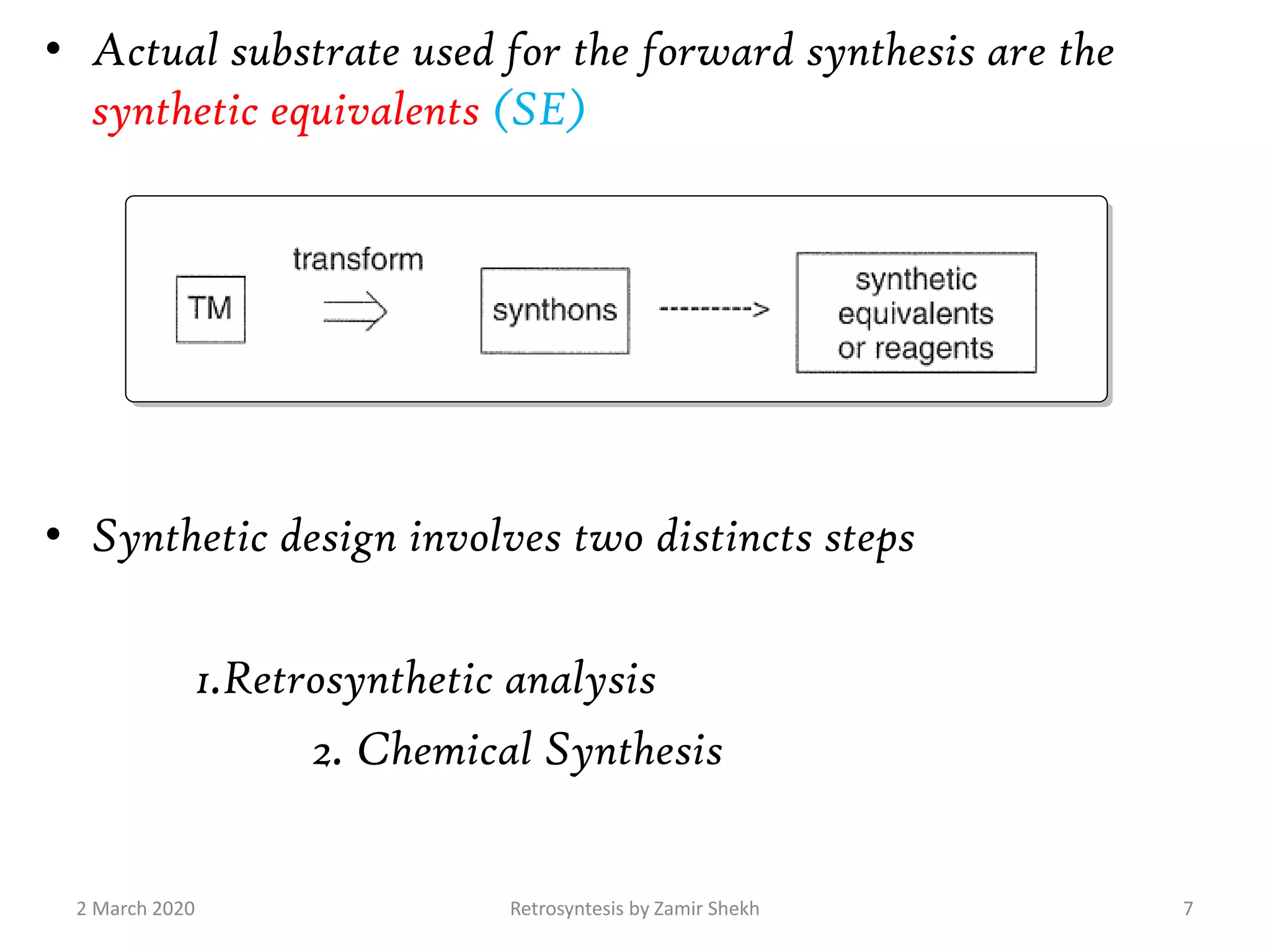
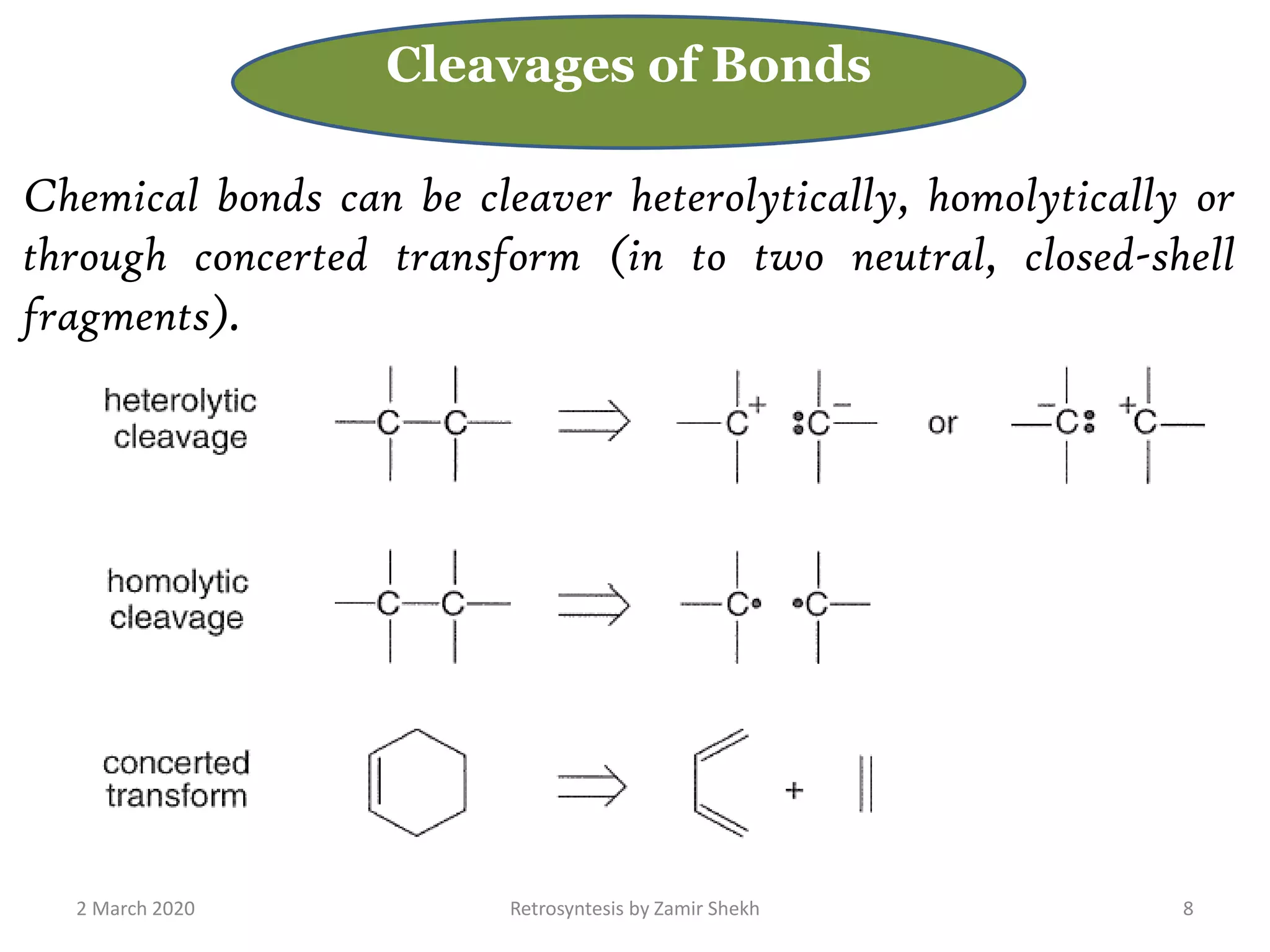
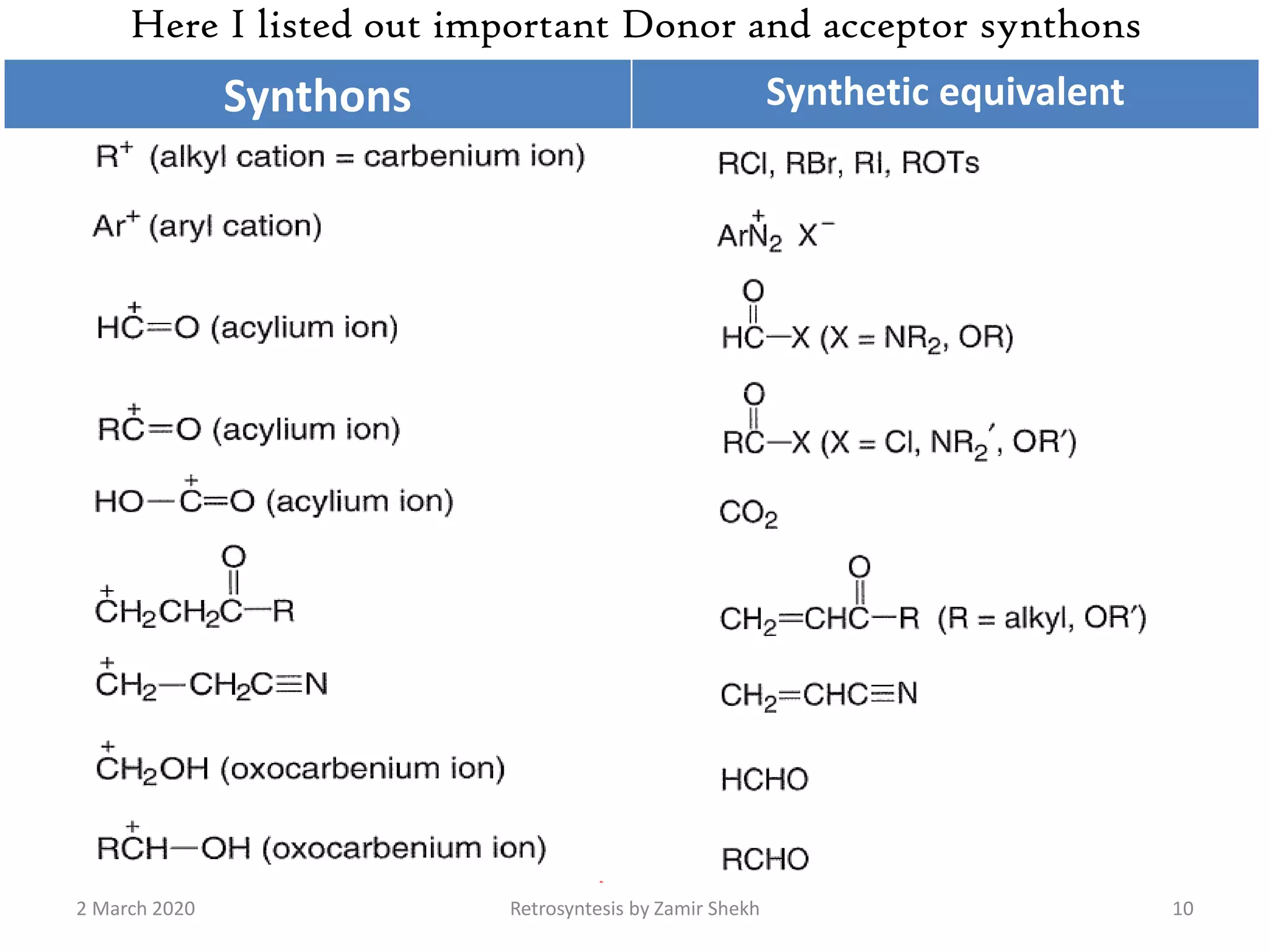
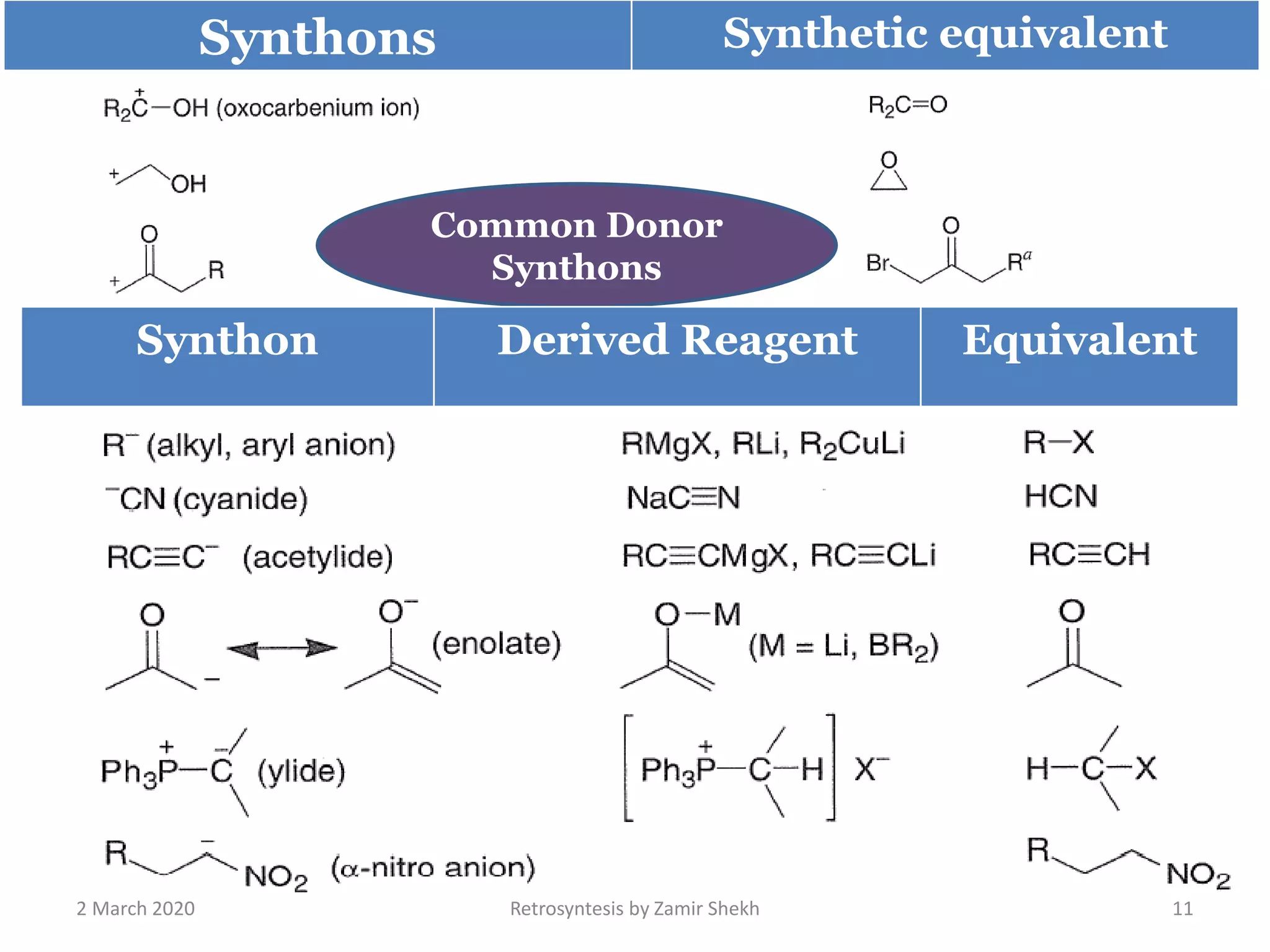
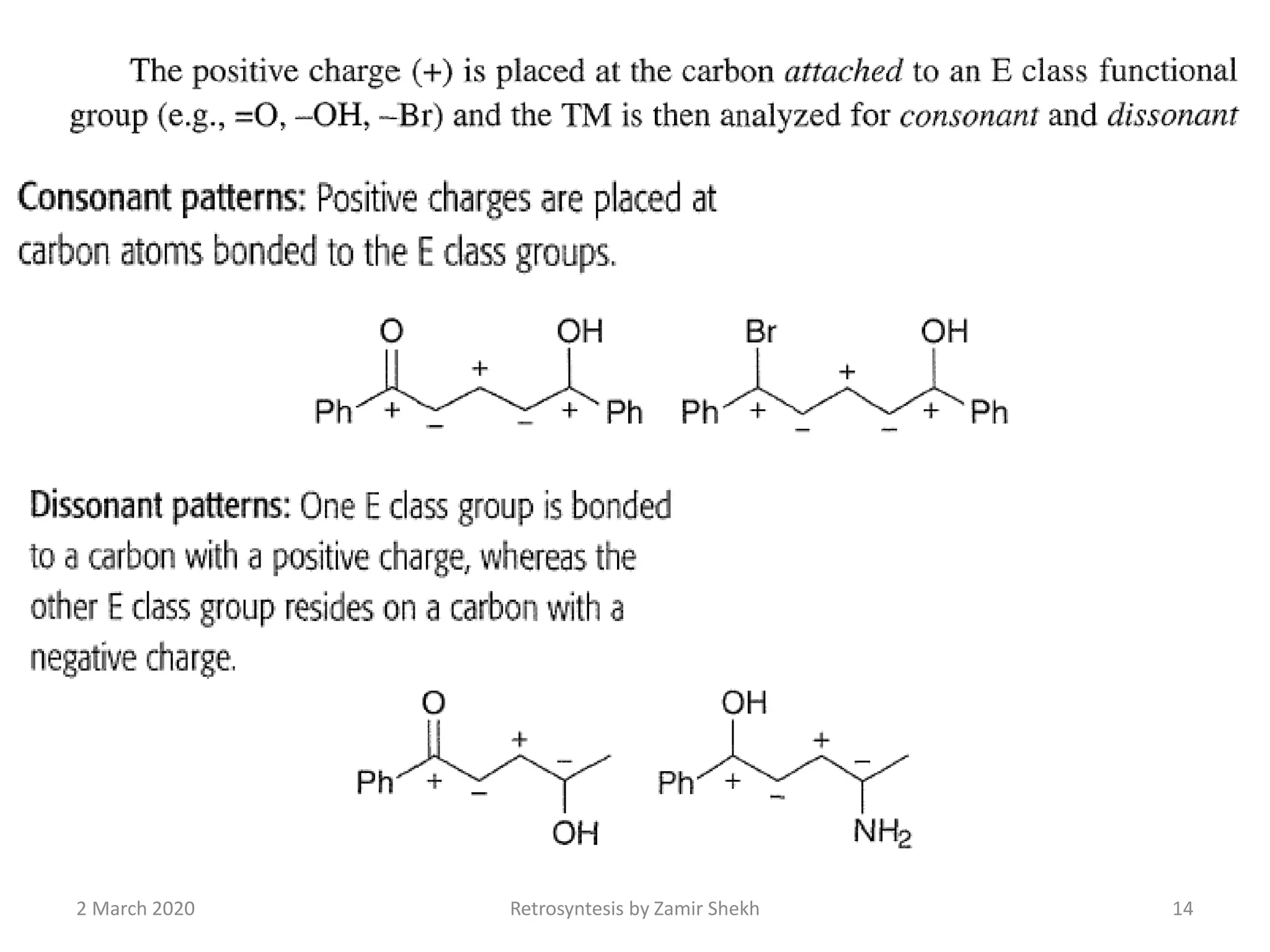
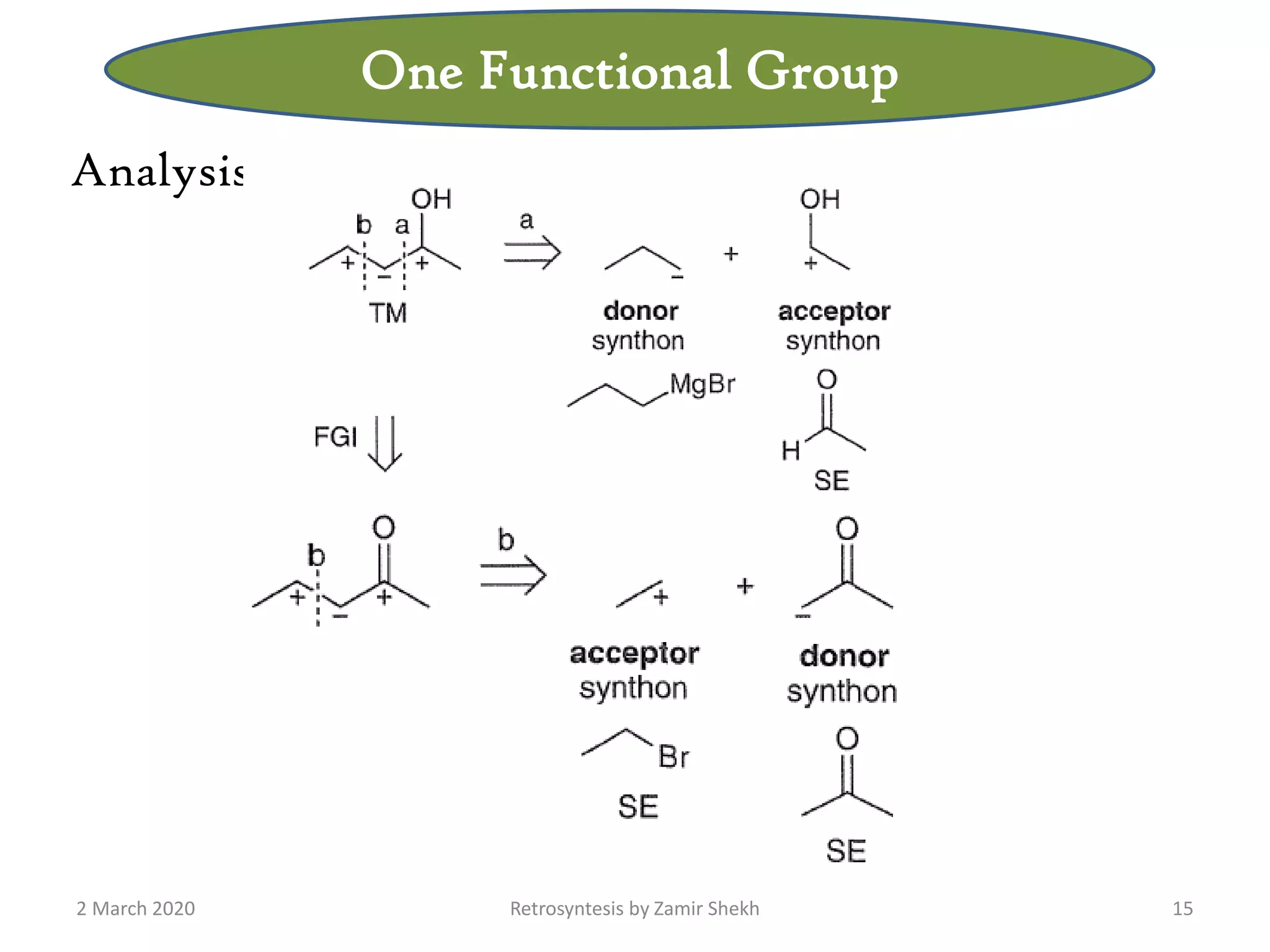
