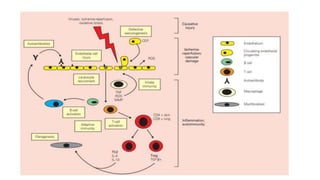
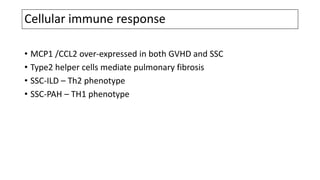
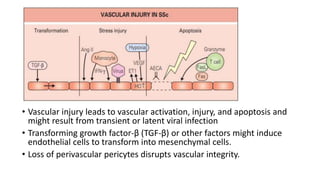
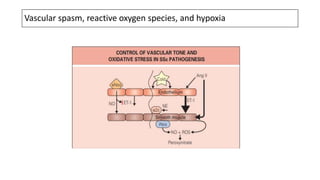
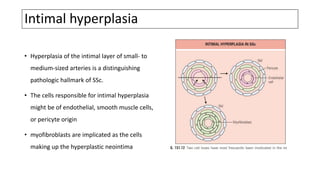
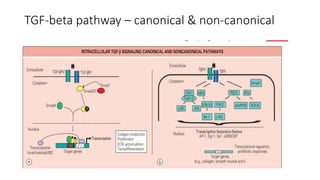
This document discusses the pathogenesis of scleroderma (SSc). It involves vascular damage, autoimmunity, and fibrosis. Innate and adaptive immunity play roles, as seen by autoantibodies, genetic polymorphisms in immune genes, and immune cell infiltration of affected tissues. Innate immune cells like macrophages and dendritic cells secrete profibrotic factors and activate pathways like TLR and inflammasome signaling that promote fibrosis. Oxidative stress, ER stress, and other cellular pathways are also involved. Various autoantibodies are implicated in tissue damage. Both humoral and cellular immune responses contribute to vascular injury and fibrosis. Myofibroblasts and TGF-beta signaling are key drivers of excessive collagen deposition.