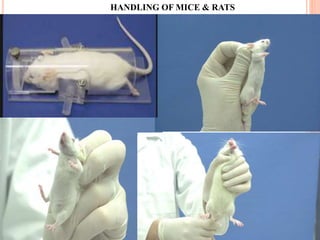
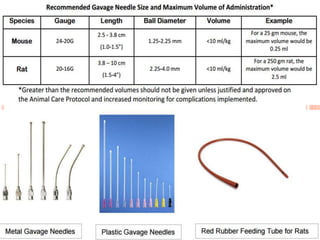
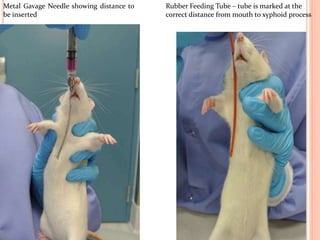
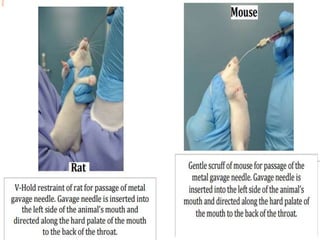
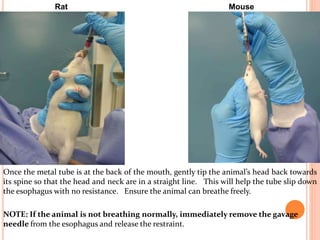
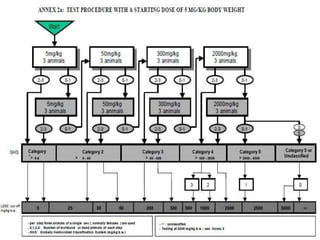
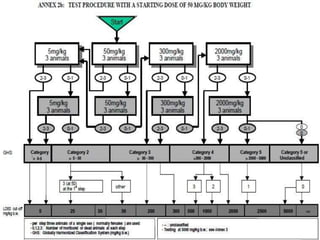
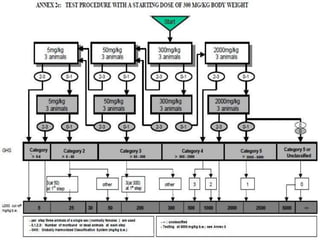
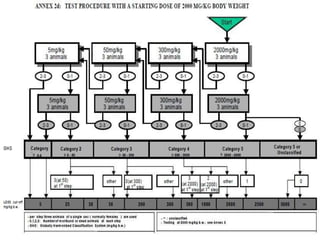
The document outlines the OECD/OCDE guideline 423 for acute oral toxicity testing which describes the proper handling and restraint of test animals, primarily rodents, during the procedure. It details methods for administering substances via gavage, the importance of humane treatment, and observations required during the testing process to evaluate acute toxicity. Key principles include the stepwise dosing method using a minimum number of animals, monitoring for signs of distress, and ensuring humane endpoints are observed throughout the testing.