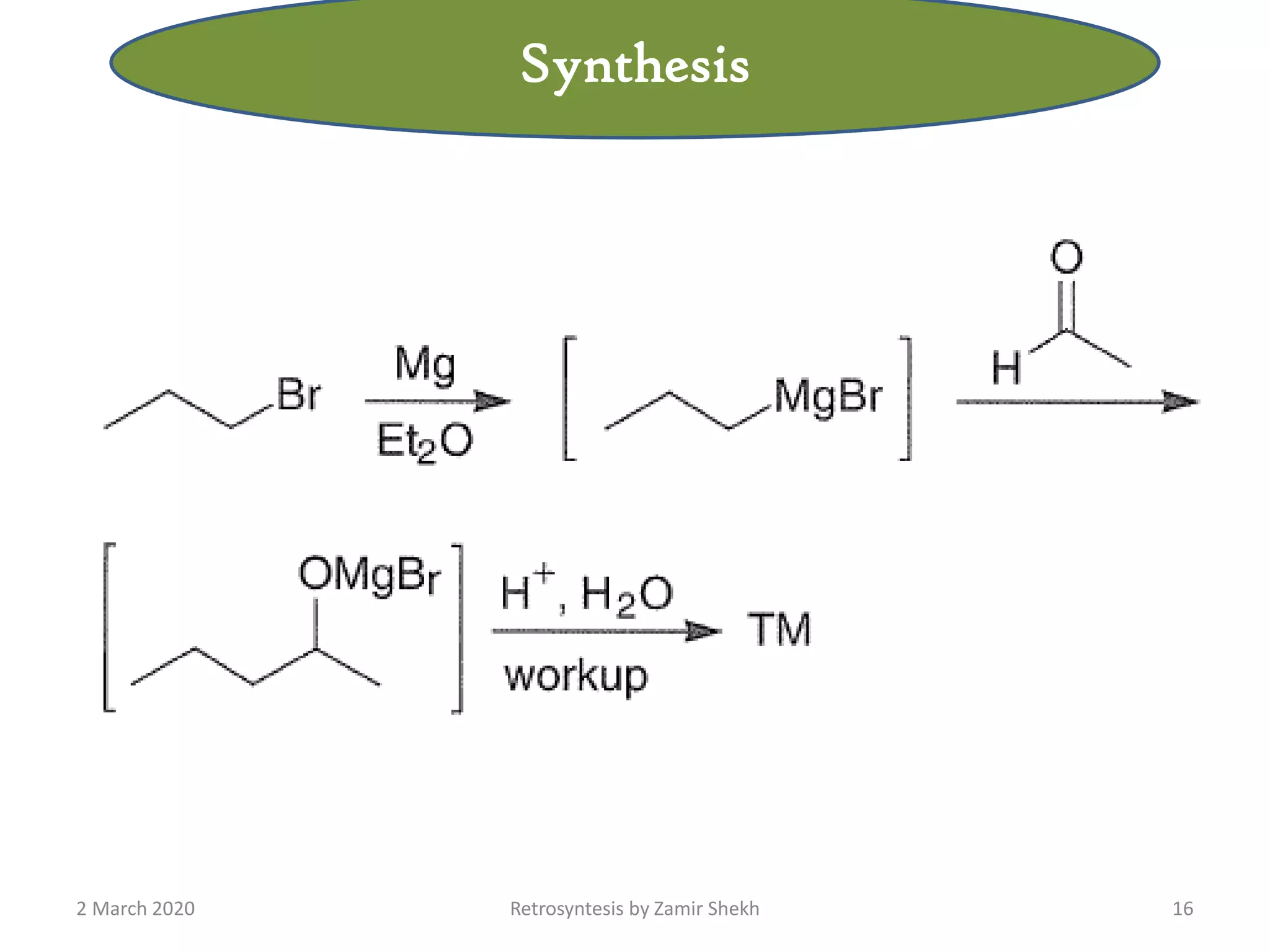
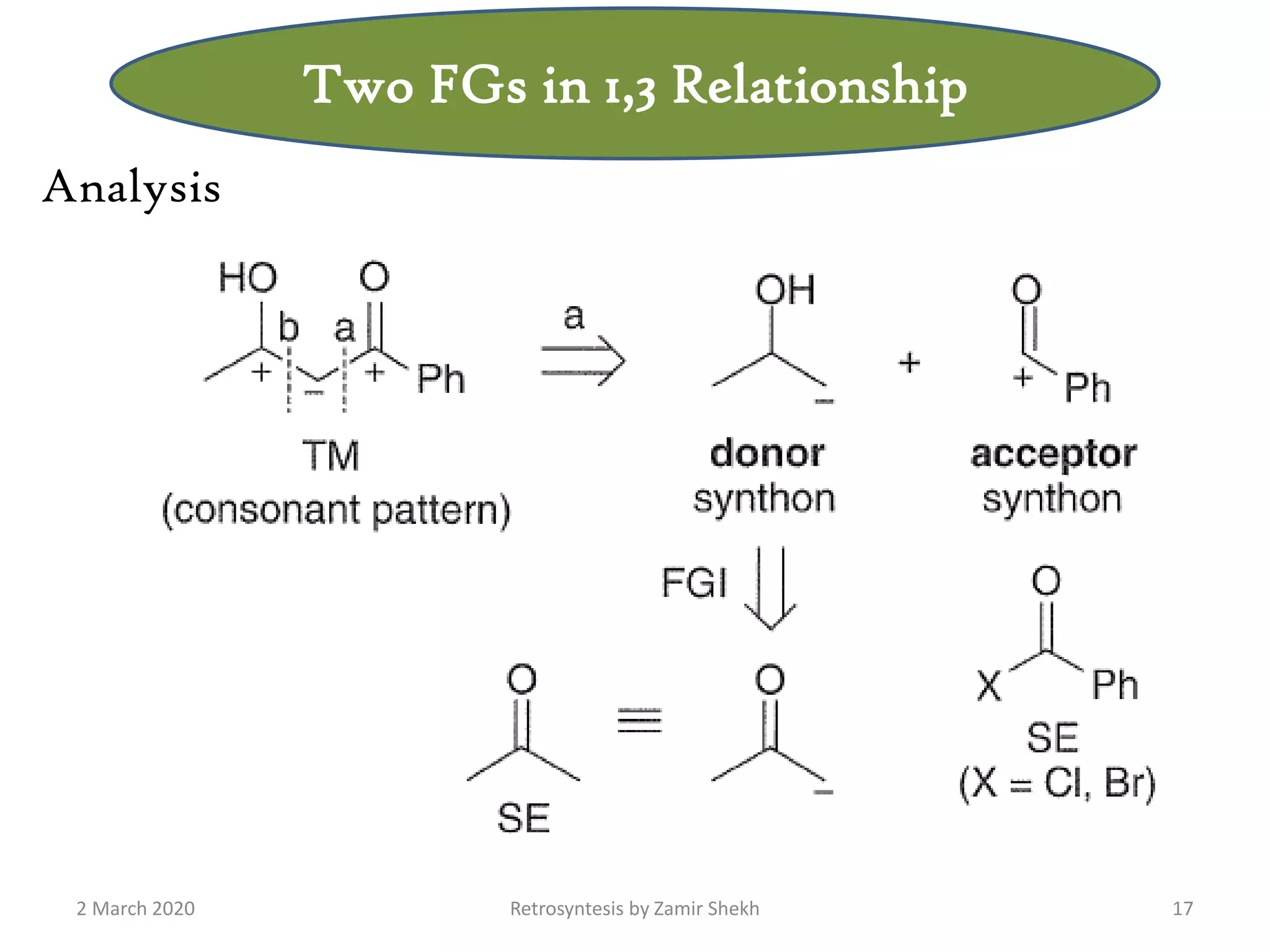
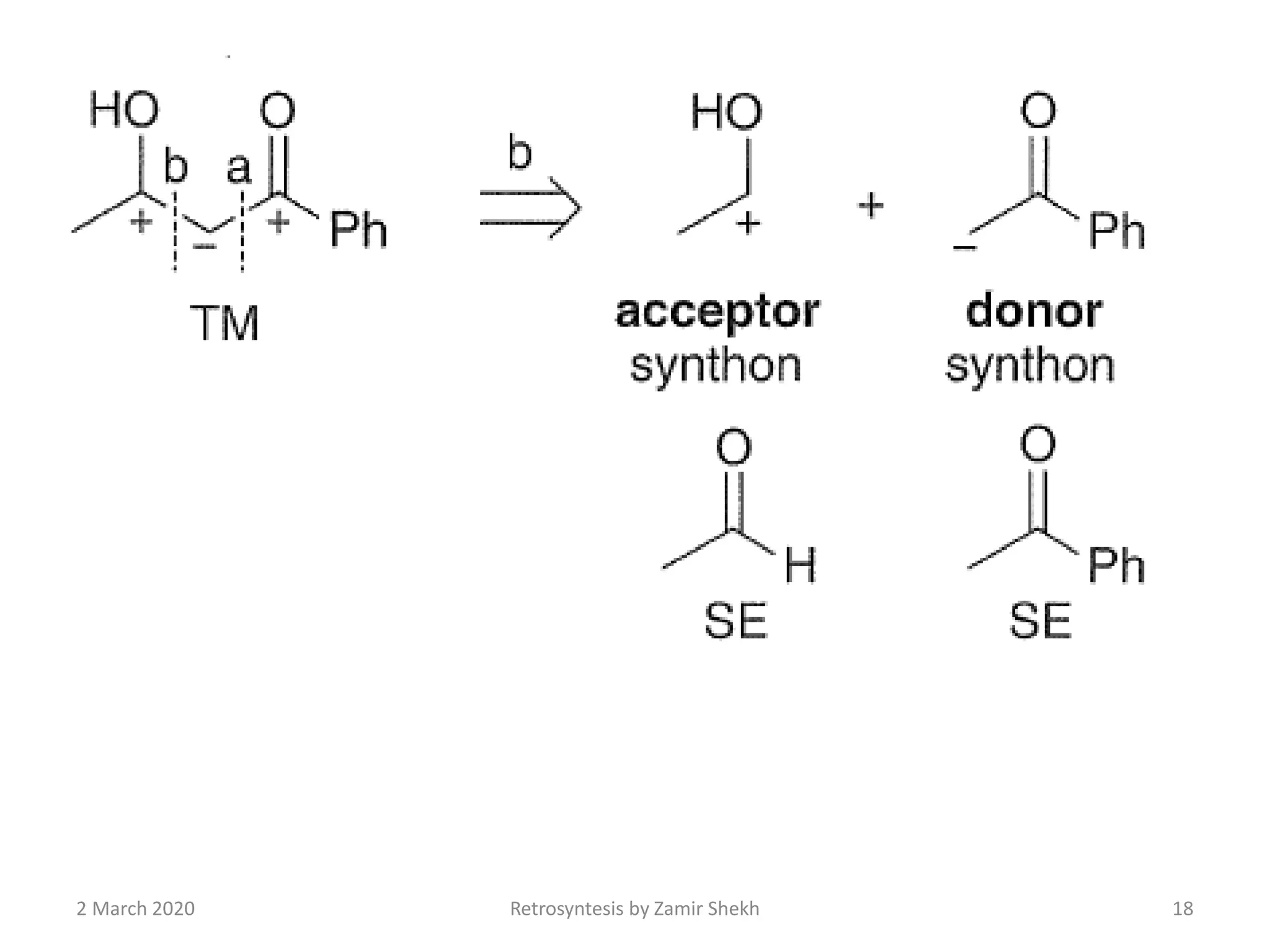
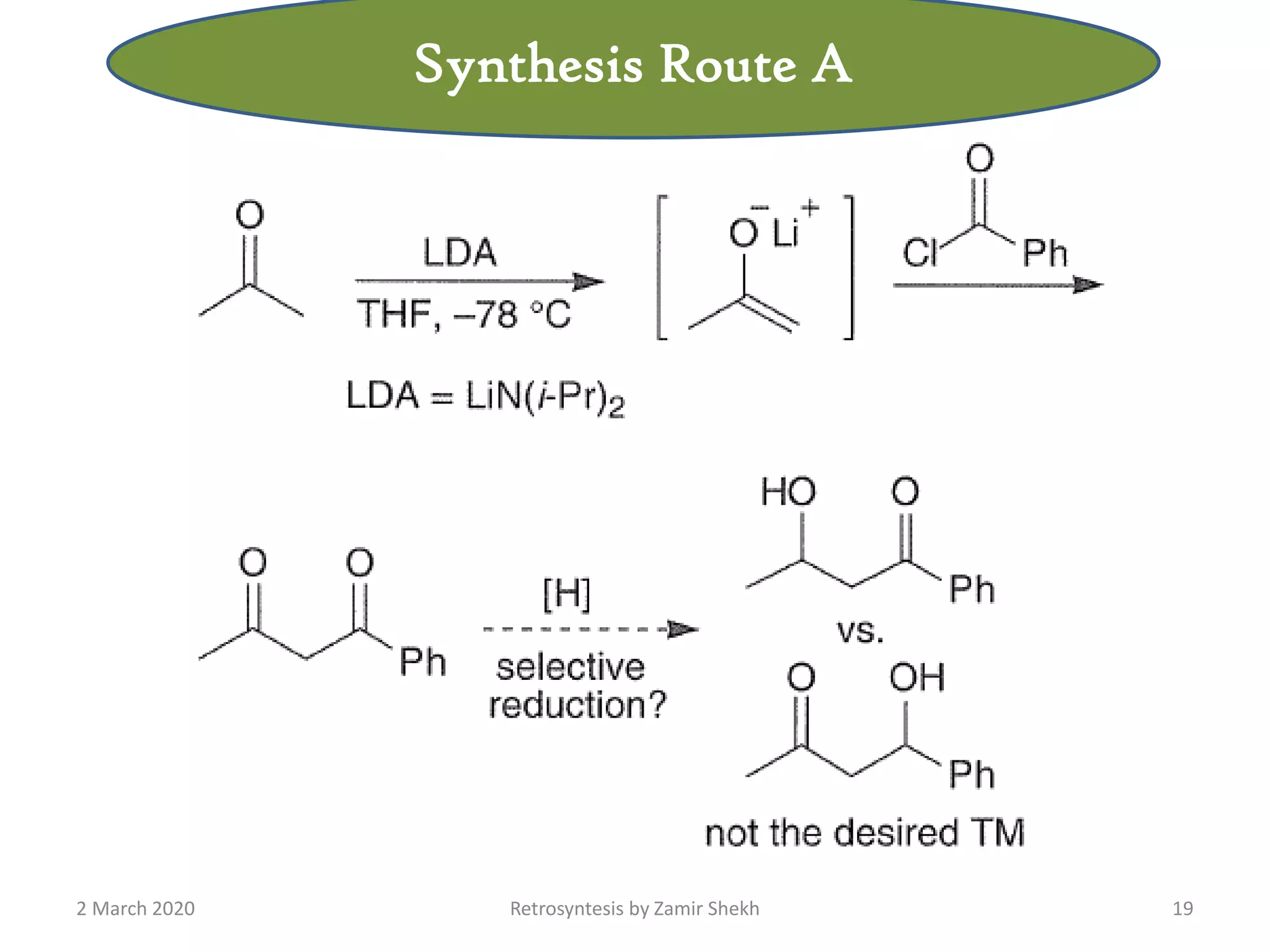
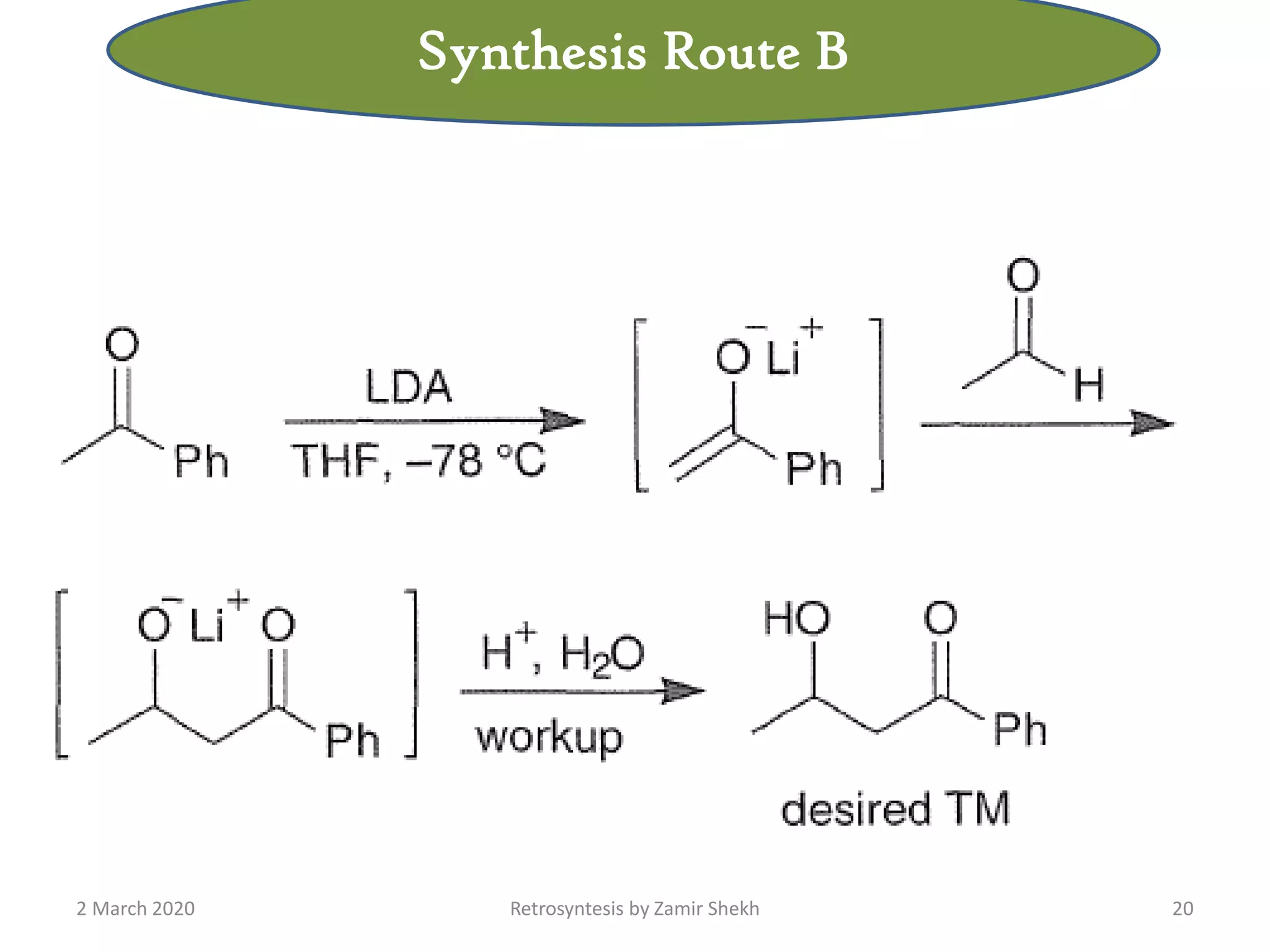
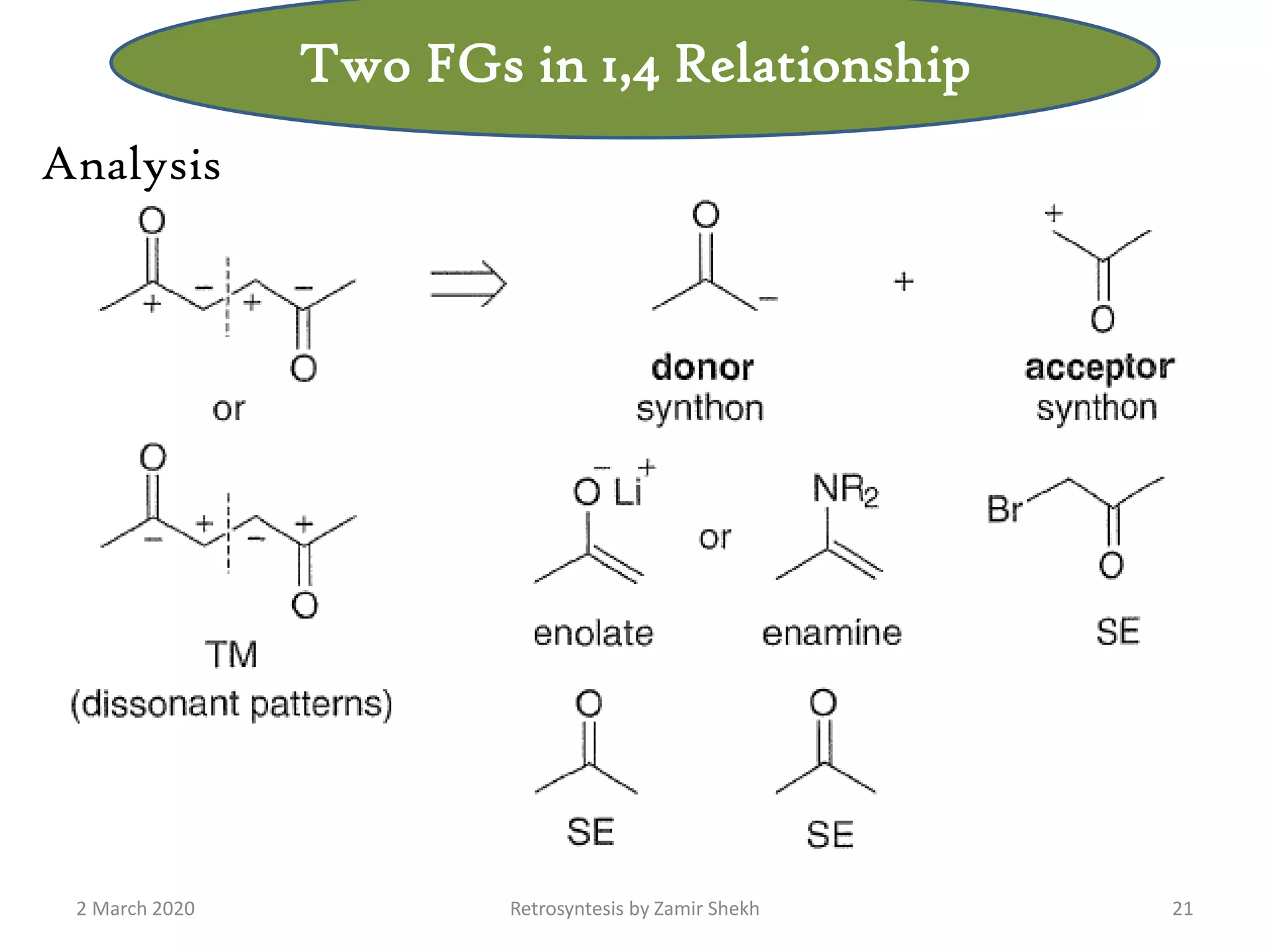
