Short and simple tricks for assign r and s configuration
•Download as PPTX, PDF•
0 likes•188 views
Assigning R and S configuration always challenging. we are providing a short trick to assign R and S simple configuration. In this method, you don't have to rotate carbon bond
Report
Share
Report
Share
Recommended
Unsaturated Carbonyl Compound

1) α,β-Unsaturated carbonyl compounds contain a carbonyl group and a conjugated carbon-carbon double bond separated by one carbon-carbon single bond.
2) These compounds undergo both electrophilic and nucleophilic addition reactions due to the conjugation between the carbonyl and double bond.
3) Common reactions include the Michael addition, in which a carbanion adds to the β-carbon, and the Diels-Alder reaction, where a conjugated diene adds to form a six-membered ring.
Von richter rearrangement

The document discusses the Von Richter rearrangement and Smiles rearrangement.
The Von Richter rearrangement involves the displacement of a nitro group by cyanide ion on an aromatic compound, with the carboxyl group entering ortho to the displaced nitro group. Evidence supports a mechanism where one oxygen of the carboxyl group comes from the nitro group and one from solvent.
The Smiles rearrangement involves an intramolecular nucleophilic substitution where a leaving group is displaced by a nucleophile activated by an ortho nitro group. Examples are given of substrates that undergo Smiles rearrangement where the linking chain can be aromatic or aliphatic. Electron withdrawing groups para to the nucleophile retard the
Electrophilic Substitution reactions

This document provides an overview of electrophilic substitution reactions. It defines electrophilic substitution as a reaction where a functional group is attached to a compound by replacing another functional group, often a hydrogen atom. It describes two main types: electrophilic aromatic substitution reactions, where an atom attached to an aromatic ring is replaced; and electrophilic aliphatic substitution reactions, where hydrogen in an aliphatic compound is usually replaced. The document also outlines the three step mechanism of electrophilic substitution reactions: 1) generating an electrophile, 2) forming a carbocation, and 3) eliminating a proton to restore aromaticity.
Organic reactions and mechanisms

The document discusses organic reactions and reaction mechanisms. It defines nucleophiles and electrophiles, and provides examples of each. It then summarizes several common types of organic reactions including addition reactions, substitution reactions, elimination reactions, and aromatic substitutions. The mechanisms and examples of nucleophilic addition, electrophilic addition, nucleophilic substitution, and electrophilic aromatic substitutions like nitration, sulfonation, and halogenation are described in detail.
fluorescence and phosphorescence

The document discusses fluorescence and phosphorescence. It begins by explaining the quantum nature of energy levels in molecules and electrons. It then defines fluorescence as the emission of light from excited singlet states, and describes the absorption and emission processes. Phosphorescence is defined as emission from triplet excited states, which have longer lifetimes than fluorescence. Examples are given of materials that exhibit fluorescence or phosphorescence, as well as applications in various fields such as analytical chemistry, life sciences, and displays.
Reaction intermediates

The document discusses various reactive intermediates in chemical reactions:
1. Intermediates are chemical species that are neither the starting reactants nor the final products but appear as transient intermediates in step-wise reactions.
2. Common types of reactive intermediates discussed include carbocations, carbanions, free radicals, carbenes, nitrenes, and arenynes.
3. Specific details are provided about the electronic structure and reactivity of each intermediate.
Arenium Ion Mechanism in Aromatic Electrophilic Substitution 

This document describes the arenium ion mechanism for aromatic electrophilic substitution reactions. The mechanism involves two steps: 1) the rate-determining attack of an electrophile on the aromatic ring to form a resonance-stabilized arenium ion intermediate, and 2) rapid departure of the leaving group to regenerate aromaticity. Evidence for this mechanism includes isolation of arenium ion intermediates and a lack of isotope effects. The first step is highly endothermic due to loss of aromaticity, making it rate-determining, while the second step regains aromatic stabilization in an exothermic process.
Favorskii and wolff rearrangement seminar

The document summarizes the Favorskii and Wolff rearrangements. The Favorskii rearrangement involves the base-catalyzed rearrangement of cyclopropanones and α-halo ketones, forming carboxylic acid derivatives. It proceeds through the formation of an enolate intermediate and a cyclopropanone, which is then attacked by the nucleophile. The Wolff rearrangement can occur thermally, photochemically, or via transition metal catalysis, and involves 1,2-migration of substituents with the migratory aptitude of H > aryl > alkyl. Both rearrangements form a carbonyl group at the migration origin and involve migration of an electron-rich carbon terminus.
Recommended
Unsaturated Carbonyl Compound

1) α,β-Unsaturated carbonyl compounds contain a carbonyl group and a conjugated carbon-carbon double bond separated by one carbon-carbon single bond.
2) These compounds undergo both electrophilic and nucleophilic addition reactions due to the conjugation between the carbonyl and double bond.
3) Common reactions include the Michael addition, in which a carbanion adds to the β-carbon, and the Diels-Alder reaction, where a conjugated diene adds to form a six-membered ring.
Von richter rearrangement

