This document outlines a reservoir rock laboratory course, including:
1) The course covers theoretical and laboratory studies of physical rock properties including porosity, permeability, compressibility, and fluid flow.
2) Students will complete class projects, laboratory experiments, and exams to assess their understanding of reservoir properties and fluid flow.
3) The goal is for students to understand factors that affect fluid flow within porous media and be able to characterize reservoir rocks through laboratory measurements and calculations.



































































































































![Gas Expansion Method (General)
Many porosimeters are designed to use the
principle of Boyle’s law of gas expansion to
determine the grain volume.
The idea is to allow the remaining volume of a chamber
in which a core is placed (V1 – Vg) at pressure P1 to
expand by an additional volume V2 and read the final
pressure P2.
From Boyle’s Law (at constant temperature).
(V1 – Vg) P1 = (V1 – Vg + V2) P2
knowing V1, V2, P1 and P2 allows the calculation of grain
volume Vg.
Vg = V1 – [(P2 / (P1 – P2)] V2
Fall 14 H. AlamiNia Reservoir Rock Laboratory Course (2nd Ed.) 136](https://image.slidesharecdn.com/rplbt5u3qruxgyfxdg6x-signature-d9e691f50924050a5574af6cb5a87f49cf8ea1b64d07db8b8ce172e0273b2b58-poli-150131005539-conversion-gate02/85/Q931-rrl-reference-en-le-cs-135-320.jpg)
![The helium porosimeter
The helium porosimeter uses the principle of gas
expansion, as described by Boyle’s law.
A known volume (reference cell volume) of helium gas
[V2], at a predetermined pressure [PA], is isothermally
expanded into a sample chamber.
After expansion,
the resultant equilibrium pressure is measured [PB].
This pressure depends on the volume of the sample
chamber [V1] minus the rock grain volume [Vg], and
then the porosity can be calculated.
PA*V2=PB*(V2+V1-Vg)
Vg=V1+V2-(PA/PB)V2=V1-[(PA-PB)/PB]V2
Fall 14 H. AlamiNia Reservoir Rock Laboratory Course (2nd Ed.) 137](https://image.slidesharecdn.com/rplbt5u3qruxgyfxdg6x-signature-d9e691f50924050a5574af6cb5a87f49cf8ea1b64d07db8b8ce172e0273b2b58-poli-150131005539-conversion-gate02/85/Q931-rrl-reference-en-le-cs-136-320.jpg)










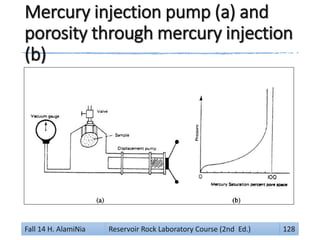
![Gas Expansion Method:
by mercury pump (with a vacuum)
The mercury pump (with a vacuum) gauge is used.
After the bulk volume is determined and mercury
fills the chamber but does not penetrate the sample,
the air in the pores is allowed to expand
by withdrawing the mercury from the chamber.
If the volume of mercury withdrawn is V which is read
on the pump scale then from Boyle’s Law:
Vp P1 = (Vp + V) P2 So: Vp = V[(P2 / (P1 – P2)]
• P1 is initial pressure (atmospheric)
• P2 is the final pressure read on the vacuum gauge
It is clear that if P2 = ½ P1 then Vp = V
• So the pore volume would be equal to the volume of mercury
withdrawn from the chamber to reduce the pressure in the
chamber to half its original (atmospheric) value.
Fall 14 H. AlamiNia Reservoir Rock Laboratory Course (2nd Ed.) 148](https://image.slidesharecdn.com/rplbt5u3qruxgyfxdg6x-signature-d9e691f50924050a5574af6cb5a87f49cf8ea1b64d07db8b8ce172e0273b2b58-poli-150131005539-conversion-gate02/85/Q931-rrl-reference-en-le-cs-147-320.jpg)