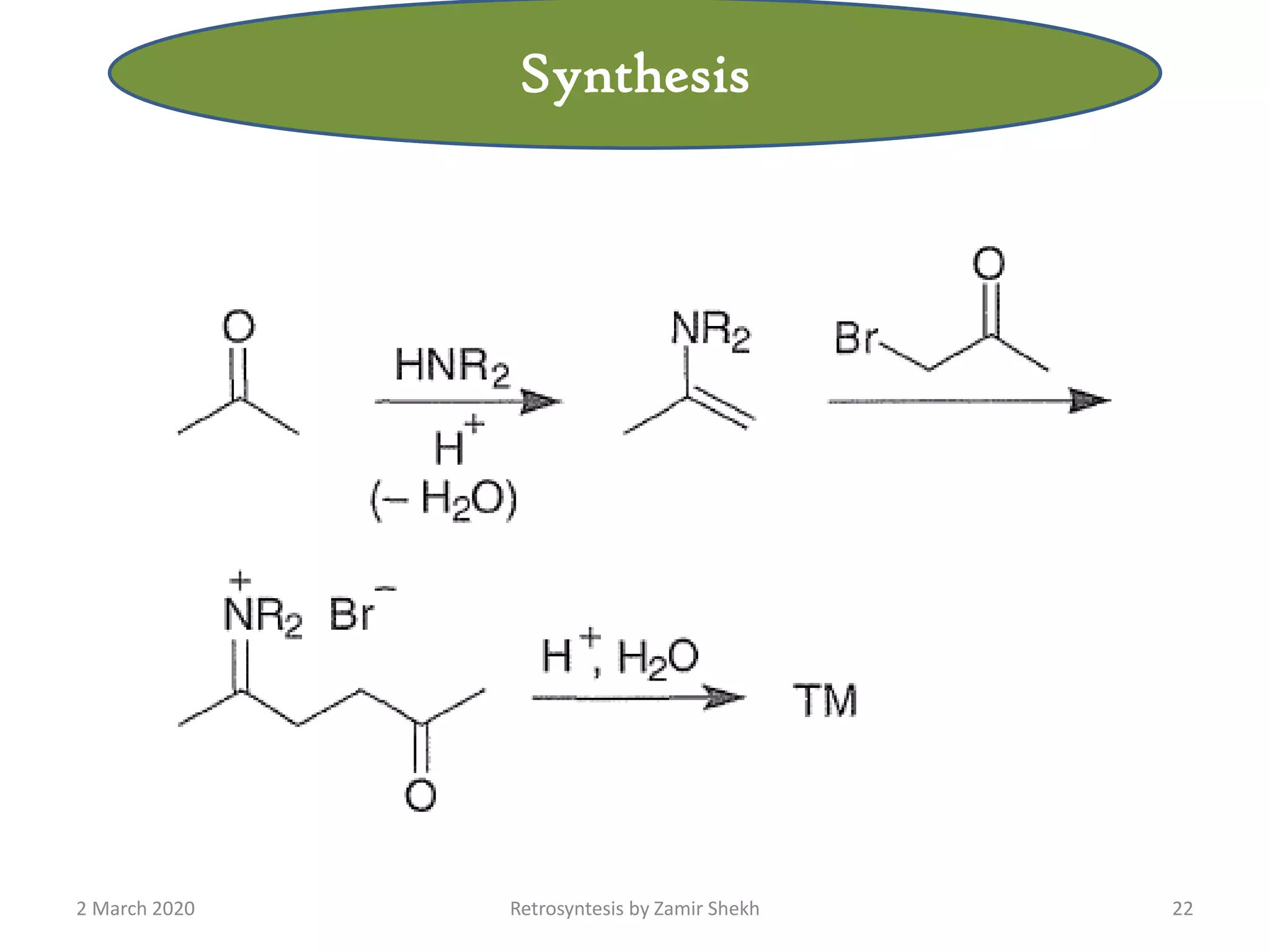
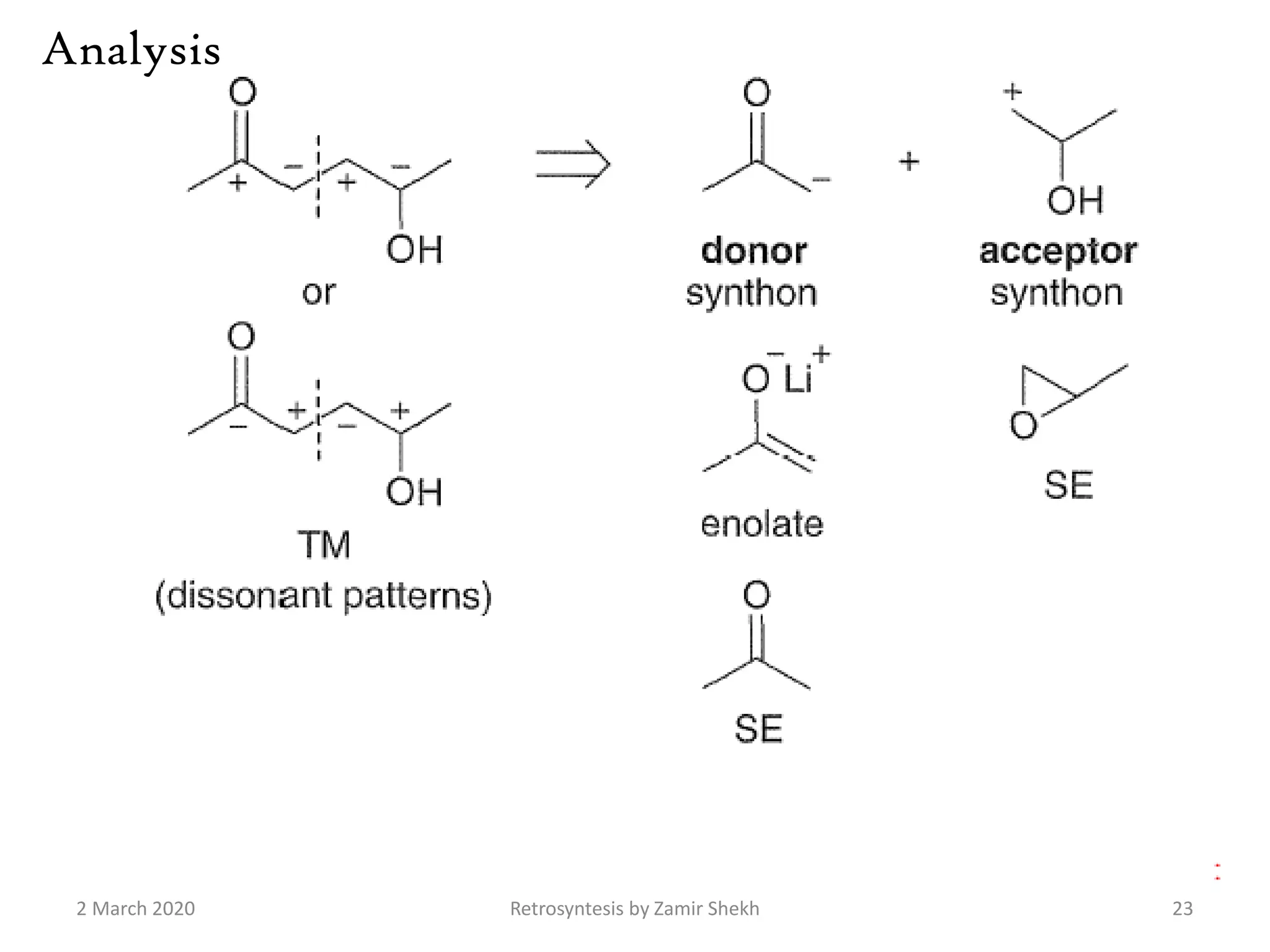
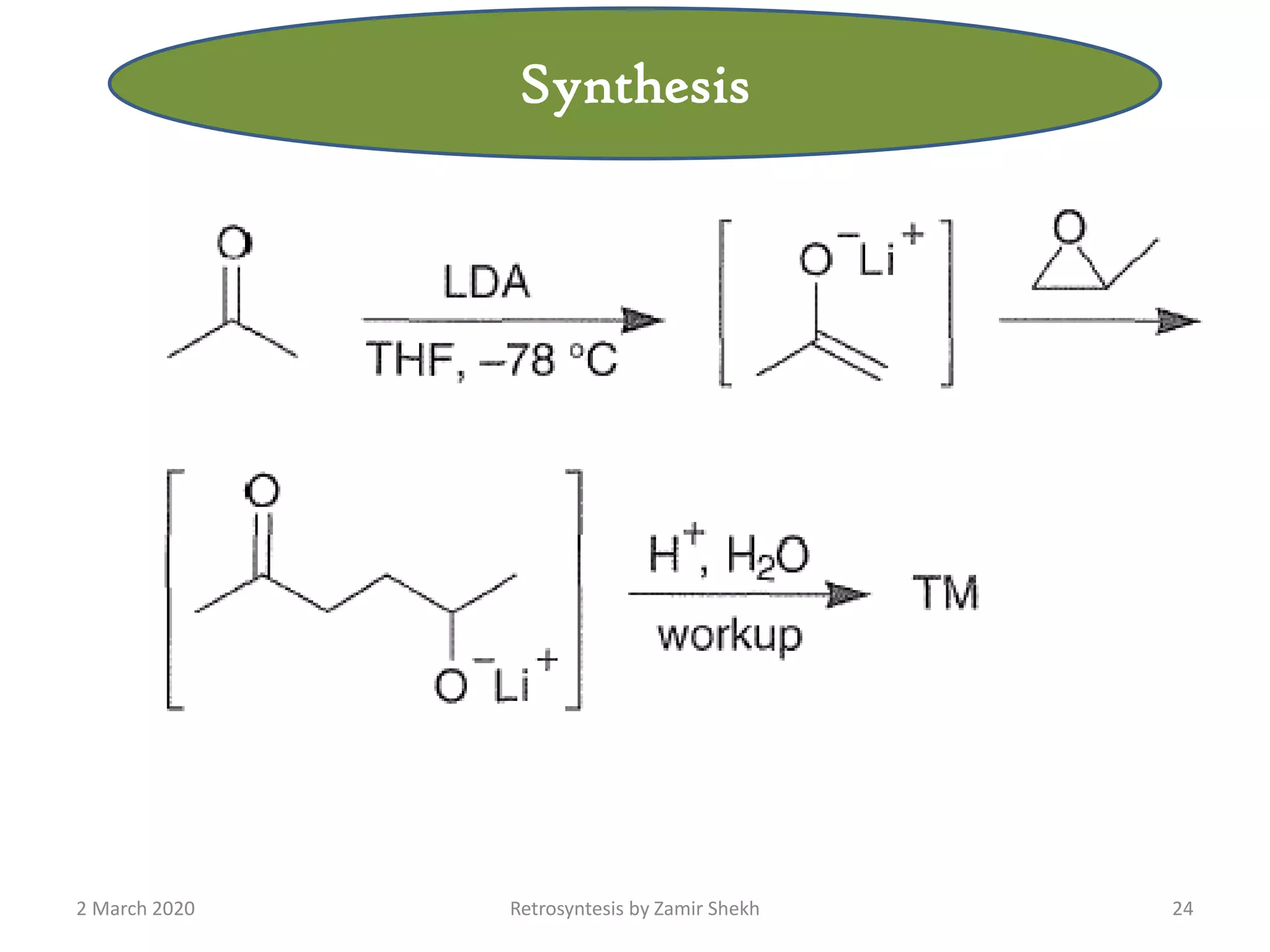
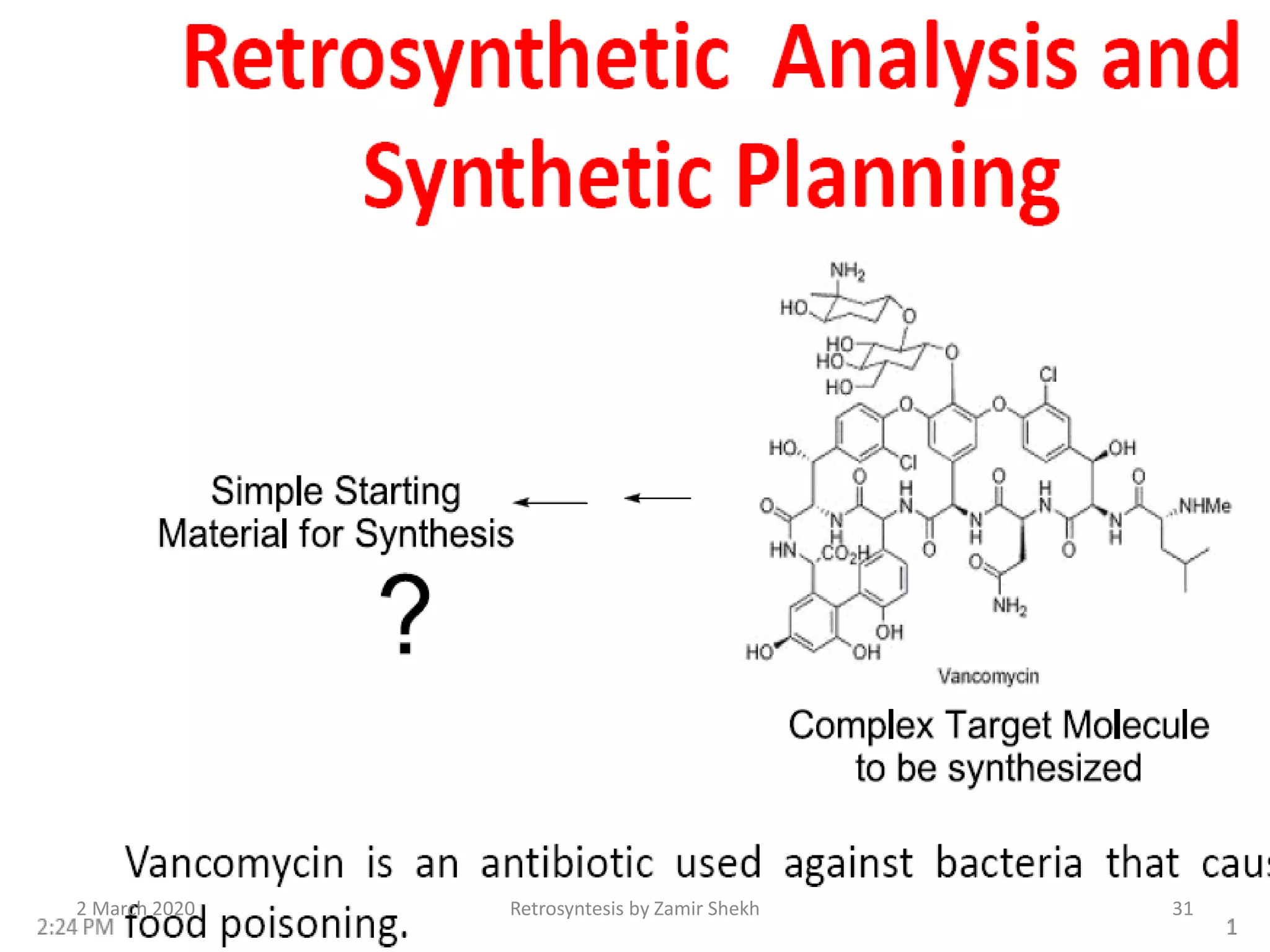
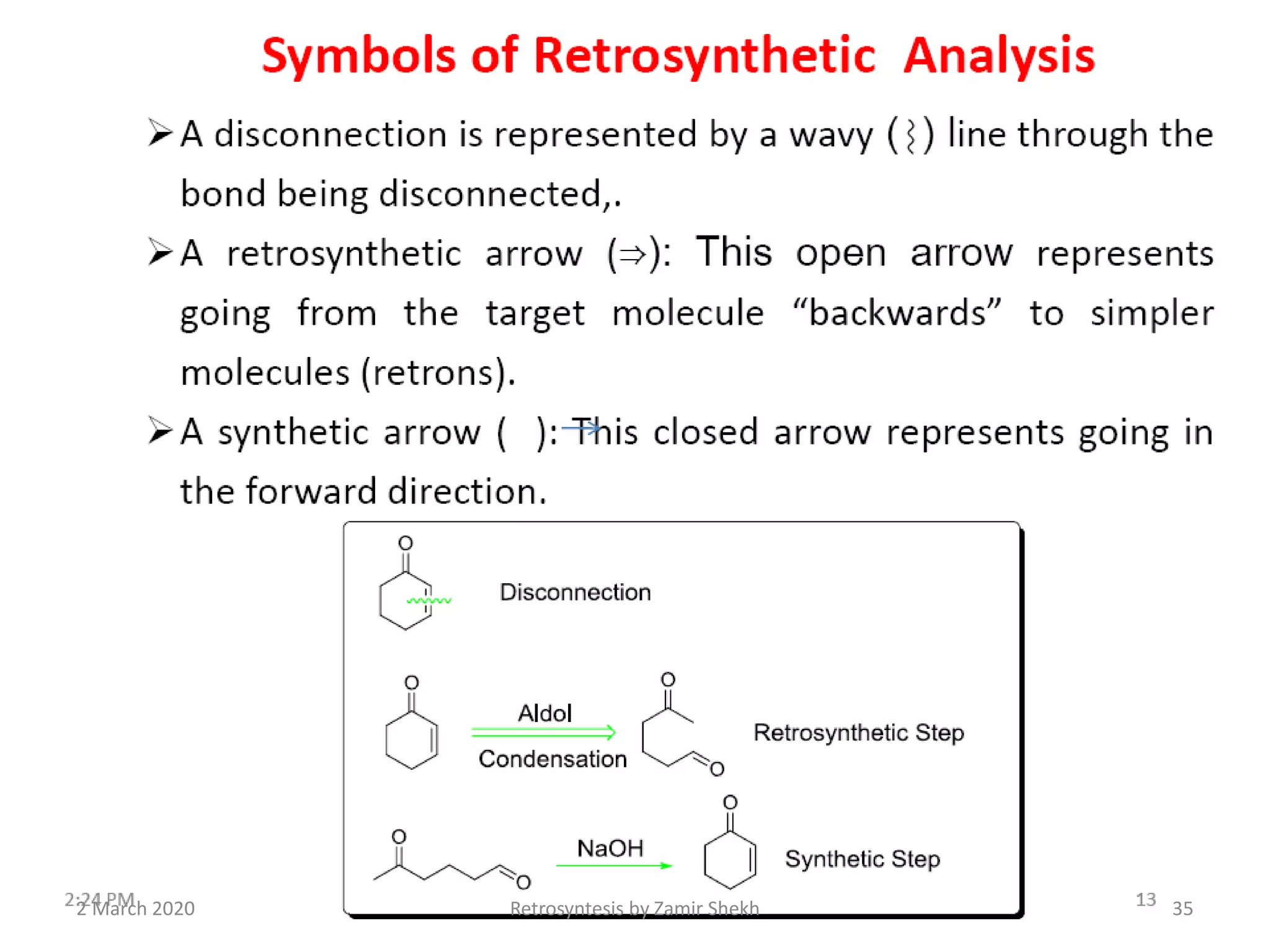