The document discusses the Von Richter rearrangement and Smiles rearrangement.
The Von Richter rearrangement involves the displacement of a nitro group by cyanide ion on an aromatic compound, with the carboxyl group entering ortho to the displaced nitro group. Evidence supports a mechanism where one oxygen of the carboxyl group comes from the nitro group and one from solvent.
The Smiles rearrangement involves an intramolecular nucleophilic substitution where a leaving group is displaced by a nucleophile activated by an ortho nitro group. Examples are given of substrates that undergo Smiles rearrangement where the linking chain can be aromatic or aliphatic. Electron withdrawing groups para to the nucleophile retard the
Electrophilic Substitution reactions

This document provides an overview of electrophilic substitution reactions. It defines electrophilic substitution as a reaction where a functional group is attached to a compound by replacing another functional group, often a hydrogen atom. It describes two main types: electrophilic aromatic substitution reactions, where an atom attached to an aromatic ring is replaced; and electrophilic aliphatic substitution reactions, where hydrogen in an aliphatic compound is usually replaced. The document also outlines the three step mechanism of electrophilic substitution reactions: 1) generating an electrophile, 2) forming a carbocation, and 3) eliminating a proton to restore aromaticity.
Organic reactions and mechanisms

The document discusses organic reactions and reaction mechanisms. It defines nucleophiles and electrophiles, and provides examples of each. It then summarizes several common types of organic reactions including addition reactions, substitution reactions, elimination reactions, and aromatic substitutions. The mechanisms and examples of nucleophilic addition, electrophilic addition, nucleophilic substitution, and electrophilic aromatic substitutions like nitration, sulfonation, and halogenation are described in detail.
fluorescence and phosphorescence

The document discusses fluorescence and phosphorescence. It begins by explaining the quantum nature of energy levels in molecules and electrons. It then defines fluorescence as the emission of light from excited singlet states, and describes the absorption and emission processes. Phosphorescence is defined as emission from triplet excited states, which have longer lifetimes than fluorescence. Examples are given of materials that exhibit fluorescence or phosphorescence, as well as applications in various fields such as analytical chemistry, life sciences, and displays.
Reaction intermediates

The document discusses various reactive intermediates in chemical reactions:
1. Intermediates are chemical species that are neither the starting reactants nor the final products but appear as transient intermediates in step-wise reactions.
2. Common types of reactive intermediates discussed include carbocations, carbanions, free radicals, carbenes, nitrenes, and arenynes.
3. Specific details are provided about the electronic structure and reactivity of each intermediate.
Arenium Ion Mechanism in Aromatic Electrophilic Substitution 

This document describes the arenium ion mechanism for aromatic electrophilic substitution reactions. The mechanism involves two steps: 1) the rate-determining attack of an electrophile on the aromatic ring to form a resonance-stabilized arenium ion intermediate, and 2) rapid departure of the leaving group to regenerate aromaticity. Evidence for this mechanism includes isolation of arenium ion intermediates and a lack of isotope effects. The first step is highly endothermic due to loss of aromaticity, making it rate-determining, while the second step regains aromatic stabilization in an exothermic process.
Favorskii and wolff rearrangement seminar

The document summarizes the Favorskii and Wolff rearrangements. The Favorskii rearrangement involves the base-catalyzed rearrangement of cyclopropanones and α-halo ketones, forming carboxylic acid derivatives. It proceeds through the formation of an enolate intermediate and a cyclopropanone, which is then attacked by the nucleophile. The Wolff rearrangement can occur thermally, photochemically, or via transition metal catalysis, and involves 1,2-migration of substituents with the migratory aptitude of H > aryl > alkyl. Both rearrangements form a carbonyl group at the migration origin and involve migration of an electron-rich carbon terminus.
Sn2 reaction

The document summarizes key aspects of SN2 reactions including reaction mechanism, kinetics, stereochemistry, and factors that affect the rate of the reaction. It describes the SN2 reaction as a bimolecular nucleophilic substitution where the nucleophile attacks the substrate simultaneously as the leaving group departs, resulting in an inversion of configuration. Rate depends on both the nucleophile and substrate concentrations. The stability of the transition state is affected by substrate structure, nucleophilicity, leaving group ability, solvent properties, and conjugation effects in allylic and benzylic systems. Cyclic substrates and those without available orbital overlap do not undergo SN2 reactions as easily.
Inductive effect pptx

This document discusses electronic displacement in organic compounds. It describes two types of electronic displacement: permanent displacement including inductive, resonance, and mesomeric effects; and temporary displacement through electromeric effects. Inductive effects are further broken down into +I effects where groups donate electron density and -I effects where groups withdraw electron density. Examples of inductive effects include their impact on acid/base strength, stability of carbocations/carbanions, and dipole moments.
Jablonski diagram

The document discusses the Jablonski diagram, which was developed by Alexander Jablonski to illustrate electronic transitions in molecules. It shows energy levels and the transitions between them that occur during absorption and emission of light. Absorption involves excitation of electrons to higher energy levels via straight arrows. Emission can occur through fluorescence, phosphorescence, or non-radiative pathways like internal conversion and intersystem crossing, represented by curved arrows. The diagram provides insight into the timescales of the different transition processes.
The chemoselectivity

