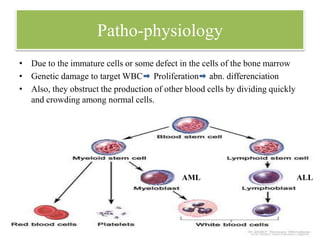
Leukemia is a group of blood cancers that begin in the bone marrow and result in abnormal blood cells. The most common types are acute lymphoblastic leukemia (ALL) in children and chronic lymphocytic leukemia (CLL) and acute myeloid leukemia (AML) in adults. Treatment involves chemotherapy, targeted therapy, radiation therapy, bone marrow transplant, immunotherapy or CAR-T cell therapy to kill leukemia cells and achieve remission in phases including induction, consolidation, and maintenance therapy.