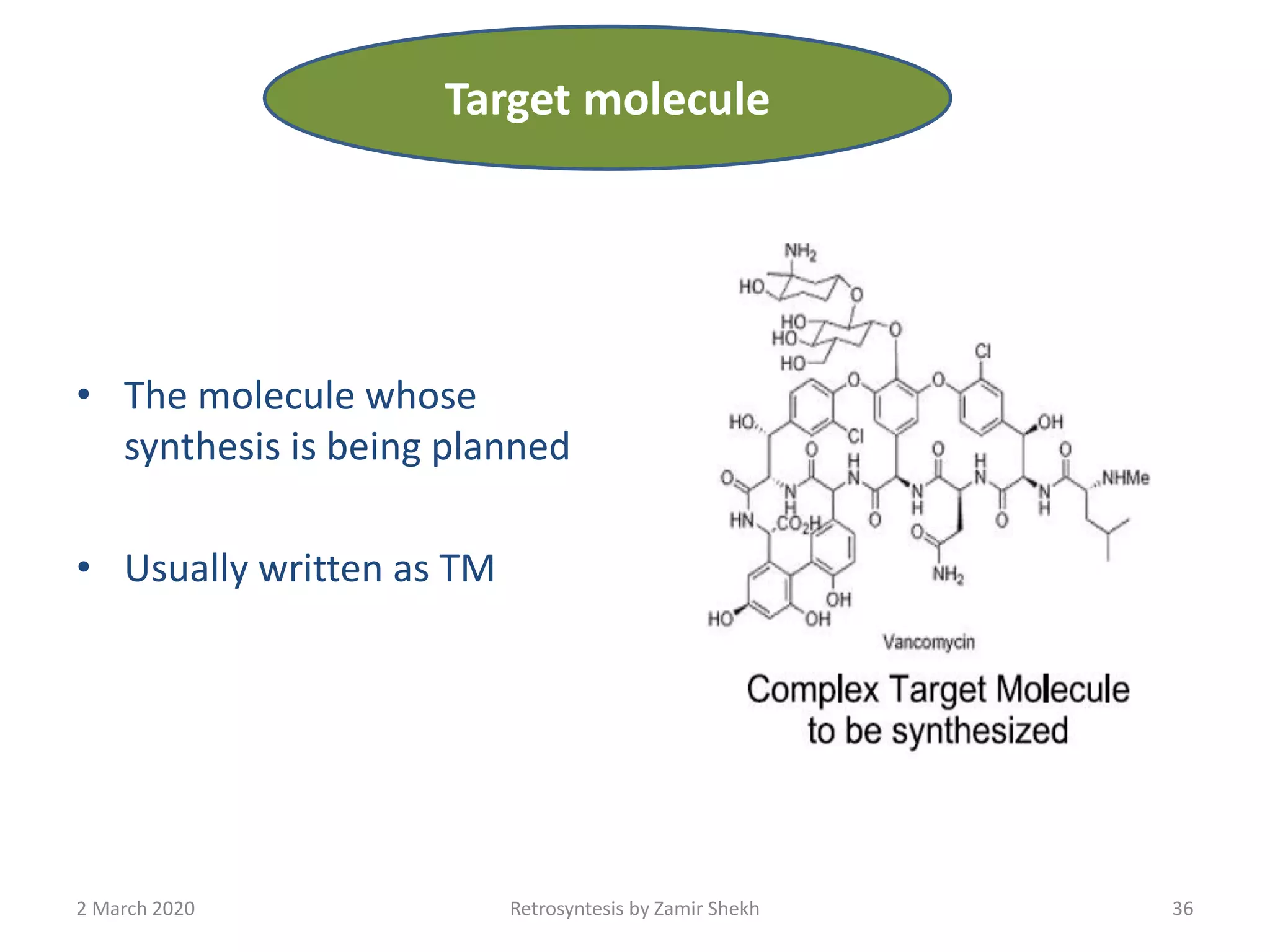
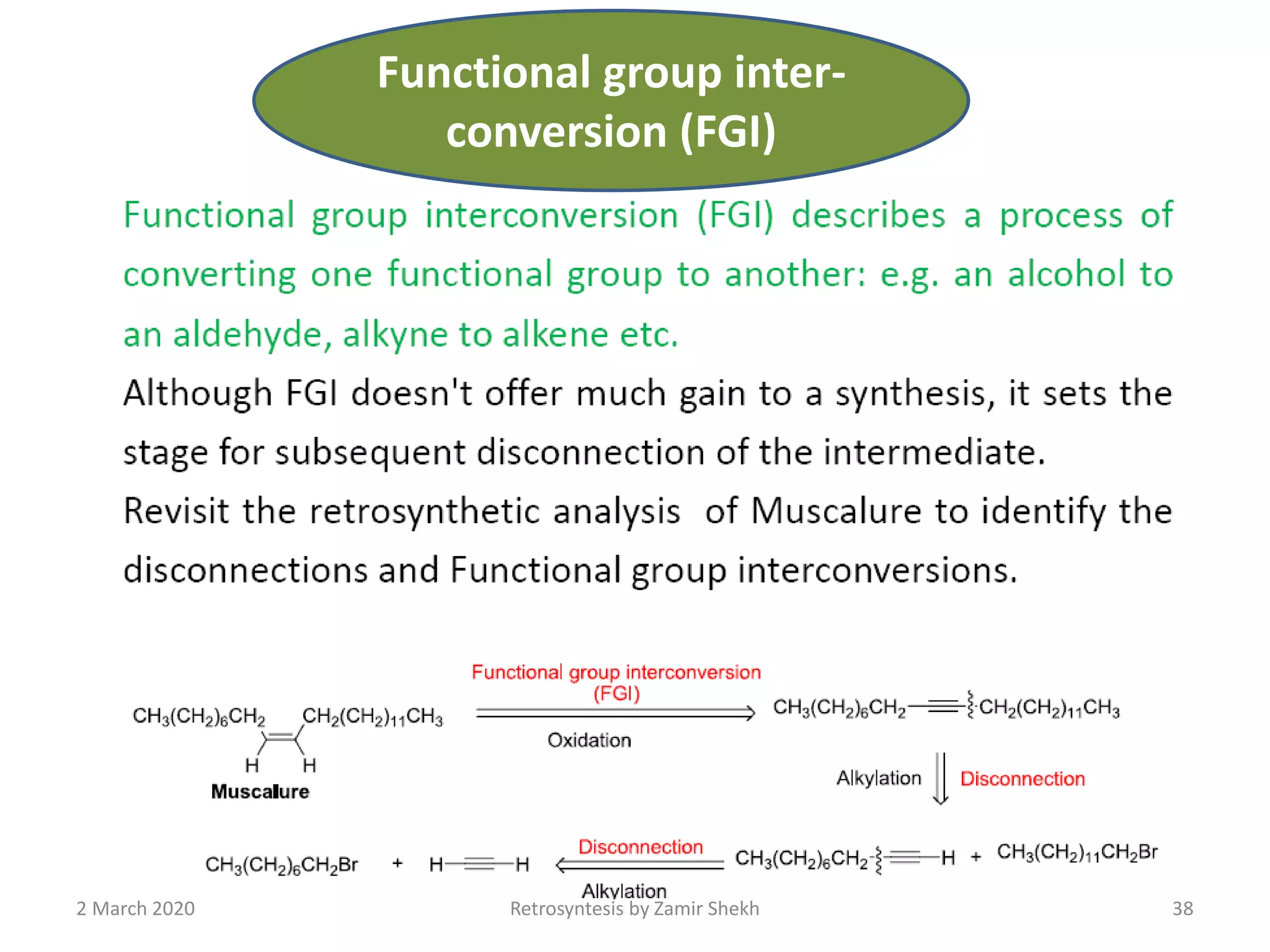
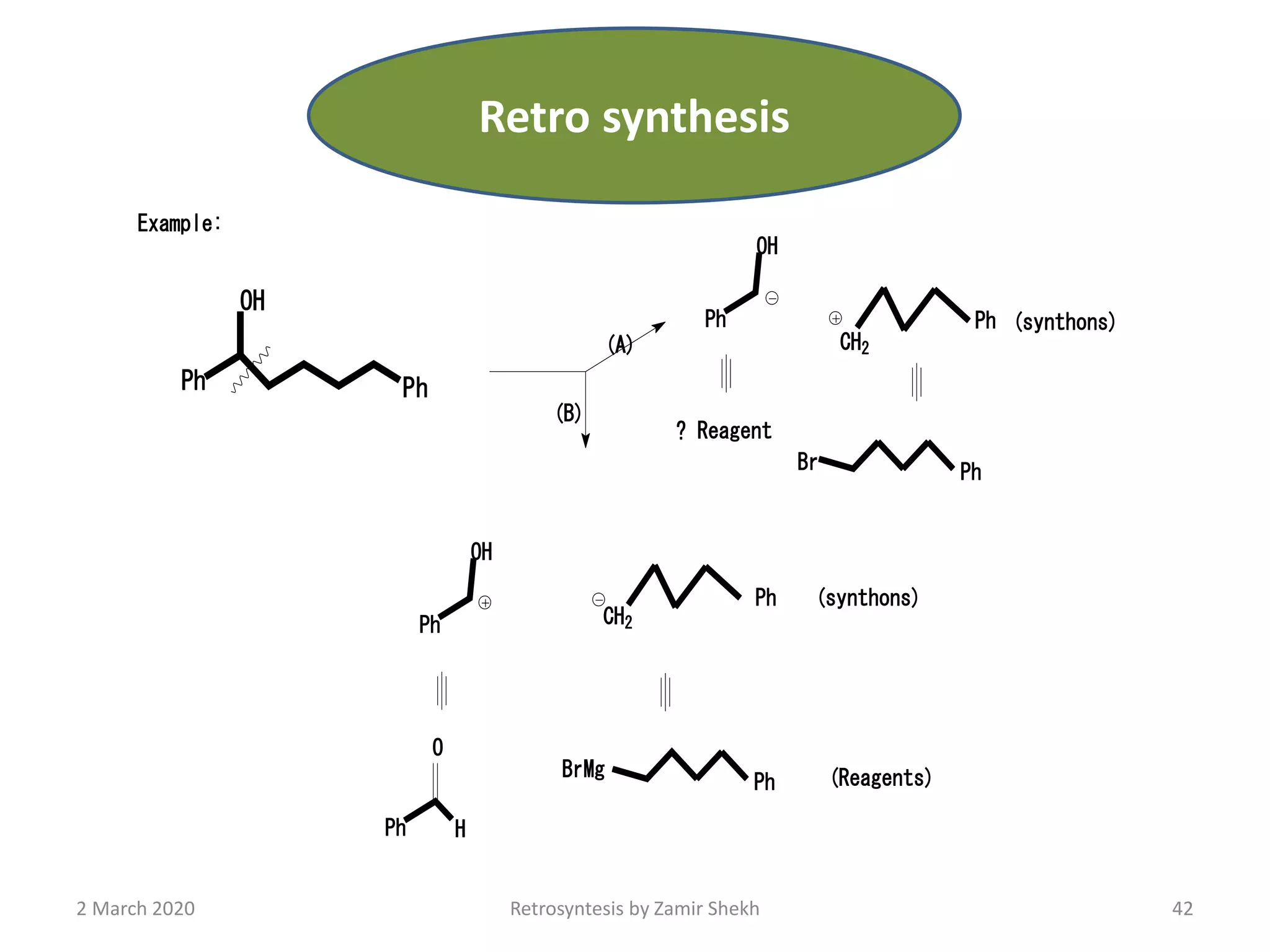
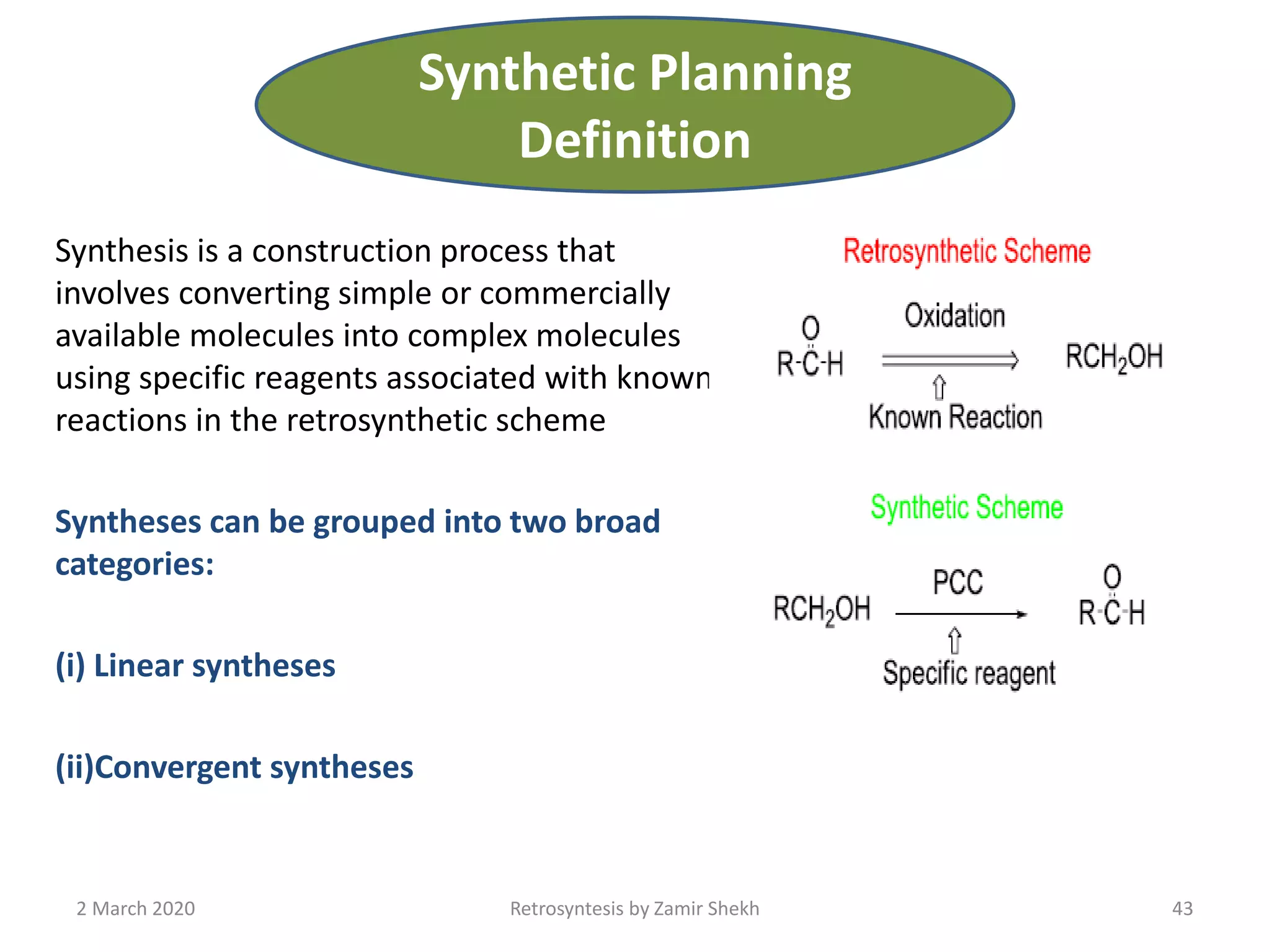
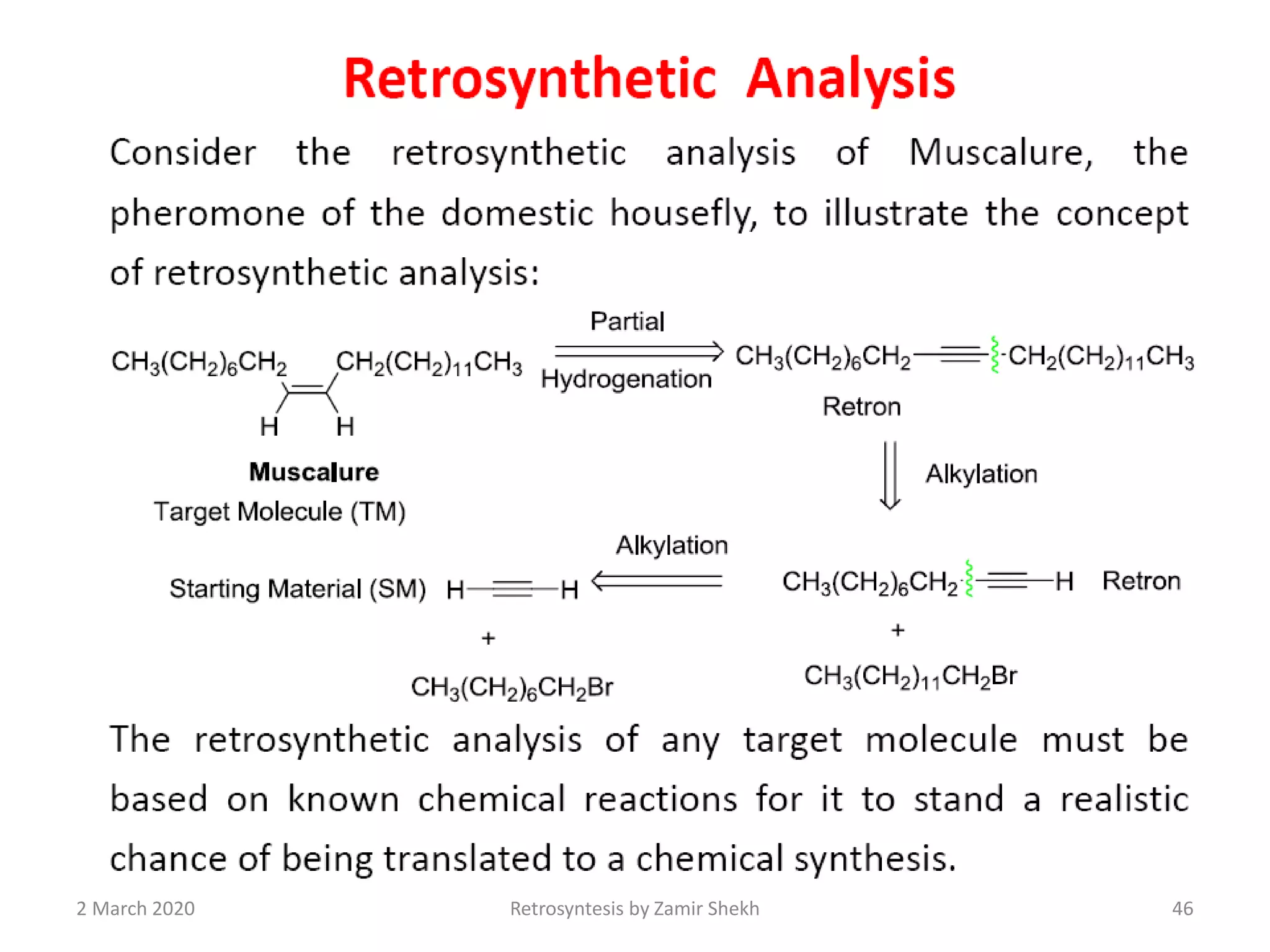
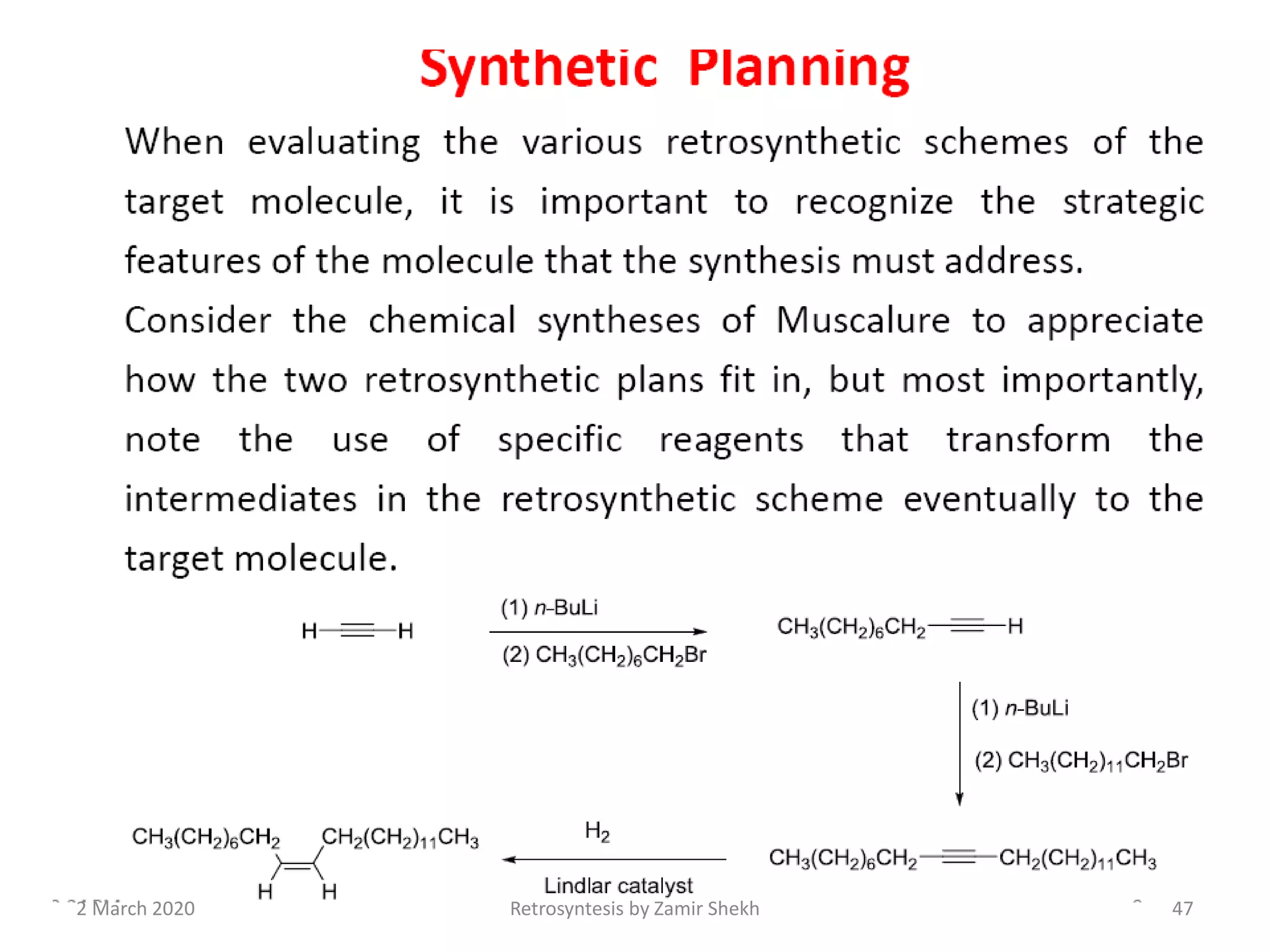