This document discusses chemoselectivity and which functional groups react first when multiple groups are present. It also covers protecting groups used to control selectivity and different oxidizing agents for converting alcohols to aldehydes and ketones or acids, including Dess-Martin and Swern oxidations for primary alcohols and chromium and manganese agents for secondary alcohols. Lastly, it prompts the reader to think about selectivity considerations.
Nanosecond photolysis

Photolysis is a chemical process where molecules are broken down by light absorption. Flash photolysis is commonly used to study short-lived intermediates in photochemical reactions, employing a photolysis flash to initiate reactions followed by a monitoring flash to measure absorption spectra. To study processes in the nanosecond time range, lasers can be used to generate photolysis pulses less than 20 nanoseconds, allowing observation of excited singlet state lifetimes and other fast reactions. Laser flash photolysis systems employ a laser pulse to synchronize a photolysis spark and provide pulses to initiate reactions and monitor absorption on nanosecond timescales, enabling identification of transient intermediates and insight into fast reaction mechanisms.
Neucleophilic aromatic substitution

Nucleophilic aromatic substitution reactions follow an addition-elimination mechanism known as SNAr. The rate-determining step is the formation of a cyclohexadienyl anion intermediate through nucleophilic attack. Electron-withdrawing groups stabilize this intermediate through resonance, making the reaction faster. Nucleophilic aromatic substitution is most favorable when the leaving group is fluoride and least with iodide, and occurs readily with strong nucleophiles like hydroxide or cyanide in the presence of electron-withdrawing groups ortho or para to the reaction site.
13. SeO2 & raney ni

Selenium dioxide (SeO2) and Raney nickel are both useful reagents in organic synthesis. SeO2 can be used to oxidize alkenes to allylic alcohols or carbonyls. It also oxidizes carbonyls to 1,2-dicarbonyls and internal alkynes to 1,2-dicarbonyls. Raney nickel catalyzes hydrogenation of aromatics and reduction of carbonyl groups by cleaving C-S bonds. Both reagents have applications in functional group transformations.
Diels alder reaction

Diels alder reaction is activated by photochemical reaction.it plays a vital role in drug discovery and synthesis.
Nucleophilic substitution reactions 

Nucleophilic substitution reactions in organicchemistry, sterochemistry and factors affecting it. SN1, Sn2, Sni.
Aromatic elctrophilic and nucleophilic substitution reaction

Aromatic elctrophilic and nucleophilic substitution reactionDr.Mohd Kamil Hussain, Department of Chemistry, Govt Raza PG College Rampur
Benzene has 6π electrons delocalized in 6p orbitals that overlap above and below the plane of the ring. Because benzene’s six pie electrons satisfy Huckel’s rule, benzene is especially stable. Reaction that keep the aromatic ring intact are therefore favoured
Carbanions

Carbanions are carbon atoms with a negative charge that are formed through various mechanisms. They can be classified based on their formation method such as through heterocyclic cleavage, proton abstraction using a base, decarboxylation, addition of a nucleophile to an alkene, or formation of an organometallic compound. Carbanion stability depends on factors like the electronegativity of the carbon, inductive effects, resonance effects, and attachment to sulfur or phosphorus. Aromatic carbanions and those with electron-withdrawing groups are particularly stable due to resonance delocalization. Carbanions have applications in reactions like the Perkin reaction, Claisen condensation, benzoin condensation,
Addition reaction

The document discusses addition reactions, which involve all atoms from reactants being added to unsaturated compounds to form products. There are three main types: electrophilic addition, nucleophilic addition, and free radical addition. Electrophilic addition involves an electrophile attacking the π bond. Nucleophilic addition involves a nucleophile attacking carbocations. Free radical addition occurs through radical intermediates formed from reactants. Addition reactions can result in stereospecific or non-stereospecific products depending on the mechanism and reactants involved.
Acid Base Hydrolysis in Octahedral Complexes

This document discusses acid and base hydrolysis in octahedral complexes. It covers factors that affect the rate of acid hydrolysis, including the charge on the complex, steric hindrance effects, and the strength of the leaving group. A higher positive charge, more steric hindrance, or stronger metal-leaving group bond each decrease the rate of acid hydrolysis according to first-order kinetics through a dissociative SN1 mechanism. Base hydrolysis of octahedral complexes can proceed by either associative SN2 or dissociative SN1 pathways depending on conditions.
ORGANIC INTERMEDIATES- CARBANIONS

The slides contains the structural features, formation methods, stability and application of carbanion produces in organic reaction.
Dienone phenol rearragment

The document summarizes the dienone-phenol rearrangement, which is the acid- or base-catalyzed migration of alkyl groups in cyclohexadienones, resulting in highly substituted phenols. It was first described in 1893 for the rearrangement of santonin to desmotroposantonin under acidic conditions, but was more fully characterized in 1930. The rearrangement requires only moderately strong acids and is exothermic. It proceeds by a [1,3] sigmatropic migration of C-C bonds, which actually occurs through two subsequent [1,2] alkyl shifts. Depending on the migrating group, other rearrangements such as [1,2], [1,3], [
Overtone & fermi resonance in IR

