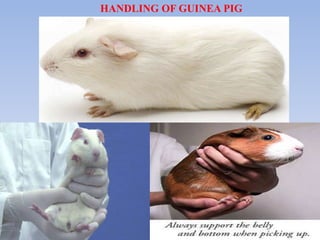
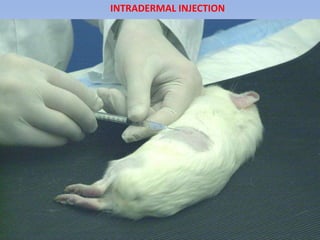
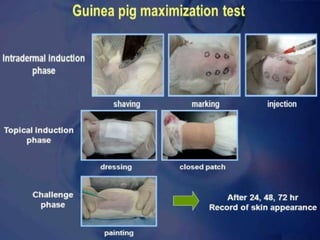
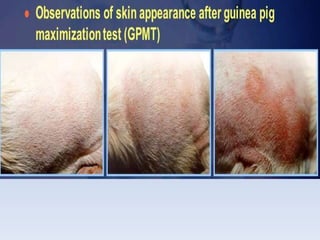
The document outlines guidelines for the handling and testing of guinea pigs for skin sensitization using OECD tests, specifically the guinea pig maximization test (GPMT) and the Buehler test. It details proper restraining techniques, injection procedures, housing and feeding conditions, and the methodology for conducting both induction and challenge phases of the tests. Additionally, it emphasizes the importance of using appropriate concentrations of test substances and monitoring reactions to ensure valid and reliable results.