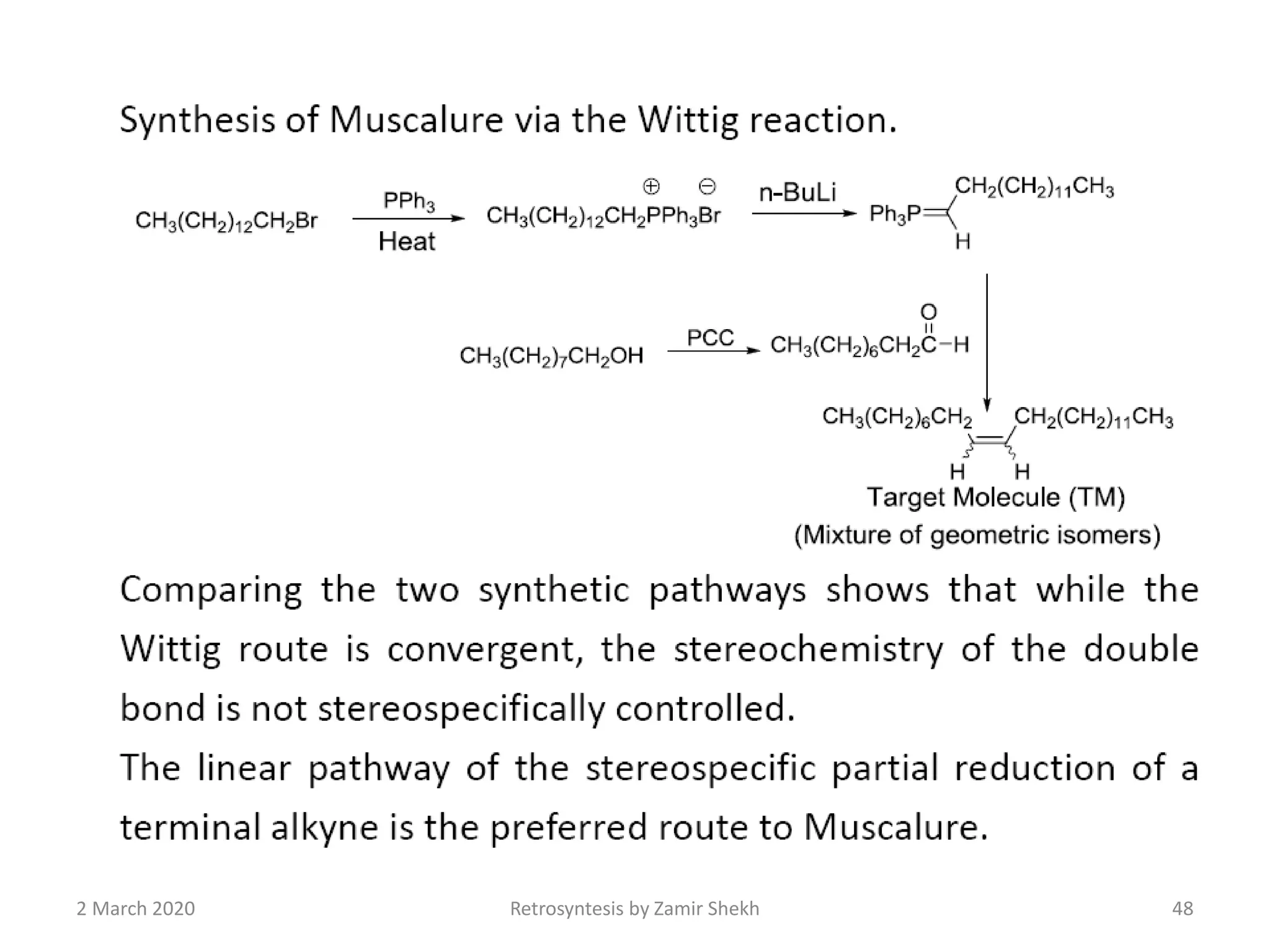
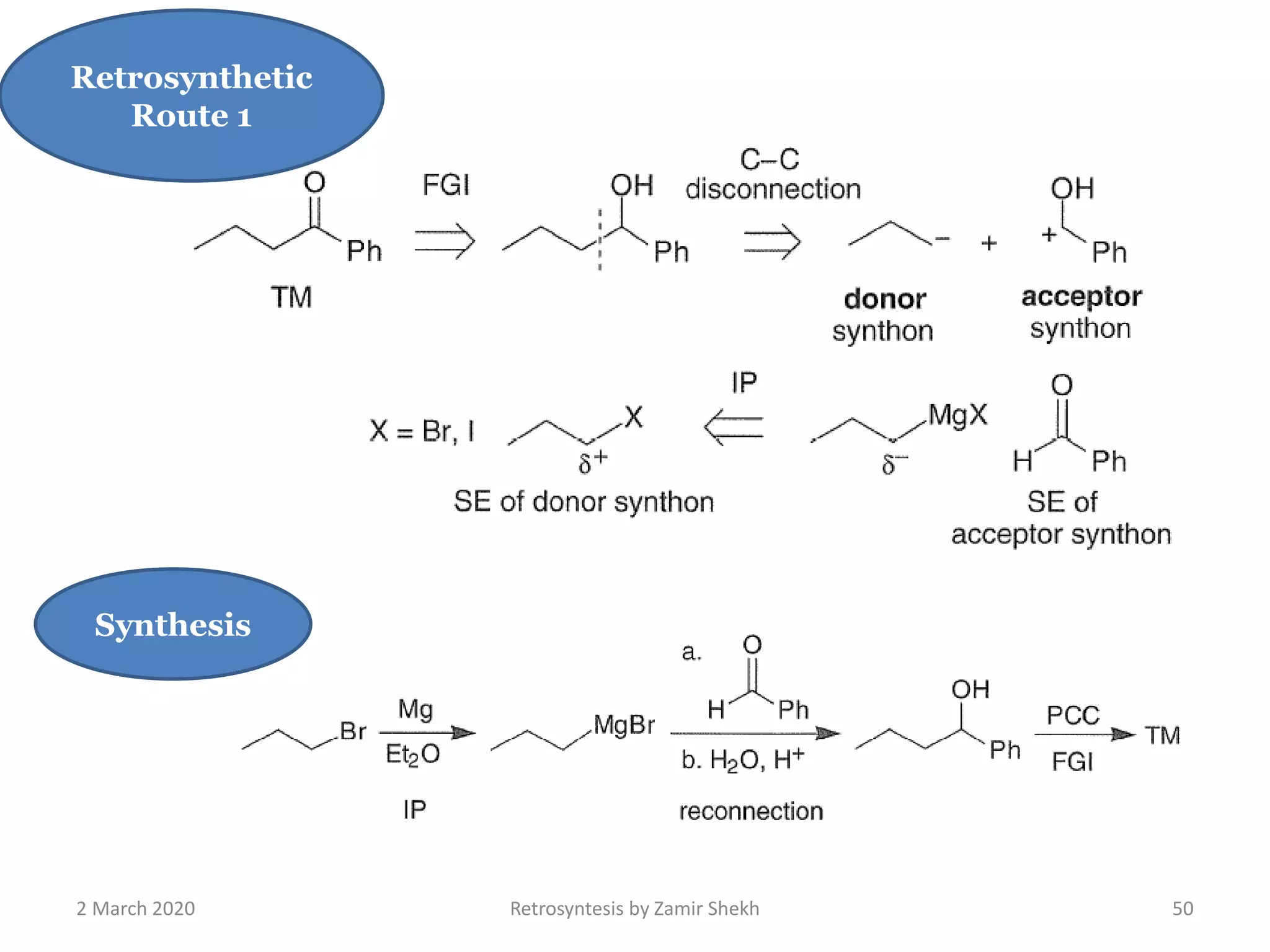
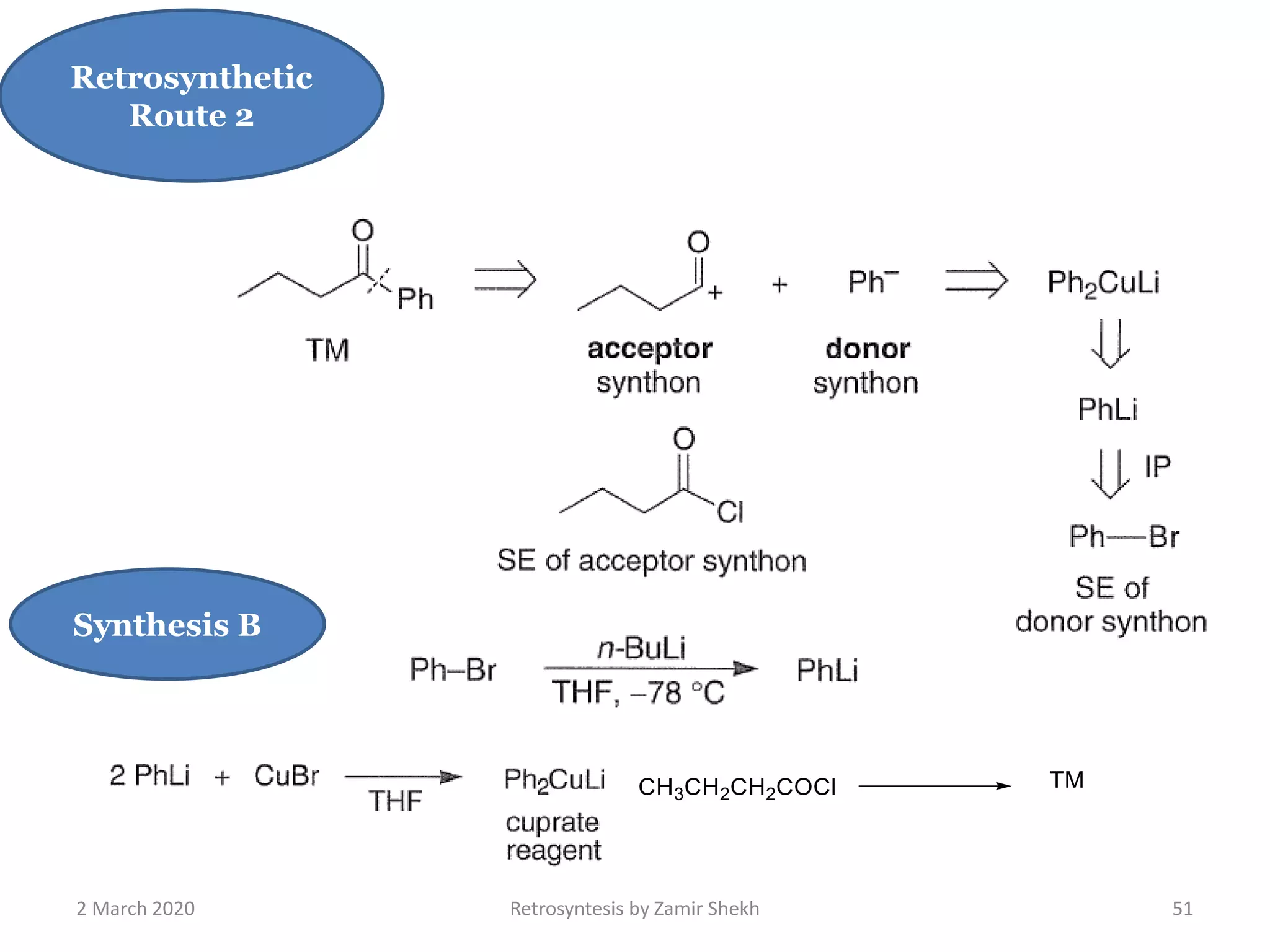
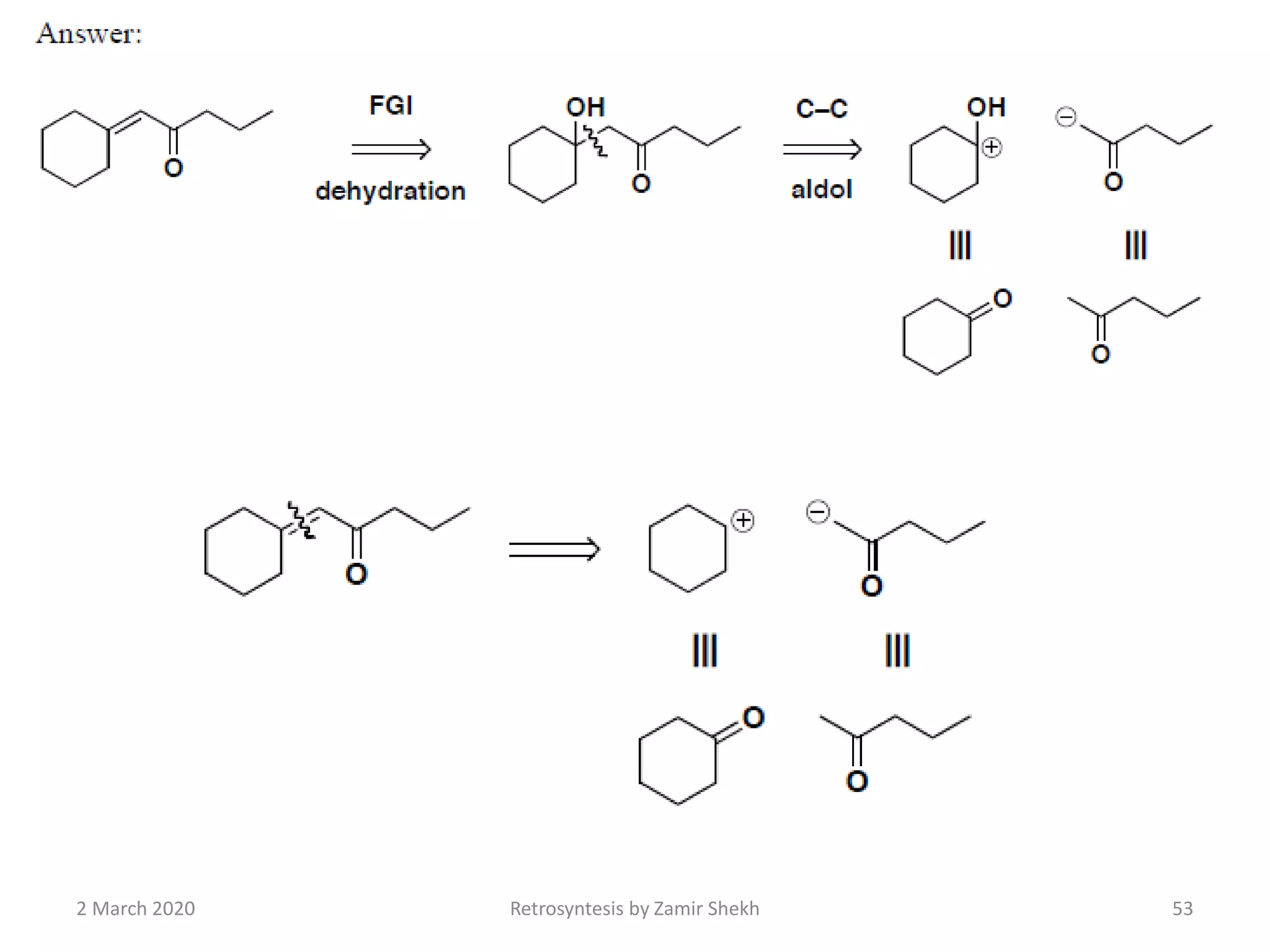
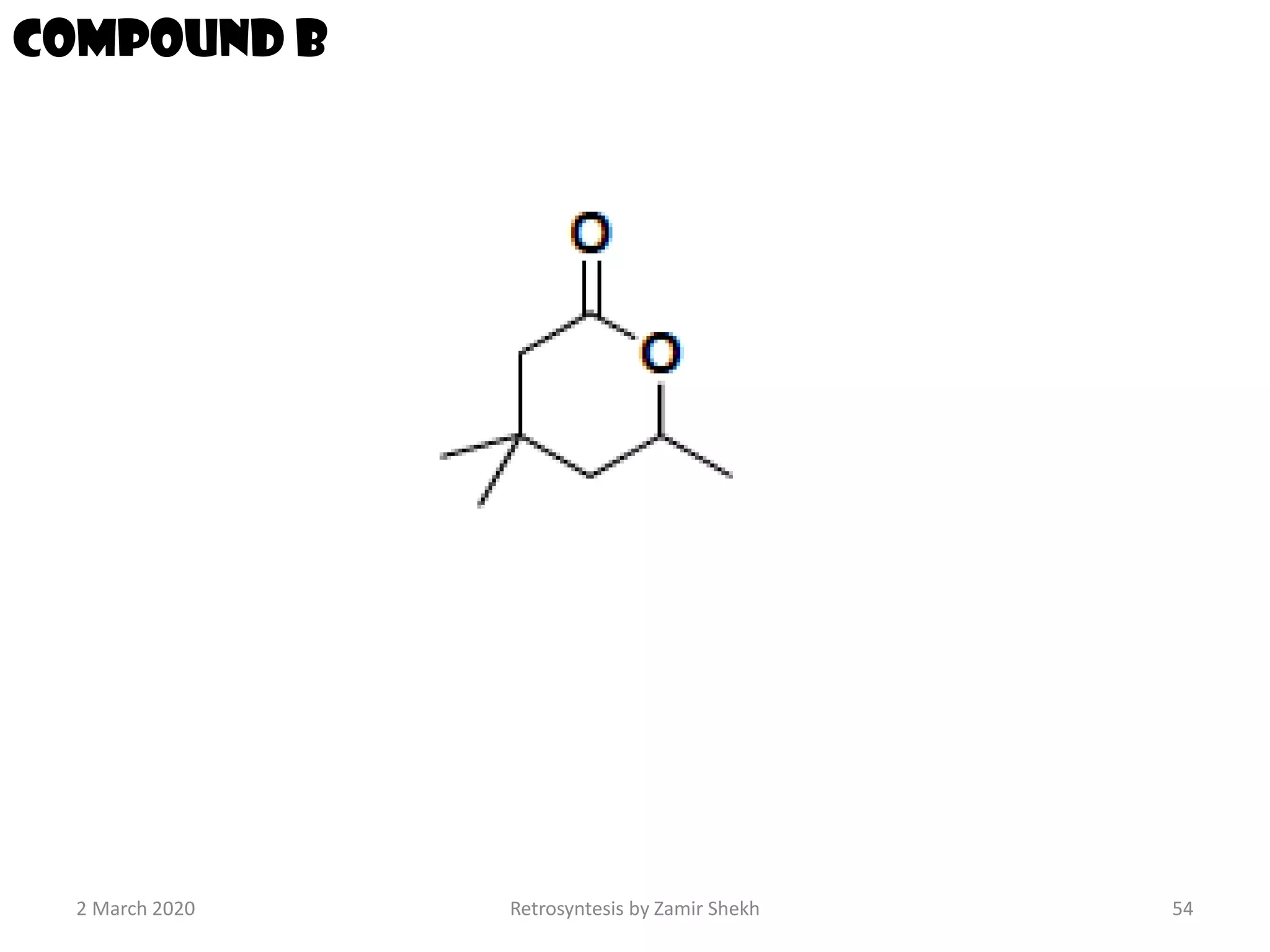
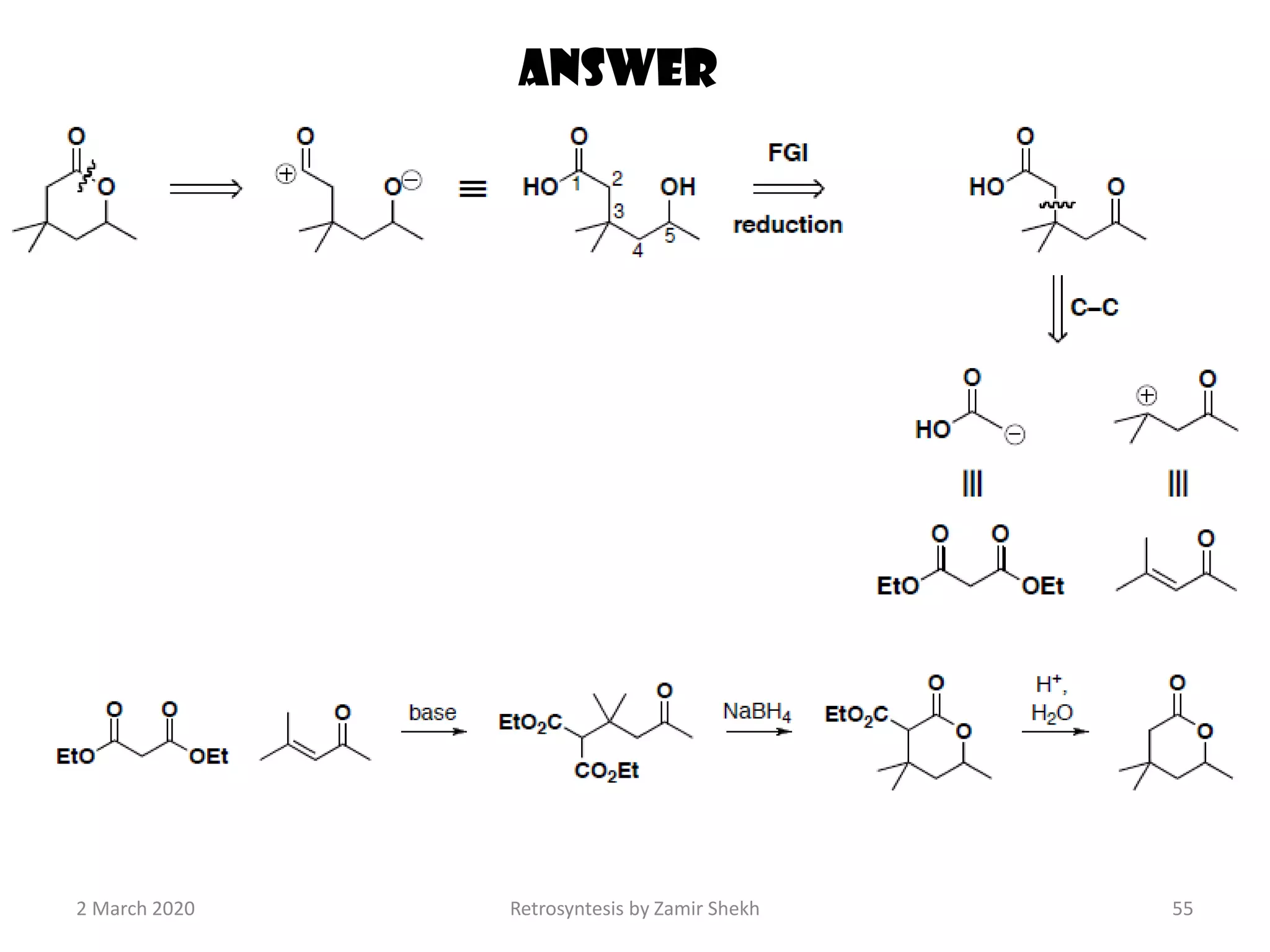