This document discusses overtones and Fermi resonance in infrared spectroscopy. It defines overtones as absorptions that occur at integral multiples of the fundamental frequency, such as a band at 1000 cm-1 accompanying a fundamental at 500 cm-1. Fermi resonance occurs when a fundamental and overtone band have similar energies, causing them to interact and shift in intensity and frequency. This can result in a "Fermi doublet" with one band increasing while the other decreases in energy. The document provides examples of overtones and Fermi resonance in infrared spectra.
Hypervalent organo iodines reagents in organic synthesis

Hypervalent organo iodines reagents in organic synthesisMinal Saini , student of Chaudhary bansilal University , Bhiwani, Haryana
Hypervalent refers to the main group elements that breaks the octet rule and firmly has more than right electrons in it's valence shell. These are non - metallic oxidation reagents. Kinetics of solution in reaction

Definition - Mechanism - Effect of dielectric constant on the rate of reactions in solutions - Salt effect - Primary salt effect - Bronsted – Bjerrum equation - Secondary salt effect - Effect of pressure on rate of reaction in solution - Volume of activation - Significance
Dicyclohexylcarbodiimide

DicyclohexylcarbodiimideDepartment of Pharmaceutical Science and Natural Products, central University of Punjab
Dicyclohexylcarbodiimide (DCC) is an organic compound that is commonly used as a synthetic reagent to couple amino acids during peptide synthesis. It was first introduced for this purpose in 1955. DCC is a waxy white solid with a sweet odor that is highly soluble in organic solvents but insoluble in water. It has a low melting point, which allows it to be easily melted and handled. DCC is commonly used to catalyze esterification reactions and form amide bonds, as well as synthesize peptides, ethers, acid anhydrides, and lactones. One of its key applications is in the synthesis of beta-lactam rings in penicillin.Gattermann koch

The Gattermann-Koch reaction is a chemical reaction discovered in 1897 by German chemists Ludwig Gattermann and Julius Arnold Koch. It involves the formylation (addition of an aldehyde group) of aromatic compounds using a mixture of carbon monoxide, hydrogen chloride, and anhydrous aluminum chloride catalyst. The unstable formyl chloride intermediate reacts to add the formyl group to the aromatic ring, producing an aromatic aldehyde such as benzaldehyde from benzene.
Important name reaction for csir net and gate chemistry

This document summarizes 12 important organic reactions: 1) Appel Reaction, 2) Baylis-Hillman reaction, 3) Boekelheide reaction, 4) Buchwald–Hartwig Coupling, 5) Chugaev elimination, 6) Core-Chaykovsky reaction, 7) Mitsunobu reaction, 8) Prévost trans and Woodward cis-dihydroxylation reaction, 9) Shapiro reaction, 10) Sharpless asymmetric epoxidation, 11) Corey–Bakshi–Shibata (CBS) reduction, and 12) Kulinkovich reaction. For each reaction, it provides a brief description and representative examples of the reaction mechanism.
Important name reaction for csir net and gate chemistry

Appel Reaction, Baylis–Hillman reaction, Boekelheide reaction, Buchwald–Hartwig Coupling, Chugaev elimination, Core-Chaykovsky reaction, Mitsunobu reaction, Shapiro reaction, Sharpless asymmetric epoxidation, Corey–Bakshi–Shibata (CBS) reduction, Kulinkovich reaction
More Related Content
What's hot
Sn2 reaction

The document summarizes key aspects of SN2 reactions including reaction mechanism, kinetics, stereochemistry, and factors that affect the rate of the reaction. It describes the SN2 reaction as a bimolecular nucleophilic substitution where the nucleophile attacks the substrate simultaneously as the leaving group departs, resulting in an inversion of configuration. Rate depends on both the nucleophile and substrate concentrations. The stability of the transition state is affected by substrate structure, nucleophilicity, leaving group ability, solvent properties, and conjugation effects in allylic and benzylic systems. Cyclic substrates and those without available orbital overlap do not undergo SN2 reactions as easily.
Inductive effect pptx

This document discusses electronic displacement in organic compounds. It describes two types of electronic displacement: permanent displacement including inductive, resonance, and mesomeric effects; and temporary displacement through electromeric effects. Inductive effects are further broken down into +I effects where groups donate electron density and -I effects where groups withdraw electron density. Examples of inductive effects include their impact on acid/base strength, stability of carbocations/carbanions, and dipole moments.
Jablonski diagram

The document discusses the Jablonski diagram, which was developed by Alexander Jablonski to illustrate electronic transitions in molecules. It shows energy levels and the transitions between them that occur during absorption and emission of light. Absorption involves excitation of electrons to higher energy levels via straight arrows. Emission can occur through fluorescence, phosphorescence, or non-radiative pathways like internal conversion and intersystem crossing, represented by curved arrows. The diagram provides insight into the timescales of the different transition processes.
The chemoselectivity

