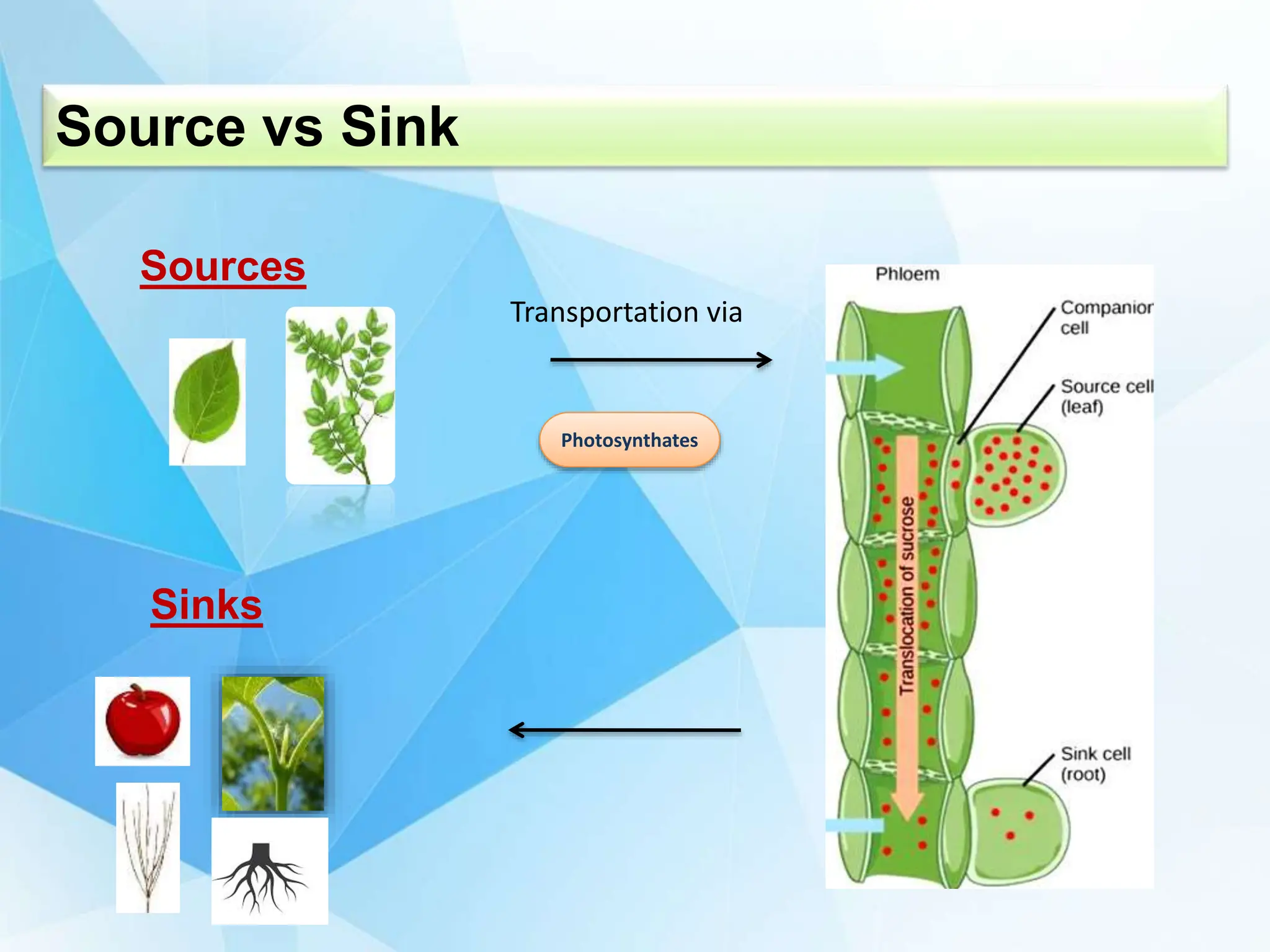
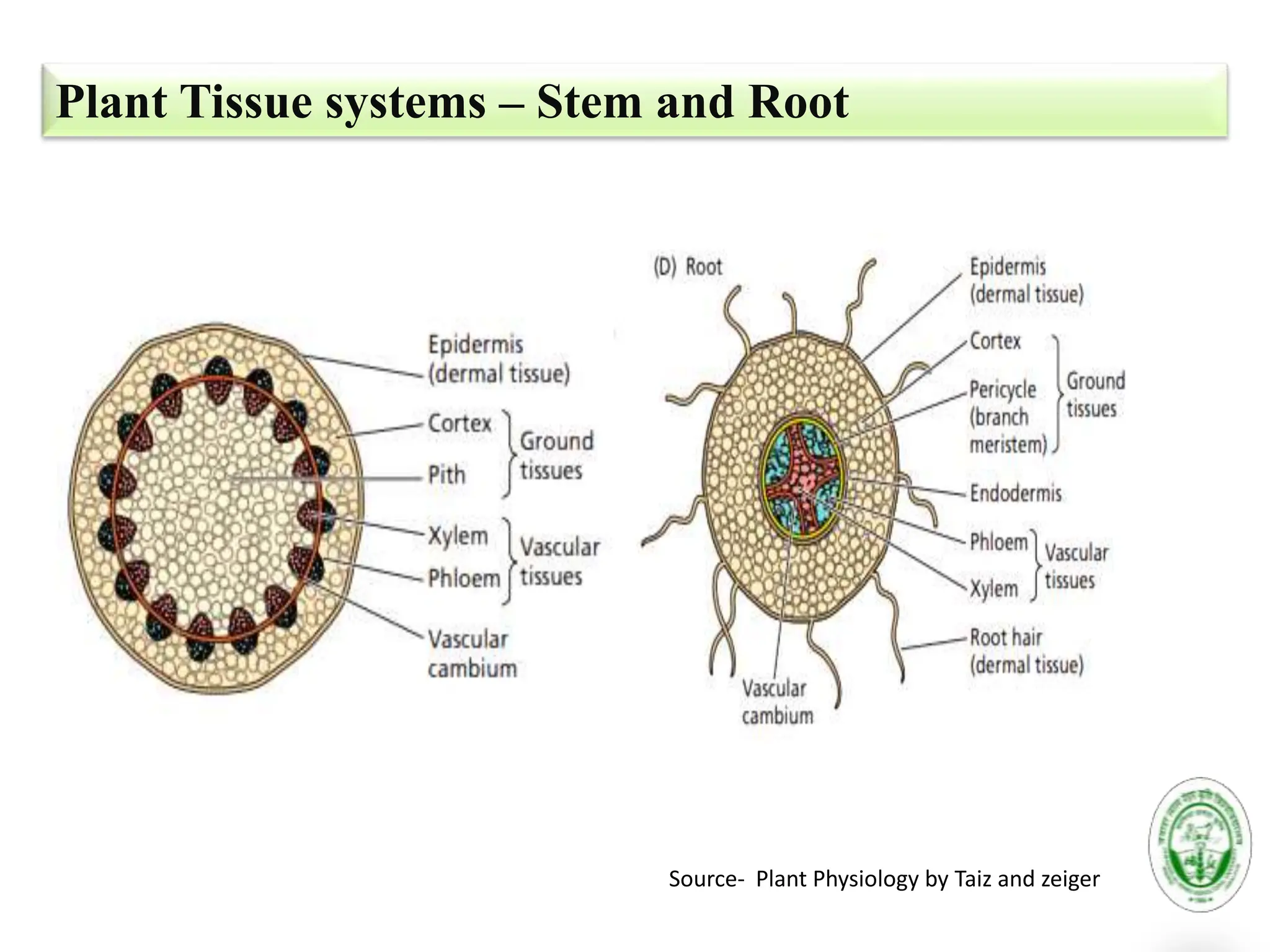
The document discusses the critical role of source-sink interactions in crop productivity, emphasizing the yield gap between actual and potential production. It highlights factors influencing source strength, sink strength, and the mechanisms of phloem transport, along with strategies to enhance yield through genetic engineering and better nutrient supply. The conclusion suggests that understanding these interactions is essential for improving agricultural output and simulations of source-sink dynamics.