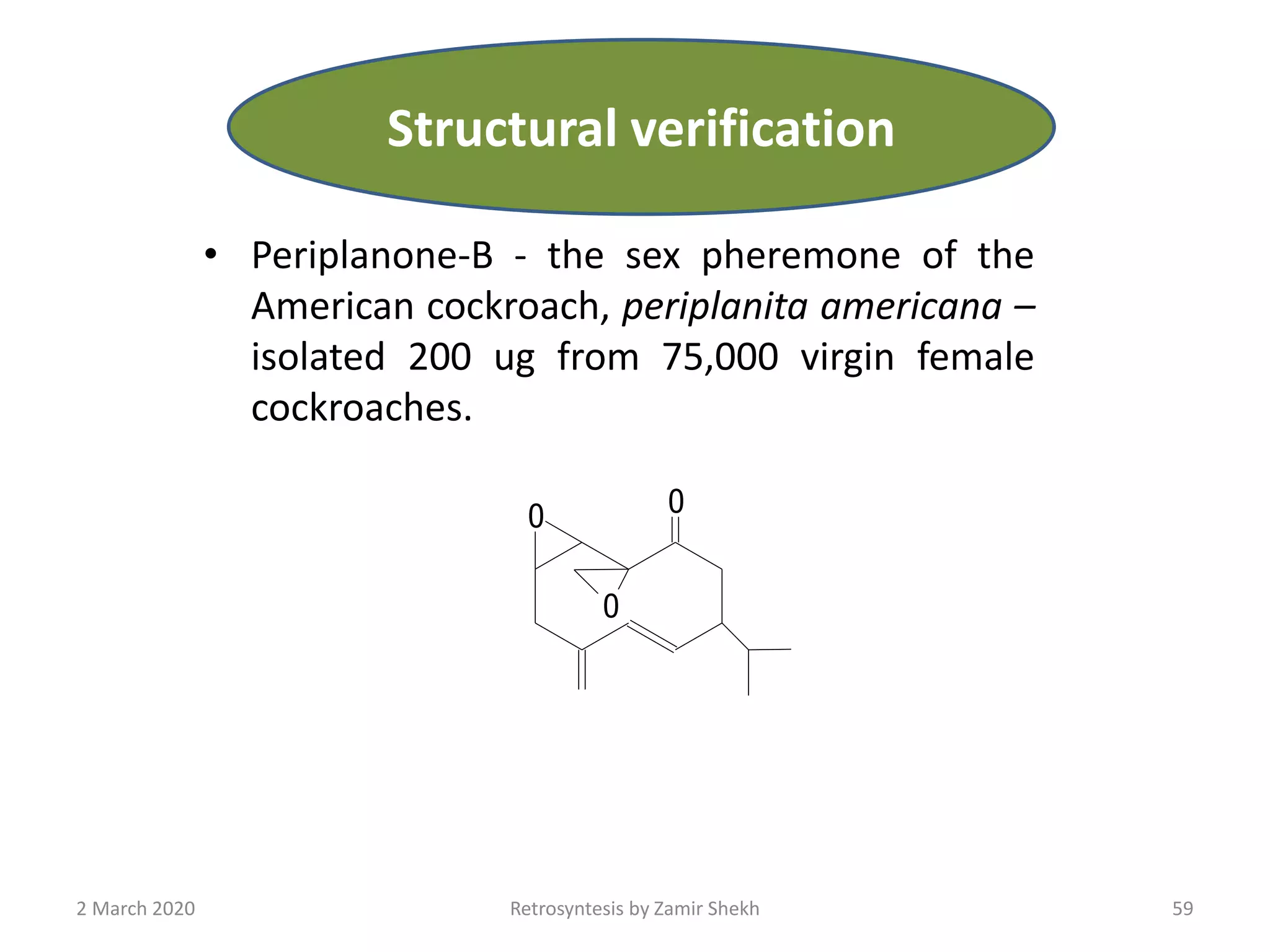
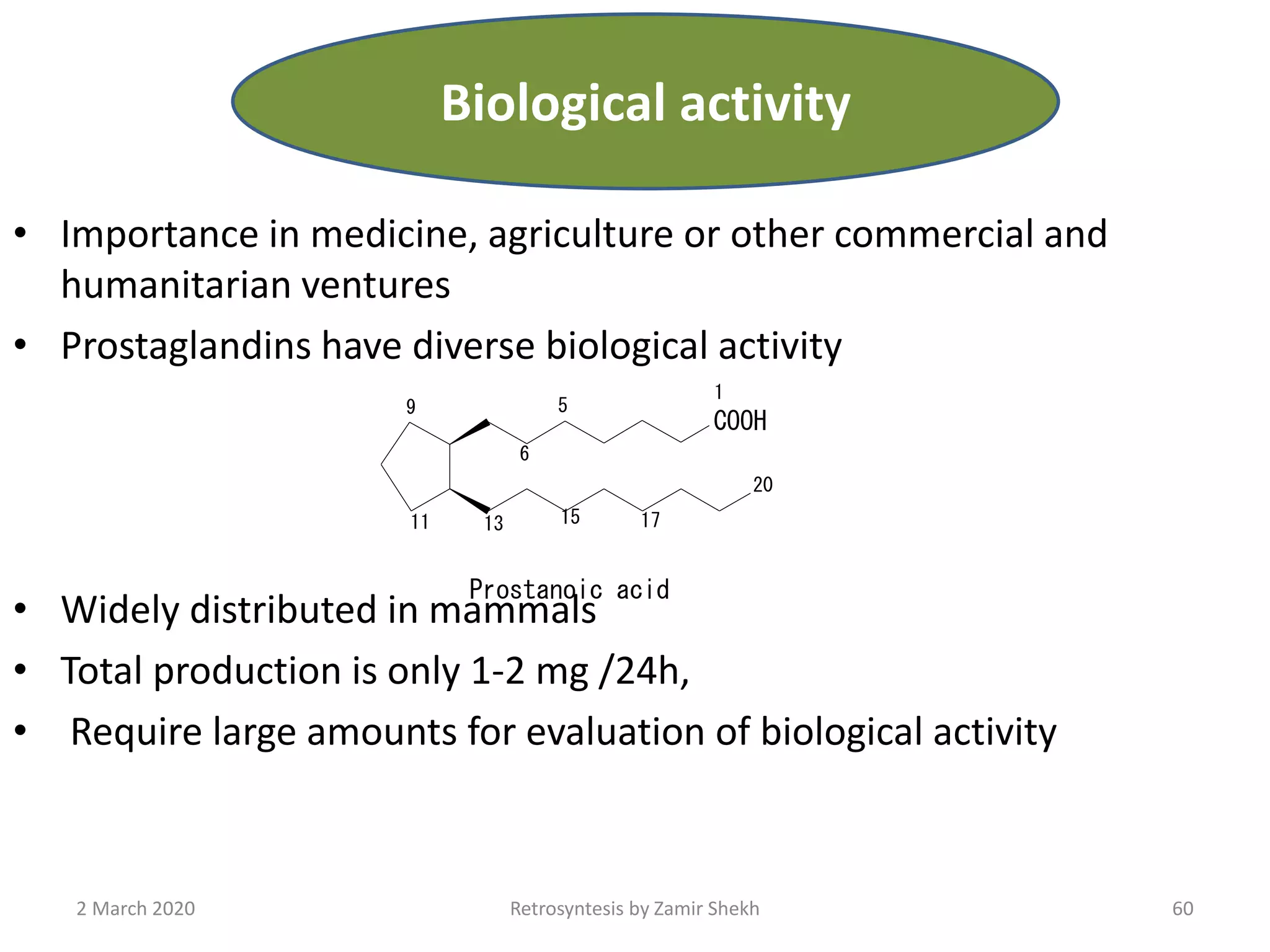
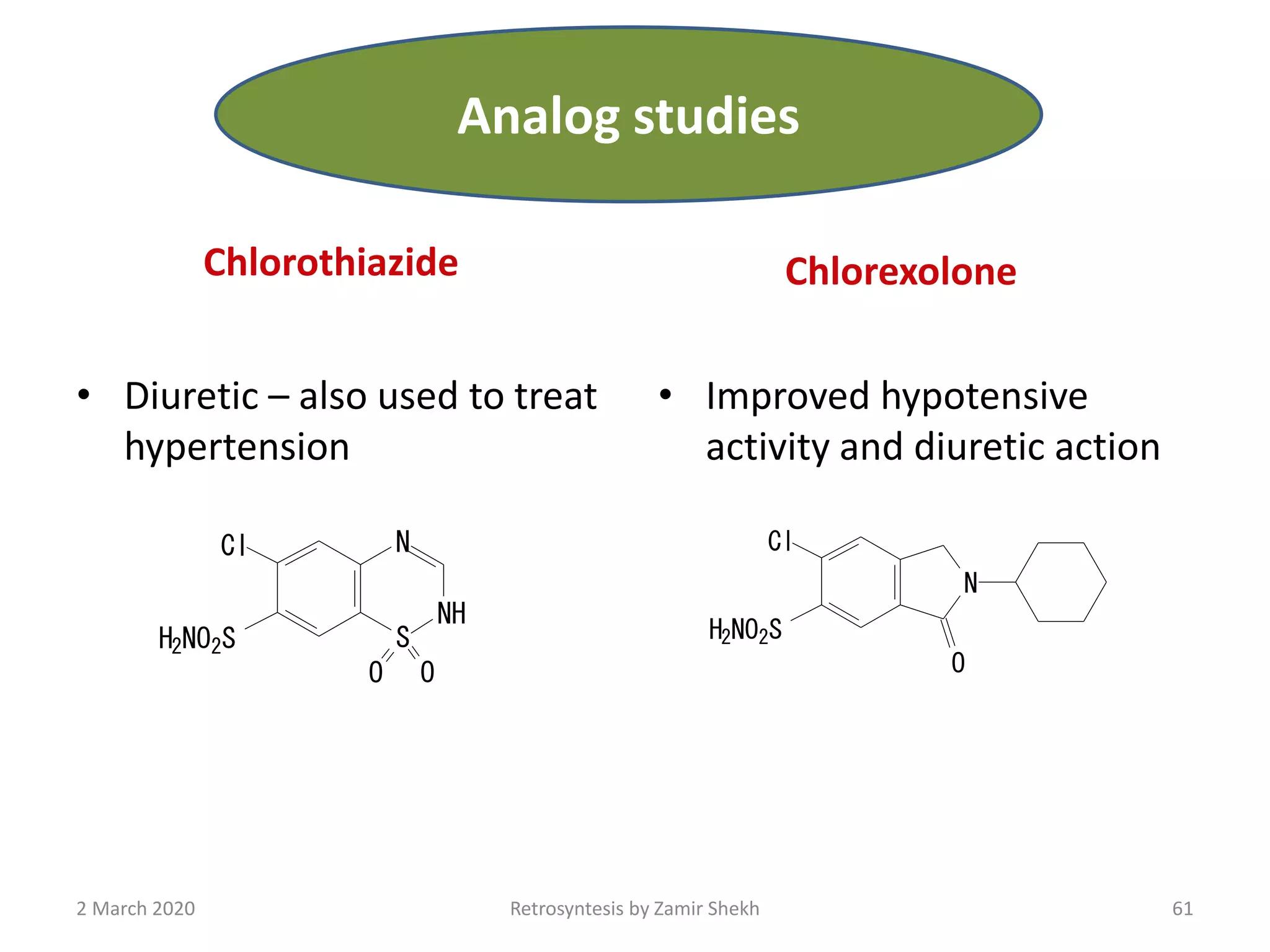
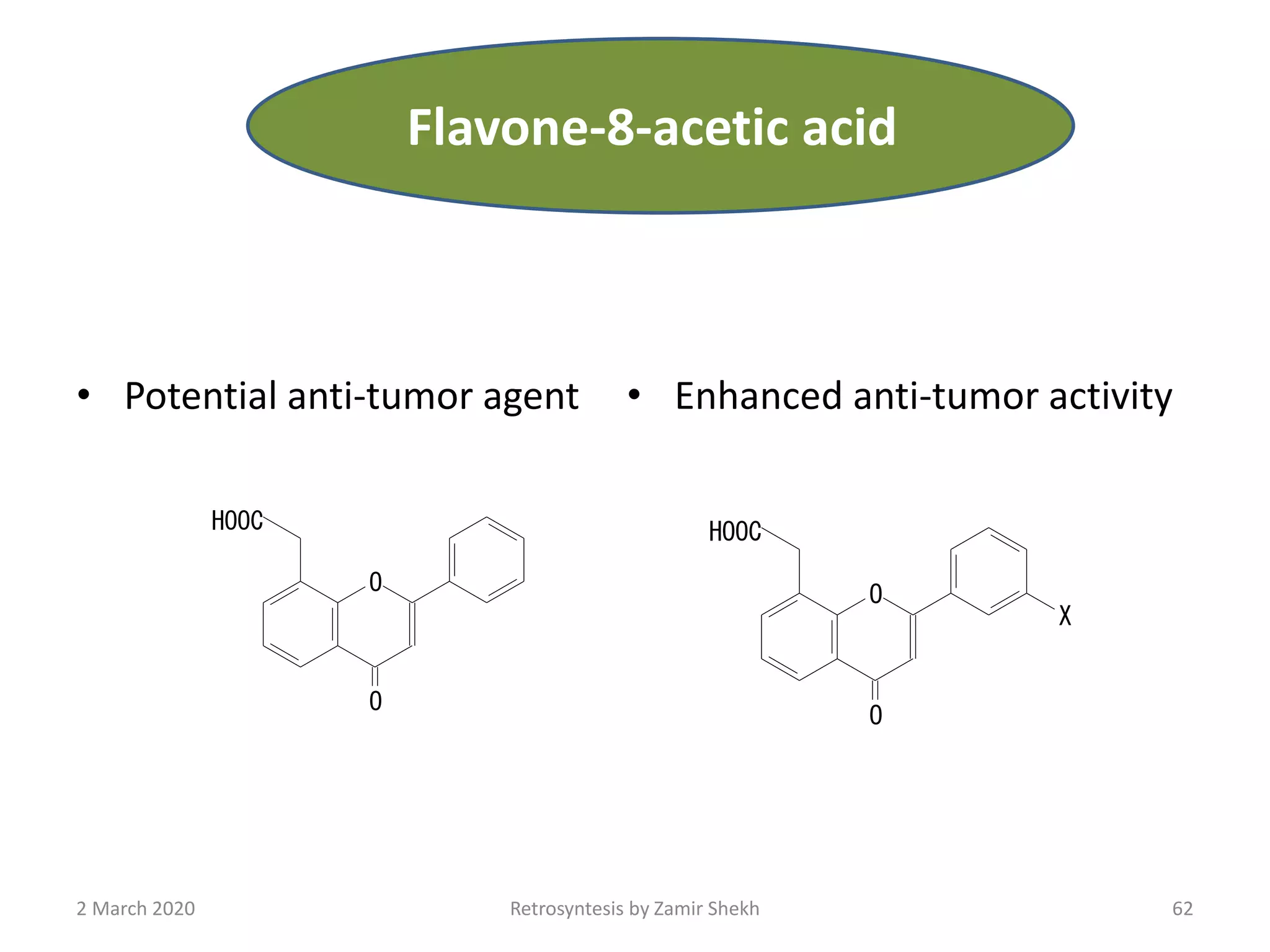
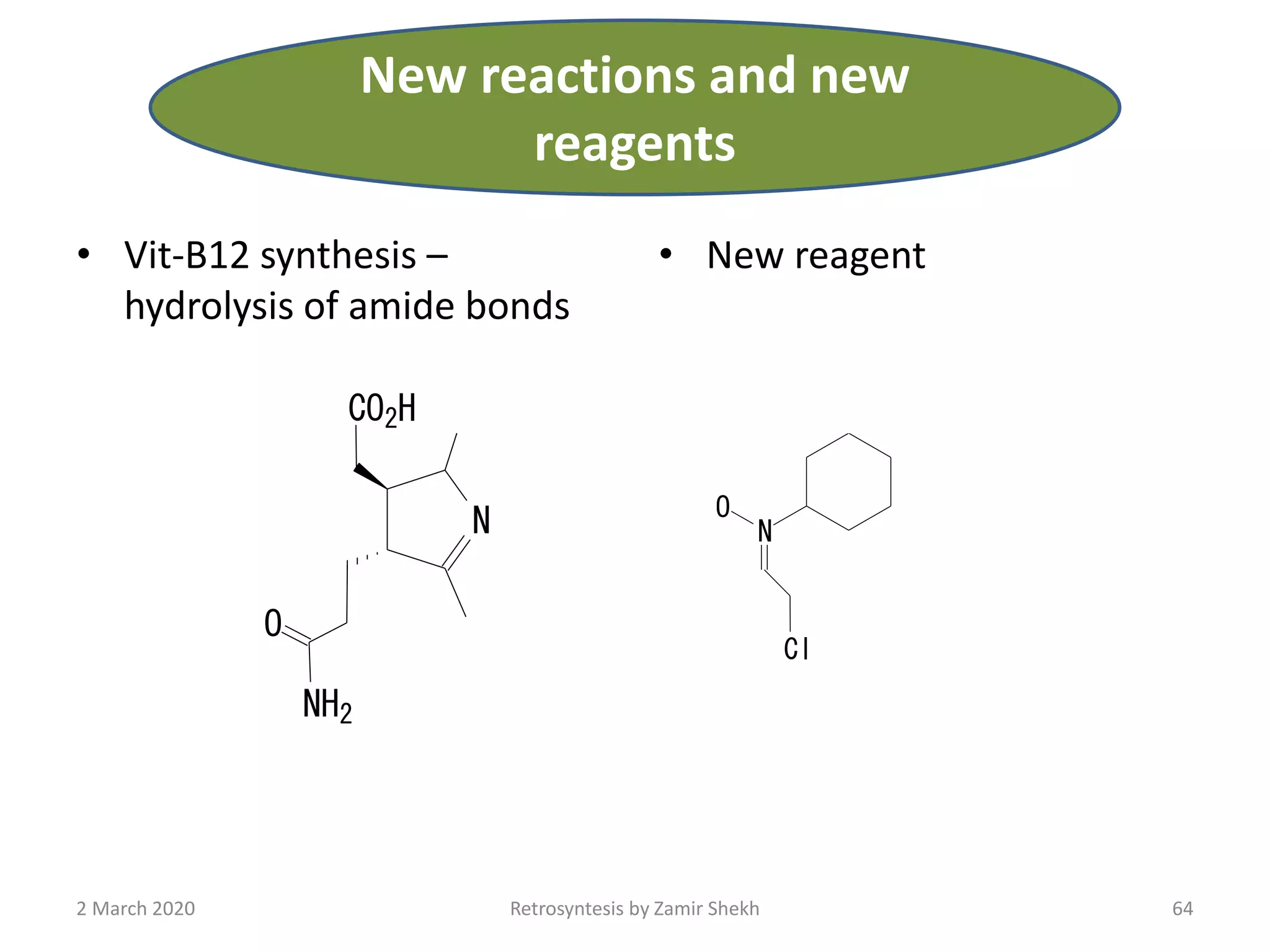
The document presents a comprehensive overview of retrosynthetic analysis in organic chemistry, detailing techniques for designing synthesis based on target molecules. It covers various concepts such as disconnections, synthons, and the strategic planning of synthesis, highlighting classifications of functional groups and synthetic routes. Additionally, the document explores the importance of retrosynthetic principles in achieving efficient and effective syntheses for complex organic compounds.