This document discusses chemoselectivity and which functional groups react first when multiple groups are present. It also covers protecting groups used to control selectivity and different oxidizing agents for converting alcohols to aldehydes and ketones or acids, including Dess-Martin and Swern oxidations for primary alcohols and chromium and manganese agents for secondary alcohols. Lastly, it prompts the reader to think about selectivity considerations.
Nanosecond photolysis

Photolysis is a chemical process where molecules are broken down by light absorption. Flash photolysis is commonly used to study short-lived intermediates in photochemical reactions, employing a photolysis flash to initiate reactions followed by a monitoring flash to measure absorption spectra. To study processes in the nanosecond time range, lasers can be used to generate photolysis pulses less than 20 nanoseconds, allowing observation of excited singlet state lifetimes and other fast reactions. Laser flash photolysis systems employ a laser pulse to synchronize a photolysis spark and provide pulses to initiate reactions and monitor absorption on nanosecond timescales, enabling identification of transient intermediates and insight into fast reaction mechanisms.
Neucleophilic aromatic substitution

Nucleophilic aromatic substitution reactions follow an addition-elimination mechanism known as SNAr. The rate-determining step is the formation of a cyclohexadienyl anion intermediate through nucleophilic attack. Electron-withdrawing groups stabilize this intermediate through resonance, making the reaction faster. Nucleophilic aromatic substitution is most favorable when the leaving group is fluoride and least with iodide, and occurs readily with strong nucleophiles like hydroxide or cyanide in the presence of electron-withdrawing groups ortho or para to the reaction site.
13. SeO2 & raney ni

Selenium dioxide (SeO2) and Raney nickel are both useful reagents in organic synthesis. SeO2 can be used to oxidize alkenes to allylic alcohols or carbonyls. It also oxidizes carbonyls to 1,2-dicarbonyls and internal alkynes to 1,2-dicarbonyls. Raney nickel catalyzes hydrogenation of aromatics and reduction of carbonyl groups by cleaving C-S bonds. Both reagents have applications in functional group transformations.
Diels alder reaction

Diels alder reaction is activated by photochemical reaction.it plays a vital role in drug discovery and synthesis.
Nucleophilic substitution reactions 

Nucleophilic substitution reactions in organicchemistry, sterochemistry and factors affecting it. SN1, Sn2, Sni.
Aromatic elctrophilic and nucleophilic substitution reaction

Aromatic elctrophilic and nucleophilic substitution reactionDr.Mohd Kamil Hussain, Department of Chemistry, Govt Raza PG College Rampur
Benzene has 6π electrons delocalized in 6p orbitals that overlap above and below the plane of the ring. Because benzene’s six pie electrons satisfy Huckel’s rule, benzene is especially stable. Reaction that keep the aromatic ring intact are therefore favoured
Carbanions

Carbanions are carbon atoms with a negative charge that are formed through various mechanisms. They can be classified based on their formation method such as through heterocyclic cleavage, proton abstraction using a base, decarboxylation, addition of a nucleophile to an alkene, or formation of an organometallic compound. Carbanion stability depends on factors like the electronegativity of the carbon, inductive effects, resonance effects, and attachment to sulfur or phosphorus. Aromatic carbanions and those with electron-withdrawing groups are particularly stable due to resonance delocalization. Carbanions have applications in reactions like the Perkin reaction, Claisen condensation, benzoin condensation,
Addition reaction

The document discusses addition reactions, which involve all atoms from reactants being added to unsaturated compounds to form products. There are three main types: electrophilic addition, nucleophilic addition, and free radical addition. Electrophilic addition involves an electrophile attacking the π bond. Nucleophilic addition involves a nucleophile attacking carbocations. Free radical addition occurs through radical intermediates formed from reactants. Addition reactions can result in stereospecific or non-stereospecific products depending on the mechanism and reactants involved.
Acid Base Hydrolysis in Octahedral Complexes

This document discusses acid and base hydrolysis in octahedral complexes. It covers factors that affect the rate of acid hydrolysis, including the charge on the complex, steric hindrance effects, and the strength of the leaving group. A higher positive charge, more steric hindrance, or stronger metal-leaving group bond each decrease the rate of acid hydrolysis according to first-order kinetics through a dissociative SN1 mechanism. Base hydrolysis of octahedral complexes can proceed by either associative SN2 or dissociative SN1 pathways depending on conditions.
ORGANIC INTERMEDIATES- CARBANIONS

The slides contains the structural features, formation methods, stability and application of carbanion produces in organic reaction.
Dienone phenol rearragment

The document summarizes the dienone-phenol rearrangement, which is the acid- or base-catalyzed migration of alkyl groups in cyclohexadienones, resulting in highly substituted phenols. It was first described in 1893 for the rearrangement of santonin to desmotroposantonin under acidic conditions, but was more fully characterized in 1930. The rearrangement requires only moderately strong acids and is exothermic. It proceeds by a [1,3] sigmatropic migration of C-C bonds, which actually occurs through two subsequent [1,2] alkyl shifts. Depending on the migrating group, other rearrangements such as [1,2], [1,3], [
Overtone & fermi resonance in IR

