) all ketones with the formula C5H10O O O pentan-2-one pentan-3-one O 3-methylbutan-2-one
- 21. KETONES Example #7 a) What is the chemical structure of the smallest aldehyde? O H H b) What is the chemical structure of the smallest ketone? O
- 22. KETONES Properties of Aldehydes and Ketones Aldehydes and ketones have lower boiling points than their corresponding alcohols. Why? They do not have OH groups so they do not participate in hydrogen-bonding. They can form weaker dipole-dipole interactions. How do these compounds compare in water solubility compared to alcohols and to hydrocarbon chains? They are less soluble because they do not form hydrogen bonds with water.