This document discusses overtones and Fermi resonance in infrared spectroscopy. It defines overtones as absorptions that occur at integral multiples of the fundamental frequency, such as a band at 1000 cm-1 accompanying a fundamental at 500 cm-1. Fermi resonance occurs when a fundamental and overtone band have similar energies, causing them to interact and shift in intensity and frequency. This can result in a "Fermi doublet" with one band increasing while the other decreases in energy. The document provides examples of overtones and Fermi resonance in infrared spectra.
Hypervalent organo iodines reagents in organic synthesis

Hypervalent organo iodines reagents in organic synthesisMinal Saini , student of Chaudhary bansilal University , Bhiwani, Haryana
Hypervalent refers to the main group elements that breaks the octet rule and firmly has more than right electrons in it's valence shell. These are non - metallic oxidation reagents. Kinetics of solution in reaction

Definition - Mechanism - Effect of dielectric constant on the rate of reactions in solutions - Salt effect - Primary salt effect - Bronsted – Bjerrum equation - Secondary salt effect - Effect of pressure on rate of reaction in solution - Volume of activation - Significance
Dicyclohexylcarbodiimide

DicyclohexylcarbodiimideDepartment of Pharmaceutical Science and Natural Products, central University of Punjab
Dicyclohexylcarbodiimide (DCC) is an organic compound that is commonly used as a synthetic reagent to couple amino acids during peptide synthesis. It was first introduced for this purpose in 1955. DCC is a waxy white solid with a sweet odor that is highly soluble in organic solvents but insoluble in water. It has a low melting point, which allows it to be easily melted and handled. DCC is commonly used to catalyze esterification reactions and form amide bonds, as well as synthesize peptides, ethers, acid anhydrides, and lactones. One of its key applications is in the synthesis of beta-lactam rings in penicillin.Gattermann koch

The Gattermann-Koch reaction is a chemical reaction discovered in 1897 by German chemists Ludwig Gattermann and Julius Arnold Koch. It involves the formylation (addition of an aldehyde group) of aromatic compounds using a mixture of carbon monoxide, hydrogen chloride, and anhydrous aluminum chloride catalyst. The unstable formyl chloride intermediate reacts to add the formyl group to the aromatic ring, producing an aromatic aldehyde such as benzaldehyde from benzene.
What's hot (20)
Aromatic elctrophilic and nucleophilic substitution reaction

Aromatic elctrophilic and nucleophilic substitution reaction
Hypervalent organo iodines reagents in organic synthesis

Hypervalent organo iodines reagents in organic synthesis
More from ScifySolution
Important name reaction for csir net and gate chemistry

This document summarizes 12 important organic reactions: 1) Appel Reaction, 2) Baylis-Hillman reaction, 3) Boekelheide reaction, 4) Buchwald–Hartwig Coupling, 5) Chugaev elimination, 6) Core-Chaykovsky reaction, 7) Mitsunobu reaction, 8) Prévost trans and Woodward cis-dihydroxylation reaction, 9) Shapiro reaction, 10) Sharpless asymmetric epoxidation, 11) Corey–Bakshi–Shibata (CBS) reduction, and 12) Kulinkovich reaction. For each reaction, it provides a brief description and representative examples of the reaction mechanism.
Important name reaction for csir net and gate chemistry

Appel Reaction, Baylis–Hillman reaction, Boekelheide reaction, Buchwald–Hartwig Coupling, Chugaev elimination, Core-Chaykovsky reaction, Mitsunobu reaction, Shapiro reaction, Sharpless asymmetric epoxidation, Corey–Bakshi–Shibata (CBS) reduction, Kulinkovich reaction
Protection and deprotection of functional groups and it application in organi...

Protection of amine, acid, alcohol, ketone, aldehyde important for organic synthesis here we are providing complete study notes on it.
visit scifysolution.com for more notes
Oxidation of carbonyl compounds 

This document provides an overview of 10 common chemical oxidation reactions that can convert alcohols to aldehydes and ketones. It describes the reagents, reaction mechanisms, advantages/disadvantages, and examples for each reaction, including Jones oxidation, PCC oxidation, Swern oxidation, Dess-Martin periodinane oxidation, MnO2 oxidation, Babler oxidation, Corey-Kim oxidation, Parikh-Doering oxidation, Fetizon oxidation, and Oppenauer oxidation.
Reduction by dissolving metals

Chemical methods of reduction can take place by addition of electrons to the unsaturated compound followed by transfer of protons or can take place by addition of hydride ion followed by protonation.
Reductions that follow the first path are generally effected by metal, the source of the electrons, and a proton donor, which may be water, an alcohol or an acid. However, in the absence of proton source, it can undergo dimerization or polymerization.
Reduction using catalytic hydrogenation

Catalytic hydrogenation is one of the most convenient available for the reduction of organic compounds. compounds. The reduction is carried out easily by stirring or shaking the substrate with the catalyst in a suitable solvent
More from ScifySolution (6)
Important name reaction for csir net and gate chemistry

Important name reaction for csir net and gate chemistry
Important name reaction for csir net and gate chemistry

Important name reaction for csir net and gate chemistry
Protection and deprotection of functional groups and it application in organi...

Protection and deprotection of functional groups and it application in organi...
Recently uploaded
Beyond Degrees - Empowering the Workforce in the Context of Skills-First.pptx

Iván Bornacelly, Policy Analyst at the OECD Centre for Skills, OECD, presents at the webinar 'Tackling job market gaps with a skills-first approach' on 12 June 2024
What is Digital Literacy? A guest blog from Andy McLaughlin, University of Ab...

What is Digital Literacy? A guest blog from Andy McLaughlin, University of Aberdeen
ANATOMY AND BIOMECHANICS OF HIP JOINT.pdf

it describes the bony anatomy including the femoral head , acetabulum, labrum . also discusses the capsule , ligaments . muscle that act on the hip joint and the range of motion are outlined. factors affecting hip joint stability and weight transmission through the joint are summarized.
How to deliver Powerpoint Presentations.pptx

"How to make and deliver dynamic presentations by making it more interactive to captivate your audience attention"
Hindi varnamala | hindi alphabet PPT.pdf

हिंदी वर्णमाला पीपीटी, hindi alphabet PPT presentation, hindi varnamala PPT, Hindi Varnamala pdf, हिंदी स्वर, हिंदी व्यंजन, sikhiye hindi varnmala, dr. mulla adam ali, hindi language and literature, hindi alphabet with drawing, hindi alphabet pdf, hindi varnamala for childrens, hindi language, hindi varnamala practice for kids, https://www.drmullaadamali.com
Advanced Java[Extra Concepts, Not Difficult].docx![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
This is part 2 of my Java Learning Journey. This contains Hashing, ArrayList, LinkedList, Date and Time Classes, Calendar Class and more.
Wound healing PPT

This document provides an overview of wound healing, its functions, stages, mechanisms, factors affecting it, and complications.
A wound is a break in the integrity of the skin or tissues, which may be associated with disruption of the structure and function.
Healing is the body’s response to injury in an attempt to restore normal structure and functions.
Healing can occur in two ways: Regeneration and Repair
There are 4 phases of wound healing: hemostasis, inflammation, proliferation, and remodeling. This document also describes the mechanism of wound healing. Factors that affect healing include infection, uncontrolled diabetes, poor nutrition, age, anemia, the presence of foreign bodies, etc.
Complications of wound healing like infection, hyperpigmentation of scar, contractures, and keloid formation.
The History of Stoke Newington Street Names

Presented at the Stoke Newington Literary Festival on 9th June 2024
www.StokeNewingtonHistory.com
Constructing Your Course Container for Effective Communication

Communicating effectively and consistently with students can help them feel at ease during their learning experience and provide the instructor with a communication trail to track the course's progress. This workshop will take you through constructing an engaging course container to facilitate effective communication.
Traditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh - RAYH...

Traditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh
Gender and Mental Health - Counselling and Family Therapy Applications and In...

A proprietary approach developed by bringing together the best of learning theories from Psychology, design principles from the world of visualization, and pedagogical methods from over a decade of training experience, that enables you to: Learn better, faster!
BÀI TẬP DẠY THÊM TIẾNG ANH LỚP 7 CẢ NĂM FRIENDS PLUS SÁCH CHÂN TRỜI SÁNG TẠO ...

BÀI TẬP DẠY THÊM TIẾNG ANH LỚP 7 CẢ NĂM FRIENDS PLUS SÁCH CHÂN TRỜI SÁNG TẠO ...Nguyen Thanh Tu Collection
https://app.box.com/s/qhtvq32h4ybf9t49ku85x0n3xl4jhr15Philippine Edukasyong Pantahanan at Pangkabuhayan (EPP) Curriculum

(𝐓𝐋𝐄 𝟏𝟎𝟎) (𝐋𝐞𝐬𝐬𝐨𝐧 𝟏)-𝐏𝐫𝐞𝐥𝐢𝐦𝐬
𝐃𝐢𝐬𝐜𝐮𝐬𝐬 𝐭𝐡𝐞 𝐄𝐏𝐏 𝐂𝐮𝐫𝐫𝐢𝐜𝐮𝐥𝐮𝐦 𝐢𝐧 𝐭𝐡𝐞 𝐏𝐡𝐢𝐥𝐢𝐩𝐩𝐢𝐧𝐞𝐬:
- Understand the goals and objectives of the Edukasyong Pantahanan at Pangkabuhayan (EPP) curriculum, recognizing its importance in fostering practical life skills and values among students. Students will also be able to identify the key components and subjects covered, such as agriculture, home economics, industrial arts, and information and communication technology.
𝐄𝐱𝐩𝐥𝐚𝐢𝐧 𝐭𝐡𝐞 𝐍𝐚𝐭𝐮𝐫𝐞 𝐚𝐧𝐝 𝐒𝐜𝐨𝐩𝐞 𝐨𝐟 𝐚𝐧 𝐄𝐧𝐭𝐫𝐞𝐩𝐫𝐞𝐧𝐞𝐮𝐫:
-Define entrepreneurship, distinguishing it from general business activities by emphasizing its focus on innovation, risk-taking, and value creation. Students will describe the characteristics and traits of successful entrepreneurs, including their roles and responsibilities, and discuss the broader economic and social impacts of entrepreneurial activities on both local and global scales.
How to Setup Warehouse & Location in Odoo 17 Inventory

In this slide, we'll explore how to set up warehouses and locations in Odoo 17 Inventory. This will help us manage our stock effectively, track inventory levels, and streamline warehouse operations.
Recently uploaded (20)
Beyond Degrees - Empowering the Workforce in the Context of Skills-First.pptx

Beyond Degrees - Empowering the Workforce in the Context of Skills-First.pptx
What is Digital Literacy? A guest blog from Andy McLaughlin, University of Ab...

What is Digital Literacy? A guest blog from Andy McLaughlin, University of Ab...
B. Ed Syllabus for babasaheb ambedkar education university.pdf

B. Ed Syllabus for babasaheb ambedkar education university.pdf
Constructing Your Course Container for Effective Communication

Constructing Your Course Container for Effective Communication
Traditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh - RAYH...

Traditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh - RAYH...
Gender and Mental Health - Counselling and Family Therapy Applications and In...

Gender and Mental Health - Counselling and Family Therapy Applications and In...
NEWSPAPERS - QUESTION 1 - REVISION POWERPOINT.pptx

NEWSPAPERS - QUESTION 1 - REVISION POWERPOINT.pptx
BÀI TẬP DẠY THÊM TIẾNG ANH LỚP 7 CẢ NĂM FRIENDS PLUS SÁCH CHÂN TRỜI SÁNG TẠO ...

BÀI TẬP DẠY THÊM TIẾNG ANH LỚP 7 CẢ NĂM FRIENDS PLUS SÁCH CHÂN TRỜI SÁNG TẠO ...
Liberal Approach to the Study of Indian Politics.pdf

Liberal Approach to the Study of Indian Politics.pdf
Philippine Edukasyong Pantahanan at Pangkabuhayan (EPP) Curriculum

Philippine Edukasyong Pantahanan at Pangkabuhayan (EPP) Curriculum
How to Setup Warehouse & Location in Odoo 17 Inventory

How to Setup Warehouse & Location in Odoo 17 Inventory
Short and simple tricks for assign r and s configuration
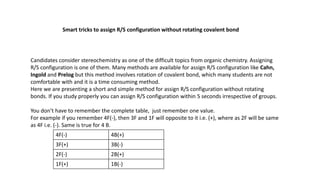
- 1. Smart tricks to assign R/S configuration without rotating covalent bond Candidates consider stereochemistry as one of the difficult topics from organic chemistry. Assigning R/S configuration is one of them. Many methods are available for assign R/S configuration like Cahn, Ingold and Prelog but this method involves rotation of covalent bond, which many students are not comfortable with and it is a time consuming method. Here we are presenting a short and simple method for assign R/S configuration without rotating bonds. If you study properly you can assign R/S configuration within 5 seconds irrespective of groups. You don’t have to remember the complete table, just remember one value. For example if you remember 4F(-), then 3F and 1F will opposite to it i.e. (+), where as 2F will be same as 4F i.e. (-). Same is true for 4 B. 4F(-) 4B(+) 3F(+) 3B(-) 2F(-) 2B(+) 1F(+) 1B(-)
- 2. 4F(-) 4B(+) 3F(+) 3B(-) 2F(-) 2B(+) 1F(+) 1B(-) 4 = 4th Position Priority group F = Front side B = Back side -Ve = Change configuration +Ve = Same configuration Clockwise = RAnitclockwise = S
- 3. Assign Priority Clockwise 11 22 33 44 Example-1 Rule 1 – Assign Priority Rule-2 Select one group above the plane or below the plane. Do not consider this group while drawing the ascending order clockwise or anticlockwise curve. In the above example, we have to select the 4th priority group which is below the plane(Backside) ( You can select a third priority group also) and have drawn the clockwise curve. As per the table 4th priority group is the backside it is 4B(+) and + indicate configuration should not change its R configuration. You can also assign using 3rd priority group – it front side it 3F(+) and configuration would be same R 4F(-) 4B(+) 3F(+) 3B(-) 2F(-) 2B(+) 1F(+) 1B(-)
- 4. 1 2Assign Priority 3 4 Anti-Clockwise 1 2 3 4 1B(-) 1st Priority group on back side and - sign indicate reverse the configuration hence its R configuration 4F(-) 4B(+) 3F(+) 3B(-) 2F(-) 2B(+) 1F(+) 1B(-)
- 5. Example -2 Assign Priority 1 2 Anti-Clockwise 3 4 1 2 3 4 1F(+) 1st Priority group on front side and + sign indicate no change in configuration hence its S configuration 4F(-) 4B(+) 3F(+) 3B(-) 2F(-) 2B(+) 1F(+) 1B(-) You can assign configuration to any chiral molecule without rotating bonds. Regular practice will help you in attaining mastery in assigning configuration.